Grade group 5 (GG5) prostate cancer represents one of the most aggressive disease subsets, associated with high rates of biochemical recurrence and metastatic progression despite standard-of-care (SOC) treatment. While androgen deprivation therapy (ADT) combined with radiotherapy—particularly high-dose rate brachytherapy (HDRBT)—has improved outcomes, long-term disease control remains suboptimal.
Given the historically limited activity of immune checkpoint inhibitors in prostate cancer, this study explored a biologically timed combination strategy. By integrating Nivolumab during the immunogenic window induced by ablative radiation and ADT, investigators aimed to enhance immune priming and improve disease control in high-risk GG5 patients.
Study Design and Methods
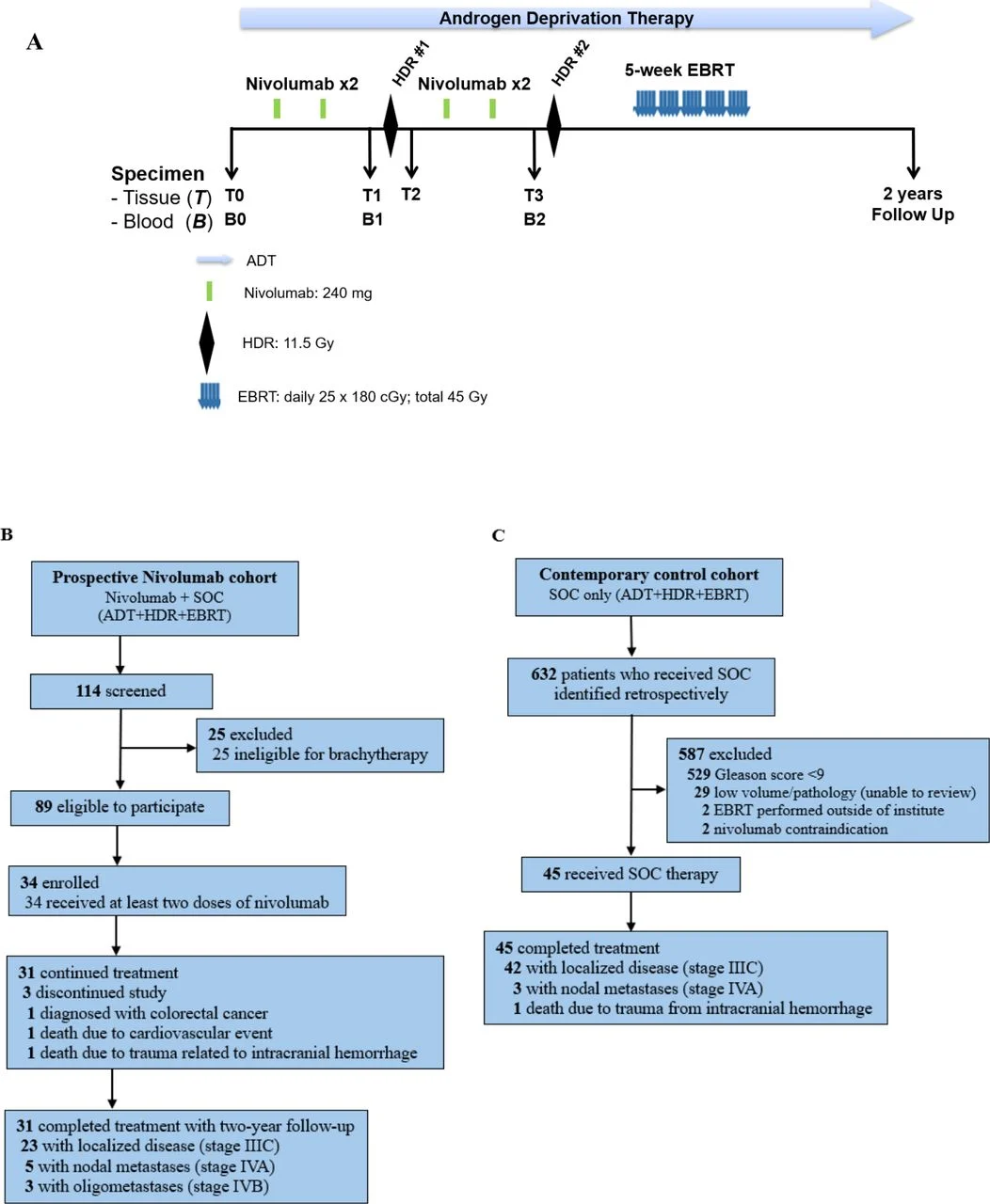
This was a single-arm, phase II trial enrolling patients with localized or oligometastatic GG5 prostate cancer with high tumor burden. The treatment strategy was intentionally multimodal, combining immune, hormonal, and radiotherapeutic mechanisms.
Patients received neoadjuvant and concurrent nivolumab (four doses, initiated prior to radiation), alongside long-course ADT and HDRBT followed by external beam radiation therapy (EBRT). The sequencing was designed to maximize immune activation—nivolumab was introduced before and during radiation to exploit antigen release and T-cell priming.
The primary endpoint was 2-year freedom from biochemical recurrence (FFBR), benchmarked against a historical control rate of 75%. Secondary and exploratory endpoints included toxicity, pathological response, and biomarker analyses, particularly genomic immune signatures.
Results
The addition of nivolumab to SOC demonstrated encouraging clinical activity in this high-risk population, exceeding predefined efficacy thresholds:
- 2-year FFBR: 90.3% (vs historical 75%, p=0.024)
- Median FFBR: not reached at median follow-up of ~38.8 months
- Biochemical recurrence: 19.3% of patients
- Metastatic progression: 19.3%, with failures often systemic
- Grade ≥3 nivolumab-related toxicity: ~6.3%, with no grade 4+ events
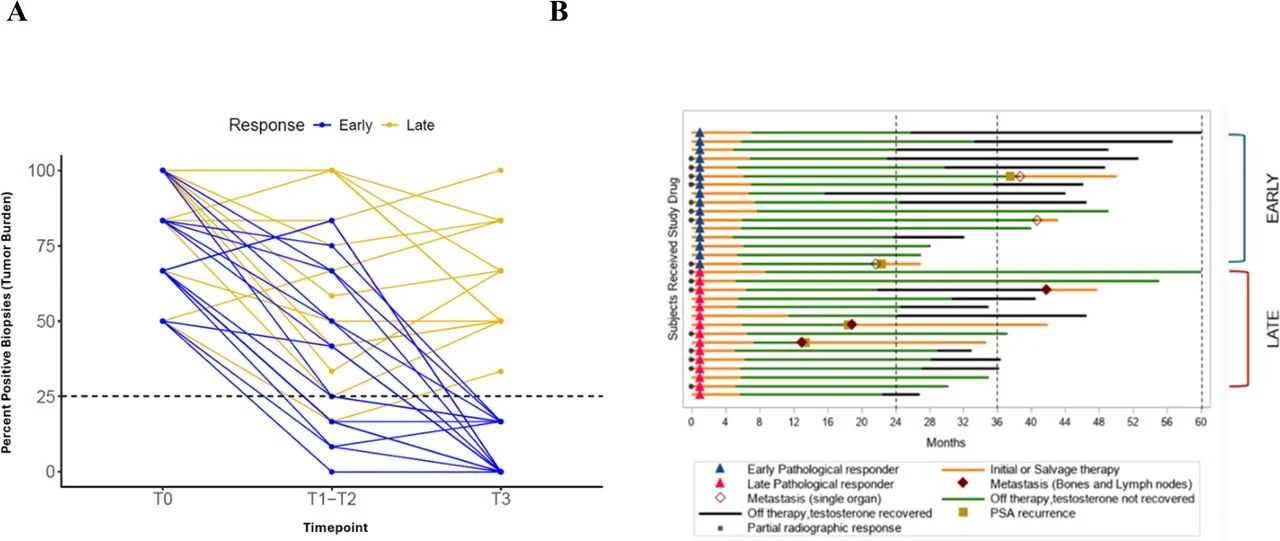
Pathological and radiologic responses further supported treatment activity. Approximately half of patients achieved early pathological response, which correlated with improved clinical outcomes and lower metastatic burden at relapse.
Importantly, early responders demonstrated distinct tumor biology, with higher baseline expression of immune-related genes such as CD8A, LAG3, STING1, and IFNG, suggesting a pre-existing immune-active microenvironment.
What These Results Mean
This study provides evidence that prostate cancer—traditionally considered immunologically “cold”—can be sensitized to checkpoint inhibition when combined with radiation and hormonal therapy in a biologically optimized sequence.
The observed FFBR improvement suggests that immunotherapy may enhance not only local tumor control but also delay systemic dissemination. The integration of nivolumab appears to amplify radiation-induced immune activation, potentially converting local therapy into a systemic anticancer response.
A particularly important finding is the identification of the Ricketts Immunosuppression Score (RIS) as a predictive biomarker. Higher RIS scores were associated with early pathological response and longer time to metastatic progression, supporting the concept that immune-enriched tumors—despite expressing inhibitory signals—may derive the greatest benefit from checkpoint blockade.
Why This Matters
This trial reflects a broader shift toward immuno-radiotherapeutic intensification in high-risk solid tumors. Rather than relying solely on prolonged androgen suppression or cytotoxic therapy, this strategy leverages:
- Radiation-induced antigen release and immune priming
- ADT-mediated immune modulation
- Checkpoint inhibition to overcome adaptive resistance
Clinically, this approach may represent an alternative or complementary strategy to androgen receptor–targeted intensification (e.g., abiraterone-based regimens), offering a shorter but potentially more immunologically durable intervention.
However, important limitations remain. The study was non-randomized, with a small sample size and reliance on historical controls. While the signal is strong, confirmation in randomized trials is essential before redefining treatment standards.
Key Takeaway Message
The integration of nivolumab with HDR brachytherapy, EBRT, and ADT achieved a 2-year biochemical control rate of over 90% in a highly aggressive GG5 prostate cancer population, with manageable toxicity.
This study suggests that timed immunotherapy combined with ablative radiation may overcome intrinsic resistance in prostate cancer, marking a step toward biologically driven, multimodal treatment strategies.
Read Full Article Here


