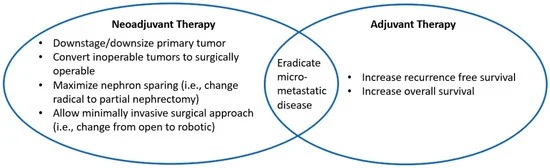
Immunotherapy is rapidly reshaping the treatment landscape of renal cancer. Once reserved mainly for metastatic disease, immune checkpoint inhibitors are now being explored across the entire disease continuum—from neoadjuvant and adjuvant settings to innovative combinations with targeted therapy, microbiome modulation, and novel biological agents. Researchers are testing strategies that integrate surgery, systemic therapy, and emerging biological mechanisms to enhance tumor control and extend survival.
Across ongoing trials, investigators are combining checkpoint inhibitors with HIF-2α inhibitors, tyrosine kinase inhibitors, microbiome-based therapies, and even stress-response pathway modulators. At the same time, studies are revisiting long-standing surgical questions, including the role of cytoreductive nephrectomy or local tumor treatment in the immunotherapy era. From biologically driven neoadjuvant approaches such as casdatifan with or without PD-1 blockade to innovative concepts like microbiome-enhanced immunotherapy or ISR pathway activation, these strategies aim to deepen immune responses and improve outcomes.
The goal is not only to control metastatic disease but also to transform the perioperative management of kidney cancer—reducing tumor burden before surgery, optimizing systemic therapy afterward, and ultimately improving long-term survival for patients with renal cell carcinoma.
Pre-NEOSHIFT-RCC: Neoadjuvant HIF-2α Inhibition With or Without Immunotherapy in Resectable ccRCC
PRENEOSHIFT-RCC is a Phase II randomized trial evaluating a new neoadjuvant treatment strategy for resectable clear cell renal cell carcinoma (ccRCC). The study investigates whether the HIF-2α inhibitor casdatifan, given alone or combined with the PD-1 inhibitor zimberelimab, can shrink tumors before surgery.
Clear cell RCC is largely driven by abnormalities in the HIF signaling pathway, making HIF-2α inhibition a promising therapeutic approach. While HIF inhibitors have shown activity in advanced disease, their role before surgery has not yet been well studied. This trial explores whether targeting tumor hypoxia, with or without immunotherapy, can improve tumor control prior to nephrectomy.
The study will enroll 32 patients with high-risk, resectable ccRCC. Participants are randomized into two groups:
- Casdatifan alone
- Casdatifan + zimberelimab
Treatment is given for three 21-day cycles before standard nephrectomy. Casdatifan is taken orally once daily, while zimberelimab is administered intravenously on day 1 of each cycle.
The primary endpoint is the tumor size reduction rate, measured before surgery. Secondary endpoints include treatment safety and adverse events.
By combining targeted HIF-2α inhibition with immune checkpoint blockade, this trial aims to test a biologically driven strategy that may enhance tumor response before surgery and help define new perioperative treatment approaches for renal cell carcinoma.
ITALIC-RCC: Primary Tumor Ablation in Metastatic Renal Cell Carcinoma Receiving Immunotherapy
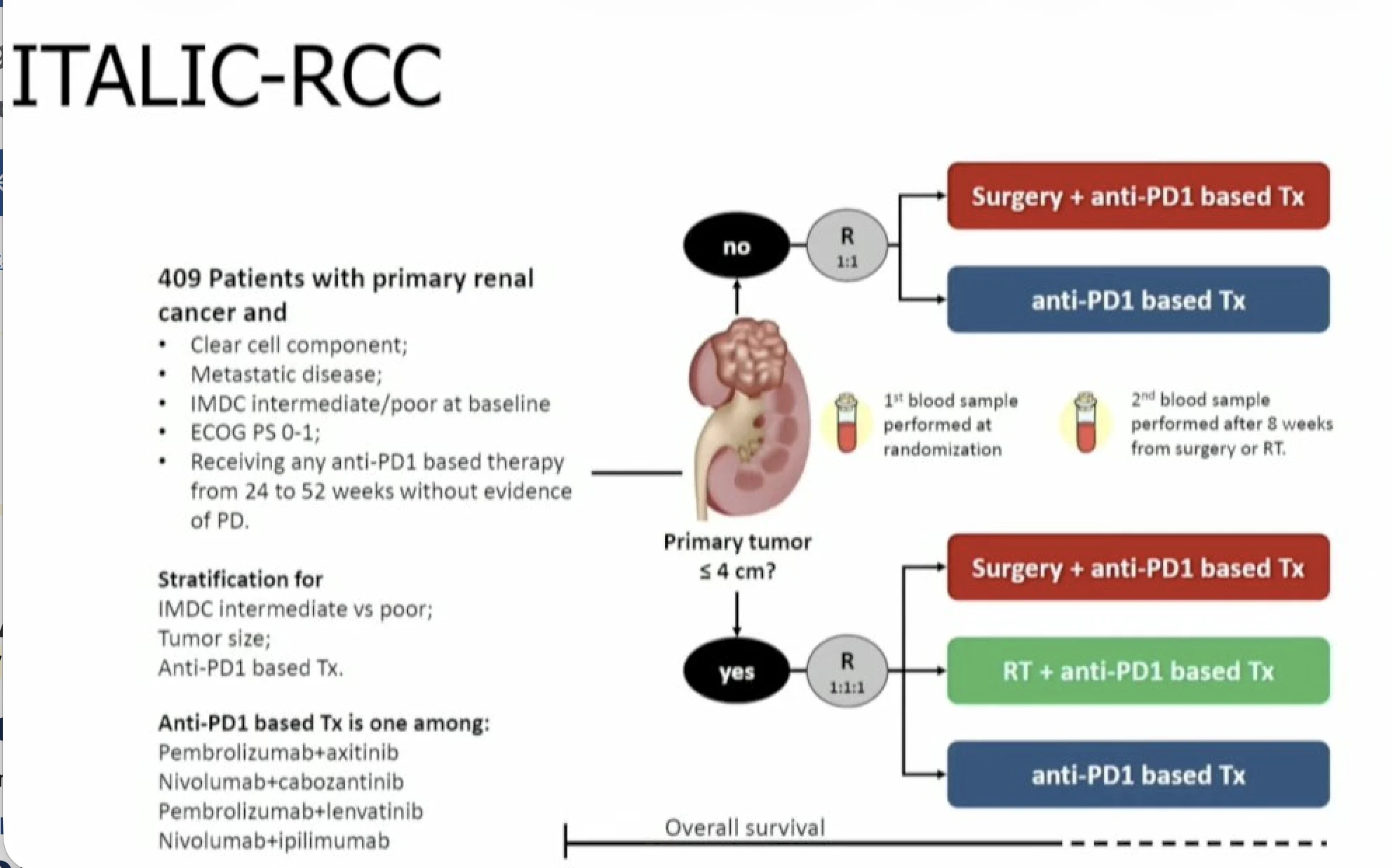
The ITALIC-RCC trial is a Phase IV randomized multicenter study investigating whether treating the primary kidney tumor can improve outcomes in patients with metastatic renal cell carcinoma (mRCC) who are already benefiting from anti–PD-1–based systemic therapy.
Eligible patients with clear cell mRCC who have received 24–52 weeks of immunotherapy-based treatment without disease progression are randomized to evaluate different strategies for managing the primary tumor while continuing systemic therapy.
Patients with primary tumors ≤4 cm are randomized to one of three approaches:
- Cytoreductive nephrectomy plus systemic therapy,
- Radiotherapy to the primary tumor plus systemic therapy, or
- Systemic therapy alone.
Patients with primary tumors >4 cm are randomized to either deferred cytoreductive nephrectomy plus systemic therapy or systemic therapy alone.
Systemic treatment continues with established immunotherapy-based combinations, including axitinib + pembrolizumab, cabozantinib + nivolumab, lenvatinib + pembrolizumab, or nivolumab monotherapy after nivolumab + ipilimumab.
The primary endpoint is 30-month overall survival, with secondary outcomes including progression-free survival, treatment safety, surgical complications, quality of life, and exploratory proteomic biomarkers.
By directly comparing local treatment of the primary tumor versus systemic therapy alone, the ITALIC-RCC study aims to clarify the evolving role of cytoreductive surgery and radiotherapy in the immunotherapy era of metastatic renal cell carcinoma.
PRIMERX Trial: Cytoreductive Nephrectomy in the Era of Immunotherapy for Metastatic RCC
The PRIMERX trial is a randomized prospective study evaluating whether cytoreductive nephrectomy (CN) improves survival in patients with primary metastatic renal cell carcinoma (mRCC) who are receiving modern immune checkpoint inhibitor–based systemic therapy.
Historically, cytoreductive nephrectomy demonstrated clinical benefit when combined with older systemic treatments. However, the treatment landscape for mRCC has evolved significantly, with immunotherapy combinations such as IO–IO or IO–TKI regimens now forming the standard of care. As a result, the role of surgery in this new therapeutic era remains uncertain and requires re-evaluation.
In this study, eligible patients with metastatic clear cell, papillary, or chromophobe RCC who have received first-line immunotherapy combinations and show no metastatic progression after six months of treatment are randomized to either cytoreductive nephrectomy (or other local ablative therapy) or continuation of systemic therapy alone. The trial uses an innovative Trial-Within-Cohort (TWiC) design embedded within the PRO-RCC registry, allowing patients already enrolled in the prospective registry to be randomized efficiently while maintaining real-world clinical relevance.
The primary endpoint is overall survival, while secondary outcomes include long-term quality of life, surgical morbidity, and translational biomarker analyses exploring immune cell infiltration and mechanisms of response or resistance. By reassessing the value of primary tumor removal in the immunotherapy era, the PRIMERX study aims to clarify whether cytoreductive surgery remains beneficial for patients with metastatic RCC treated with modern immune checkpoint inhibitors.
EXACT Trial: Zanzalintinib With or Without Nivolumab After Adjuvant Immunotherapy in Clear Cell RCC
The EXACT trial is a Phase II randomized study evaluating new treatment strategies for patients with advanced or metastatic clear cell renal cell carcinoma (ccRCC) whose disease has progressed after adjuvant PD-1/PD-L1 immunotherapy.
Participants are randomly assigned to one of two treatment groups. One group receives zanzalintinib (XL092) alone, an oral multi-target tyrosine kinase inhibitor taken daily. The second group receives zanzalintinib combined with nivolumab, a PD-1 immune checkpoint inhibitor administered intravenously every four weeks. Treatment is given in 28-day cycles and continues until disease progression or unacceptable toxicity.
The primary endpoint is the overall response rate (ORR) within six months of treatment. Secondary outcomes include progression-free survival, overall survival, safety, and duration of response.
This study aims to determine whether combining targeted therapy with immunotherapy can improve outcomes for patients whose renal cell carcinoma progresses after adjuvant checkpoint inhibitor therapy, a growing clinical challenge in modern RCC management.
PROBE Trial: Immunotherapy With or Without Cytoreductive Nephrectomy in Metastatic Renal Cell Carcinoma
The PROBE trial is a Phase III study investigating whether adding cytoreductive nephrectomy to modern immunotherapy-based treatment improves survival in patients with newly diagnosed metastatic renal cell carcinoma (mRCC).
Today, several immune checkpoint inhibitor–based combinations are used as standard first-line treatment for metastatic RCC. However, the role of removing the primary kidney tumor in patients already receiving these therapies remains unclear.
In this study, patients first receive standard systemic therapy for about 10–14 weeks. After this initial treatment period, they are randomized to one of two strategies:
- Continue immunotherapy-based systemic treatment alone
- Undergo cytoreductive nephrectomy followed by continuation of the same systemic therapy
The systemic treatment regimens used in the study include:
- Nivolumab + Ipilimumab
- Pembrolizumab + Axitinib
- Avelumab + Axitinib
The primary goal of the trial is to determine whether adding surgery improves overall survival. Secondary objectives include evaluating treatment response at metastatic sites, surgical complications, drug-related toxicity, and changes in the size of the primary tumor.
By directly comparing immunotherapy alone versus immunotherapy combined with surgery, the PROBE trial aims to clarify whether cytoreductive nephrectomy still provides benefit in the modern immunotherapy era of metastatic kidney cancer.
SHARK Trial: HC-7366 Combined With Immunotherapy in Clear Cell Renal Cell Carcinoma
The SHARK trial is a Phase 1b study evaluating a novel strategy for metastatic clear cell renal cell carcinoma (ccRCC) by combining HC-7366, a first-in-class integrated stress response (ISR) agonist, with immune checkpoint inhibitors. The study aims to determine whether activating tumor stress pathways with HC-7366 can enhance the effectiveness of immunotherapy.
The trial investigates two treatment approaches:
Doublet regimen
- HC-7366 (oral ISR agonist)
- Nivolumab (PD-1 immune checkpoint inhibitor)
Triplet regimen
- HC-7366
- Nivolumab
- Ipilimumab (CTLA-4 immune checkpoint inhibitor)
The primary objective is to evaluate the safety and tolerability of these combinations by monitoring dose-limiting toxicities and treatment-related adverse events. The study also assesses antitumor activity, including overall response rate (ORR) and disease control rate (DCR) according to RECIST criteria.Secondary endpoints include progression-free survival, overall survival, duration of response, time to response, and the incidence of immune-related adverse events. Patient-reported outcomes and quality of life are also evaluated using validated instruments.
By combining a novel ISR-targeting agent with immune checkpoint blockade, the SHARK study explores a new biological strategy to enhance immune-mediated tumor control in advanced kidney cancer.
EXL01 Trial: Microbiome-Based Therapy Combined With Immunotherapy in Metastatic Renal Cell Carcinoma
This Phase I randomized study investigates whether modifying the gut microbiome can improve the effectiveness of immune checkpoint inhibitors in patients with metastatic renal cell carcinoma (mRCC).
The trial evaluates the addition of EXL01, a live biotherapeutic product containing the bacterium Faecalibacterium prausnitzii, to standard immunotherapy. This bacterium is believed to influence immune activity through changes in the gut microbiome, potentially enhancing responses to checkpoint blockade.
Patients are randomized into two treatment arms:
Arm A – Immunotherapy + Microbiome therapy
- Nivolumab (PD-1 inhibitor)
- Ipilimumab (CTLA-4 inhibitor)
- EXL01 (oral live bacterial therapy)
Arm B – Standard immunotherapy
- Nivolumab
- Ipilimumab
During the first four treatment cycles, nivolumab and ipilimumab are administered every three weeks, with EXL01 taken orally once daily in Arm A. After these induction cycles, patients continue nivolumab maintenance therapy, with or without EXL01 depending on the assigned arm.
The primary objective is to evaluate how EXL01 affects systemic immune modulation when combined with checkpoint inhibitors. Secondary objectives include assessing safety, tolerability, and potential improvements in clinical response.
Exploratory analyses focus on changes in gut microbiome diversity and immune signaling, aiming to better understand how microbiome-based therapies may enhance immunotherapy responses in kidney cancer.
CBM588 With Nivolumab and Ipilimumab in Advanced Renal Cell Carcinoma
This Phase I open-label study is evaluating whether CBM588, a live biotherapeutic containing Clostridium butyricum MIYAIRI 588, can be safely combined with nivolumab plus ipilimumab in patients with advanced or metastatic renal cell carcinoma.
CBM588 is being studied as a microbiome-directed therapy that may improve the activity of immune checkpoint inhibitors by altering the composition and function of the gut microbiome. Because response to immunotherapy may be influenced by intestinal bacteria, this trial explores whether adding CBM588 can enhance the effects of standard dual immunotherapy in kidney cancer.
Patients receive:
- CBM588 orally twice daily
- Nivolumab intravenously
- Ipilimumab intravenously
Treatment is given in 21-day cycles for the first 4 cycles, followed by nivolumab maintenance plus CBM588 in 28-day cycles, continued until disease progression or unacceptable toxicity.
The primary objective is to determine the safety, tolerability, and best dose of CBM588 in combination with nivolumab and ipilimumab. Secondary and exploratory objectives include evaluating:
- Clinical efficacy of the combination
- Changes in the gut microbiome
- Effects on metabolic pathways
- Systemic immune modulation
This study represents a growing area of research in renal cell carcinoma: using live biotherapeutics and microbiome modulation to potentially improve outcomes with checkpoint blockade.
Bevacizumab, Erlotinib, and Atezolizumab in HLRCC-Associated and Sporadic Papillary Renal Cell Carcinoma
This Phase II study is evaluating a triple combination of bevacizumab, erlotinib, and atezolizumab in patients with advanced hereditary leiomyomatosis and renal cell cancer (HLRCC)-associated RCC and sporadic papillary renal cell carcinoma.
The rationale is biologically strong.
- Bevacizumab targets tumor angiogenesis by blocking VEGF.
- Erlotinib inhibits EGFR signaling, which may help slow tumor growth.
- Atezolizumab is a PD-L1 inhibitor designed to enhance immune-mediated antitumor activity.
Together, this regimen is being tested to determine whether combined antiangiogenic therapy, EGFR inhibition, and immunotherapy can improve disease control in these aggressive non-clear cell kidney cancer subtypes.
Patients receive:
- Bevacizumab IV on day 1
- Atezolizumab IV on day 1
- Erlotinib orally once daily on days 1–21
Treatment is given in 21-day cycles and continues until progression or unacceptable toxicity.
The primary objective is to assess the complete response rate by RECIST 1.1. Secondary endpoints include:
- Objective response rate (ORR)
- Disease control rate (DCR)
- Progression-free survival (PFS)
- Overall survival (OS)
- Duration of response
- Safety and tolerability
The trial also includes important translational analyses, exploring:
- immune-cell changes in blood and tumor tissue,
- cytokine profiles,
- PD-1/PD-L1 expression,
- and genomic alterations such as FH and NRF2 pathway abnormalities.
This study is especially important because HLRCC-associated RCC and papillary RCC remain difficult-to-treat kidney cancer subtypes, and more effective biology-driven approaches are urgently needed.
NEOPAX: Neoadjuvant Pembrolizumab and Axitinib in RCC With Inferior Vena Cava Tumor Thrombus
The NEOPAX trial is a Phase II study exploring whether neoadjuvant pembrolizumab plus axitinib can reduce the burden of inferior vena cava (IVC) tumor thrombus in patients with renal cell carcinoma before surgery.
This is an important question because RCC with IVC tumor thrombus is often surgically complex. If preoperative treatment can shrink the thrombus or lower its level of extension, surgery may become safer and less morbid, while potentially improving long-term outcomes.
Patients receive:
- Pembrolizumab 200 mg IV every 3 weeks
- Axitinib 5 mg orally twice daily, with dose escalation if tolerated
Treatment is given for 12 weeks (4 cycles), followed by repeat imaging and then definitive surgery within about 2 weeks.
The main goal of the study is to evaluate whether this regimen changes the extent and size of the IVC tumor thrombus, measured by MRI using the Mayo classification and thrombus dimensions. Secondary endpoints include:
- Surgical complications
- Treatment-related adverse events
- 1-year progression-free survival
- 1-year overall survival
The study also includes patient-reported kidney cancer outcomes, using FKSI-DRS and FKSI-19 questionnaires during neoadjuvant treatment.