Locally advanced pancreatic cancer remains one of the most difficult settings in gastrointestinal oncology, where curative surgery is often not possible at diagnosis and long-term outcomes remain limited despite multimodal treatment.
A phase II study published in Nature Communications evaluated a strategy that combined a GM-CSF-secreting allogeneic pancreatic cancer vaccine (GVAX), pembrolizumab and stereotactic body radiation therapy (SBRT) after induction chemotherapy in patients with locally advanced pancreatic cancer. The results suggest that this approach may improve outcomes in selected patients, particularly those who ultimately undergo surgical resection.
Title: ” The combination of a cancer vaccine, pembrolizumab, and stereotactic body radiation in patients with locally advanced pancreatic cancer: a single-arm, phase II study ” 27 January, 2026
Authors: Valerie Lee, Apoorvaa S. Sachidanand, Christina Rodriguez, Junke Wang, Beth Onners, Hanfei Qi, Rose Parkinson, Thomas McPhaul, Jennifer Durham, Ding Ding, Amy Hacker-Prietz, Amol K. Narang, Dung T. Le, Ana De Jesus, Michael Pishvaian, Eric Christenson, Elizabeth D. Thompson, Robert A. Anders, Arsen Osipov, Matthew Weiss, William Burns, Richard Burkhart, Kelly Lafaro, Jin He, Christopher L. Wolfgang, Elizabeth Sugar, Joseph M. Herman, Elizabeth M. Jaffee, Hao Wang, Daniel A. Laheru & Lei Zheng.
Why This Study Matters
Pancreatic ductal adenocarcinoma continues to carry a poor prognosis, and around 30% of newly diagnosed cases present as locally advanced disease. In this setting, tumors are considered surgically inoperable because of vascular involvement, and standard management typically includes induction chemotherapy followed by radiation, including SBRT. Even so, increasing the rate of successful resection and improving long-term disease control remain major clinical challenges.
Immunotherapy has transformed treatment in several malignancies, but pancreatic cancer has largely remained resistant to checkpoint blockade. The study authors note that the pancreatic tumor microenvironment is strongly immunosuppressive and poorly infiltrated by effector T cells, which may partly explain the limited activity of immune checkpoint inhibitors in this disease.
Radiation therapy may help overcome part of this resistance by acting as an in situ vaccine, and prior work with GVAX suggested that vaccination could enhance immune cell infiltration in pancreatic tumors. This trial was designed to test whether combining GVAX, pembrolizumab, and SBRT could produce meaningful clinical benefit in locally advanced pancreatic cancer.
A Triple-Modality Strategy After Chemotherapy
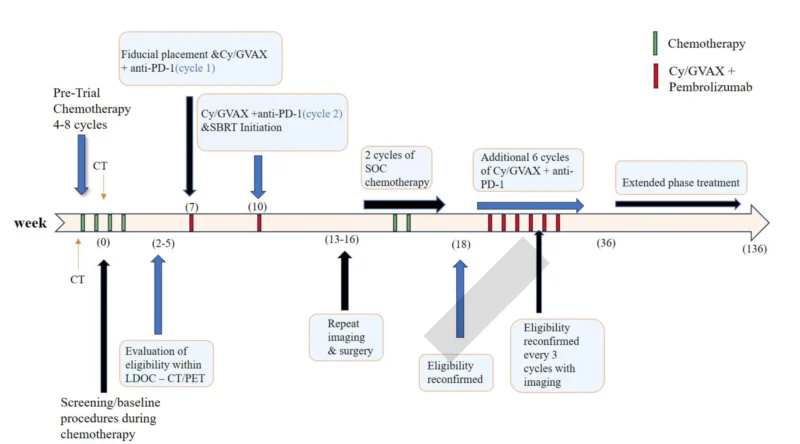
This was a single-center, single-arm, phase II trial conducted at Johns Hopkins Hospital in patients with locally advanced pancreatic cancer. Eligible patients had no metastatic disease after at least 4 and no more than 8 cycles of FOLFIRINOX or gemcitabine/nab-paclitaxel–based chemotherapy.
They then received cyclophosphamide, pembrolizumab, GVAX, and SBRT before reassessment for surgery. Patients deemed potentially resectable underwent surgical exploration. Those without metastatic disease could then continue standard chemotherapy followed by maintenance immunotherapy.
The study enrolled 58 patients between July 18, 2016, and December 14, 2020. Of these, 54 were evaluable for efficacy after receiving two cycles of the combination treatment and one follow-up scan. The primary endpoint was distant metastasis-free survival, while secondary endpoints included overall survival, surgical resectability, and pathologic response.
Resectability Emerged As A Key Turning Point
Among the 54 evaluable patients, 35 patients, or 64.8%, were considered potentially resectable after treatment and underwent exploratory laparotomy. Ultimately, 24 patients underwent R0 or R1 resection, corresponding to an overall resection rate of 44.4%. Among those resected, 91.7% achieved R0 resections, indicating a high rate of margin-negative surgery in the resected subgroup. The rate of resectability did not differ according to whether induction chemotherapy had been FOLFIRINOX or gemcitabine/nab-paclitaxel.
This distinction between resected and unresected disease proved central to the study’s outcome analysis. Patients who underwent surgery appeared to derive the greatest benefit from the treatment strategy, while outcomes remained substantially poorer in those whose disease could not be resected despite exploration.
Survival Signals Were Most Pronounced In Resected Patients
At a median follow-up of 19.6 months, median distant metastasis-free survival for all evaluable patients was 9.8 months, which was the primary endpoint of the study. Among patients who underwent R0 or R1 resection, median distant metastasis-free survival reached 20.3 months. By contrast, patients who were explored but not resected had a median distant metastasis-free survival of only 2.4 months.
Median overall survival from the start of immunotherapy was 21.8 months in the full evaluable cohort. In the resected group, median overall survival was 29.7 months, compared with 12.1 months in patients who underwent surgical exploration but not resection. When calculated from the time of initial diagnosis, median overall survival was 28.2 months in all evaluable patients, 36.7 months in the resected group, and 19.7 months in the unresected group.
The timing analysis also showed that immunotherapy did not appear to delay surgery. Median time from the start of immunotherapy to surgical exploration was 75.5 days, and the median time from the end of immunotherapy to surgical exploration was 53 days.
Radiotherapy for Pancreatic Cancer: Types, Success Rate, Side Effects And More
Comparison With Historical Controls Adds Context
To better understand the treatment effect, investigators compared the trial population with a historical control cohort of patients from the Johns Hopkins Pancreatic Cancer Registry who met the same eligibility criteria and had received neoadjuvant chemotherapy followed by SBRT.
Among surgically explored patients, median distant metastasis-free survival was significantly longer in the trial cohort than in the historical control group, at 10.2 versus 9.5 months, with a hazard ratio of 0.34. Among resected patients, median distant metastasis-free survival was 20.3 months in the trial cohort versus 10.2 months in the historical control cohort, with a hazard ratio of 0.40.
Overall survival comparisons were more modest. In surgically explored patients, median overall survival from the start of study treatment was 27.9 months in the trial cohort versus 26.0 months in the historical control group, and from diagnosis it was 35.6 versus 33.1 months. These differences did not reach statistical significance, although the study authors noted a numerical trend favoring the investigational strategy, especially among resected patients.
Safety Was Manageable
The regimen demonstrated a manageable safety profile. All patients experienced grade 1 or 2 vaccine injection-site reactions. Grade 3 or 4 treatment-related adverse events occurred in 12% of patients and were mainly immune-related. These included pneumonitis, diabetic ketoacidosis related to immune-mediated pancreatitis, abdominal pain and colitis, elevated liver enzymes, nephritis, and rash. Elevated liver enzymes were not associated with clinical hepatitis. Importantly, the study reported no surgery delays due to treatment-related adverse events and no unexpected postoperative complications.
What The Findings Suggest
The trial did not meet its primary goal of prolonging median distant metastasis-free survival to 13.6 months in the entire cohort. However, the data suggest that this radioimmunotherapy approach may be particularly relevant for patients who can ultimately proceed to surgical resection. In this subgroup, both distant metastasis-free survival and overall survival appeared more favorable, and the improvement in distant metastasis-free survival compared with historical controls was notable.
At the same time, the study highlights a continuing unmet need for patients who remain unresectable after neoadjuvant treatment. The authors suggest that while the combination may help control micrometastatic disease in patients with minimal residual tumor burden, it appears less effective in patients with persistent unresected tumors. They point to tumor-associated macrophages as a possible contributor to resistance and suggest that future treatment strategies may need to incorporate macrophage-targeting approaches.
A Step Forward, With Important Limits
This was a single-arm study conducted at a single center, and the authors acknowledge the possibility of selection bias, since enrolled patients had already completed several months of induction chemotherapy without developing metastatic disease.
Although the historical comparison helps provide context, randomized studies will be needed to determine whether this strategy can produce a definitive survival benefit and to identify which patients are most likely to benefit.
Still, this phase II study adds to growing interest in combining radiation, vaccination, and immune checkpoint blockade in pancreatic cancer. In a disease where therapeutic advances remain difficult to achieve, the observed resection outcomes and prolonged distant metastasis-free survival in resected patients offer an important signal for further investigation.
Discover more articles like this on OncoDaily.
Written by Nare Hovhannisyan, MD



