The treatment landscape of squamous non-small cell lung cancer (sqNSCLC) continues to evolve, yet durable responses remain limited for many patients. A new development from Henlius signals a potential shift toward biologically driven combination strategies, integrating EGFR targeting with immunotherapy.
On April 24, 2026, Henlius announced that its Phase 2/3 trial (HLX07-sqNSCLC-301) evaluating pimurutamab (HLX07) in combination with serplulimab and chemotherapy has successfully completed Clinical Trial Notification (CTN) in Australia—marking a key step in global clinical expansion.
Non-Small Cell Lung Cancer: Causes, Symptoms, Diagnosis, Treatment Options
Why EGFR Still Matters in sqNSCLC
Despite major advances with PD-1/PD-L1 inhibitors, sqNSCLC remains a disease with:
- High recurrence rates
- Limited long-term survival
- Few biomarker-driven strategies
Unlike non-squamous NSCLC, EGFR mutations are rare in squamous disease. However, EGFR overexpression is extremely common, approaching ~90% in sqNSCLC.
This distinction is critical:
- EGFR mutation → targetable with TKIs
- EGFR overexpression → not effectively addressed by TKIs, but potentially targetable with monoclonal antibodies
This creates a large, underexploited therapeutic population, where EGFR-directed antibodies could play a role—especially in combination with immunotherapy.
The Strategy: Dual Targeting of Tumor Growth and Immune Evasion
This investigational regimen is built on a simple but powerful concept: target the tumor directly while simultaneously activating the immune system.
The combination brings together three complementary components:
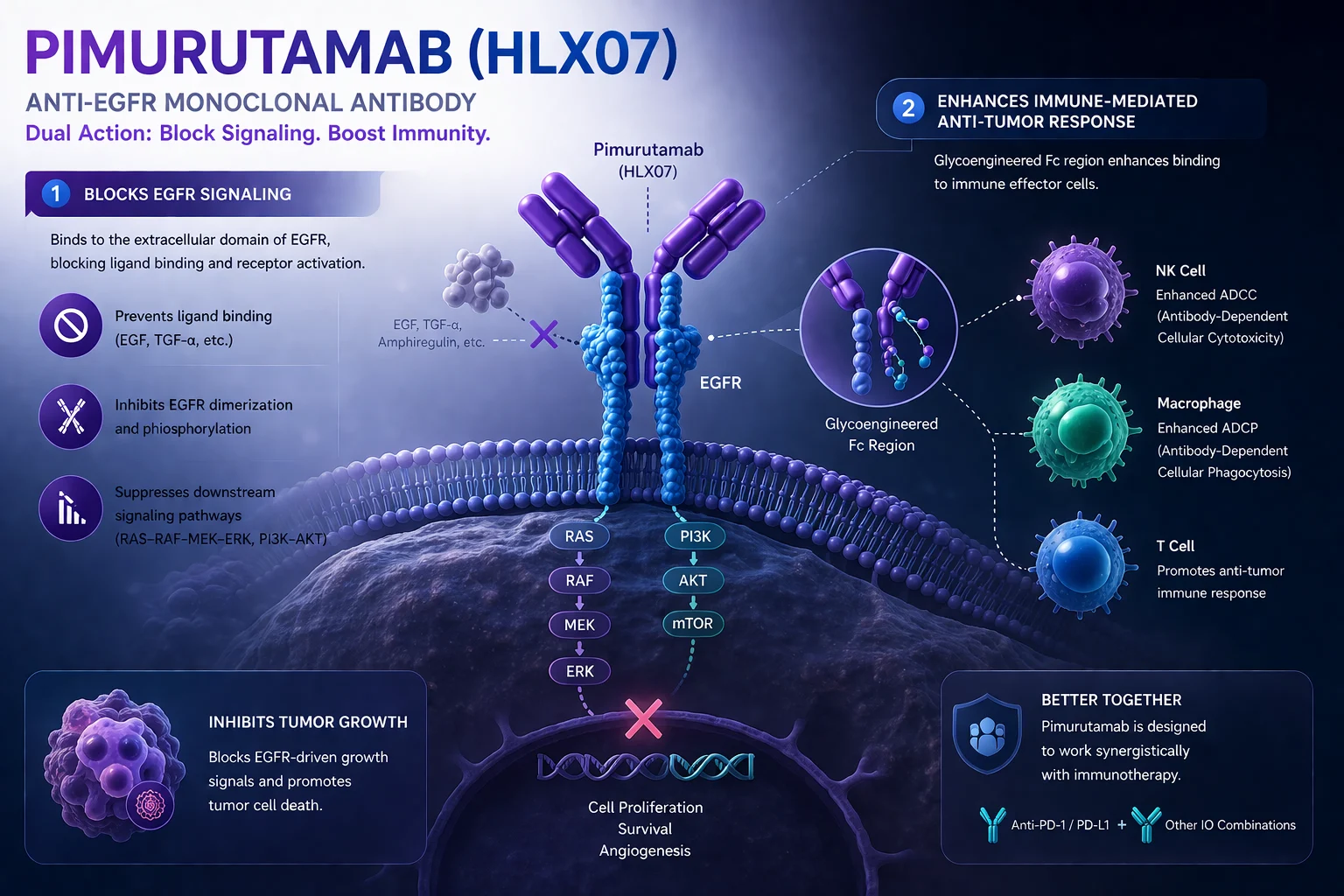
- Pimurutamab (HLX07) – an anti-EGFR monoclonal antibody
- Serplulimab – an anti-PD-1 immune checkpoint inhibitor
- Chemotherapy – the cytotoxic backbone
Rather than acting independently, these therapies are designed to work together across different layers of tumor biology.
At the tumor level, EGFR blockade interferes with key signaling pathways that drive cancer cell proliferation and survival. At the same time, PD-1 inhibition helps restore T-cell activity, allowing the immune system to recognize and attack tumor cells more effectively. Chemotherapy adds a third dimension by inducing tumor cell death, increasing antigen release, and enhancing immune priming.
In essence, the strategy aims to:
- Directly suppress tumor growth through EGFR inhibition
- Reinvigorate anti-tumor immunity via PD-1 blockade
- Amplify immune recognition through chemotherapy-induced antigen release
Preclinical findings suggest that this is not merely an additive approach, but a synergistic one, where each component enhances the activity of the others—potentially translating into deeper and more durable clinical responses.
Phase 2/3 Trial Design (HLX07-sqNSCLC-301)
The HLX07-sqNSCLC-301 study is designed as a randomized, double-blind, multicenter Phase 2/3 trial, aiming to rigorously evaluate whether adding pimurutamab to an immunotherapy–chemotherapy backbone can improve outcomes in the first-line treatment of advanced sqNSCLC.
The study follows a two-part, adaptive design, allowing early signal detection and dose optimization before moving into confirmatory evaluation.
Phase 2: Dose Selection and Signal Detection
The first part of the study focuses on identifying the optimal dose of pimurutamab and confirming early clinical activity.
Participants are randomized in a 1:1:1 ratio into three treatment groups:
- High-dose pimurutamab + serplulimab + chemotherapy
- Low-dose pimurutamab + serplulimab + chemotherapy
- Placebo + serplulimab + chemotherapy
All regimens are administered on a 3-week cycle (Q3W) for induction, followed by maintenance therapy with pimurutamab (or placebo) plus serplulimab.
The primary endpoints are designed to capture both early efficacy and response dynamics:
- Progression-free survival (PFS)
- Objective response rate (ORR)
This phase will determine not only whether the combination is active, but also which dose provides the best balance of efficacy and tolerability for further development.
Phase 3: Confirmatory Efficacy Evaluation
Once the optimal dose is selected, the trial transitions into a Phase 3 confirmatory stage, directly comparing the investigational strategy with current standard approaches.
Patients are randomized in a 1:1 ratio into:
Experimental arm:
- Pimurutamab + serplulimab + chemotherapy
- Followed by maintenance pimurutamab + serplulimab
Control arm:
- Placebo + pembrolizumab + chemotherapy
- Followed by maintenance pembrolizumab
This design is particularly relevant because it benchmarks the regimen against a PD-1–based standard of care, rather than chemotherapy alone.
The primary endpoints reflect long-term clinical benefit:
- Progression-free survival (PFS) (assessed by blinded independent central review)
- Overall survival (OS)
Biomarker Strategy: Toward Precision Selection
A key strength of the trial is its integrated biomarker exploration, aiming to better understand which patients derive the greatest benefit.
The study will evaluate:
- EGFR expression levels in tumor tissue
- PD-L1 status
- The interaction between EGFR and PD-L1 biology and treatment response
This approach reflects a broader shift toward biology-driven treatment strategies, moving beyond one-dimensional biomarkers and exploring how multiple signaling and immune pathways intersect.
Early Clinical Signal (Phase 2)
Data from the Phase 2 study (HLX07-sqNSCLC-201) provide the rationale for Phase 3 development:
- Objective response rate (ORR): ~70%
- Disease control rate (DCR): >90%
- Median PFS: up to 17.4 months (high-dose cohort)
- Durability: responses remained stable with extended follow-up (~23.5 months)
- Safety: manageable, no new safety signals
These results are notable when compared with historical chemo-immunotherapy benchmarks in sqNSCLC, suggesting potential incremental benefit beyond current standards.
Why This Matters
This study is not just another combination trial—it reflects a broader shift in how we approach sqNSCLC.
Over the past decade, chemo-immunotherapy built around PD-1/PD-L1 inhibitors has become the standard of care. However, despite these advances, outcomes have started to plateau, with many patients still experiencing progression.
What makes this strategy different is that it moves beyond simply adding immunotherapy to chemotherapy. Instead, it introduces a biologically driven combination, targeting both tumor signaling and immune evasion.
At its core, this approach represents three important shifts:
- Beyond PD-1–based combinations
While PD-1 inhibitors remain central, there is growing recognition that additional mechanisms are needed to further improve outcomes. - Revisiting EGFR in a new way
Rather than focusing on rare EGFR mutations, this strategy targets EGFR overexpression, a far more common feature in sqNSCLC—potentially expanding relevance to a much broader patient population. - Toward precision combination therapy
The future may not rely on a single biomarker, but on integrated strategies, such as:dual-pathway targeting (EGFR + PD-1)
combining tumor biology with immune modulation
What Doctors May Be Thinking
From a clinical perspective, this study raises several important and very practical questions:
- Can EGFR overexpression evolve into a clinically useful predictive biomarker in sqNSCLC?
- Will this combination meaningfully outperform current PD-1 + chemotherapy standards?
- Is any potential efficacy gain balanced by toxicity, cost, and real-world feasibility?
- Could this approach ultimately reshape first-line treatment algorithms?
You Can Read Full Article Here


