Dual checkpoint blockade with nivolumab and ipilimumab has transformed metastatic melanoma, producing durable responses and long-term survival in a subset of patients. However, its clinical use is limited by substantial immune-related toxicity, particularly with systemic anti-CTLA-4 exposure.
The NIVIPIT trial explored a biologically rational alternative: delivering low-dose ipilimumab directly into tumor lesions while maintaining systemic nivolumab. The goal was to preserve local immune activation and antitumor efficacy, while reducing systemic toxicity.
Study Design and Methods
NIVIPIT was a randomized, multicenter phase 1b trial enrolling patients with previously untreated metastatic melanoma. Patients were randomized 2:1 to receive intravenous nivolumab combined with either intratumoral low-dose ipilimumab or standard intravenous ipilimumab.
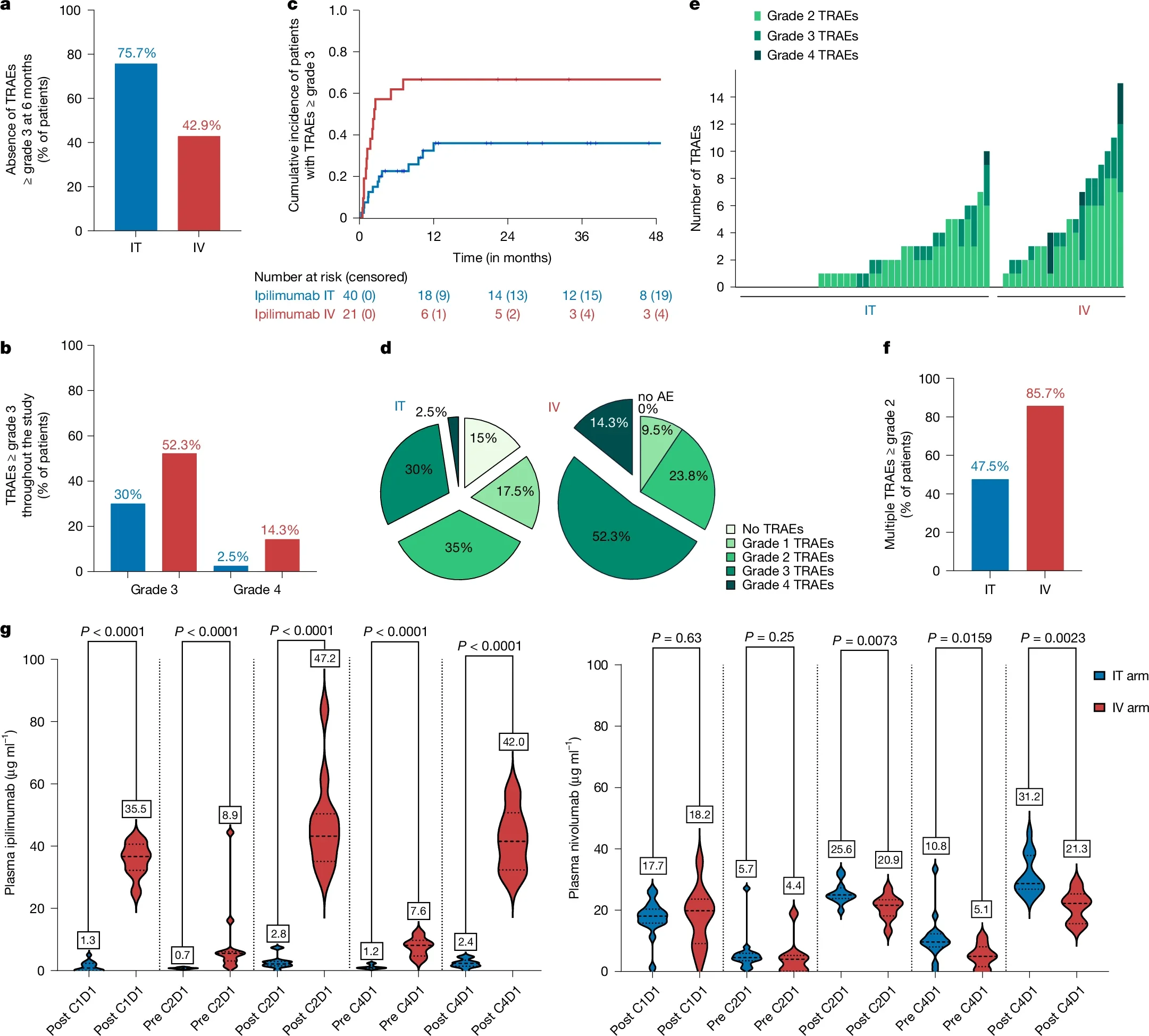
The intratumoral approach used a tenfold lower ipilimumab dose, aiming to create high local drug exposure within the tumor microenvironment while minimizing systemic circulation. The primary endpoint was safety, specifically grade 3–4 treatment-related adverse events at 6 months. The trial also included extensive pharmacokinetic, pharmacodynamic, and tumor immune profiling analyses.
Key Results
The trial met its primary endpoint, showing that intratumoral anti-CTLA-4 substantially reduced severe toxicity while maintaining clinically relevant activity.
- Grade 3–4 treatment-related adverse events at 6 months: 22.6% with intratumoral ipilimumab vs 57.1% with intravenous ipilimumab
- Cumulative grade 3–4 toxicity: 32.5% vs 66.6%
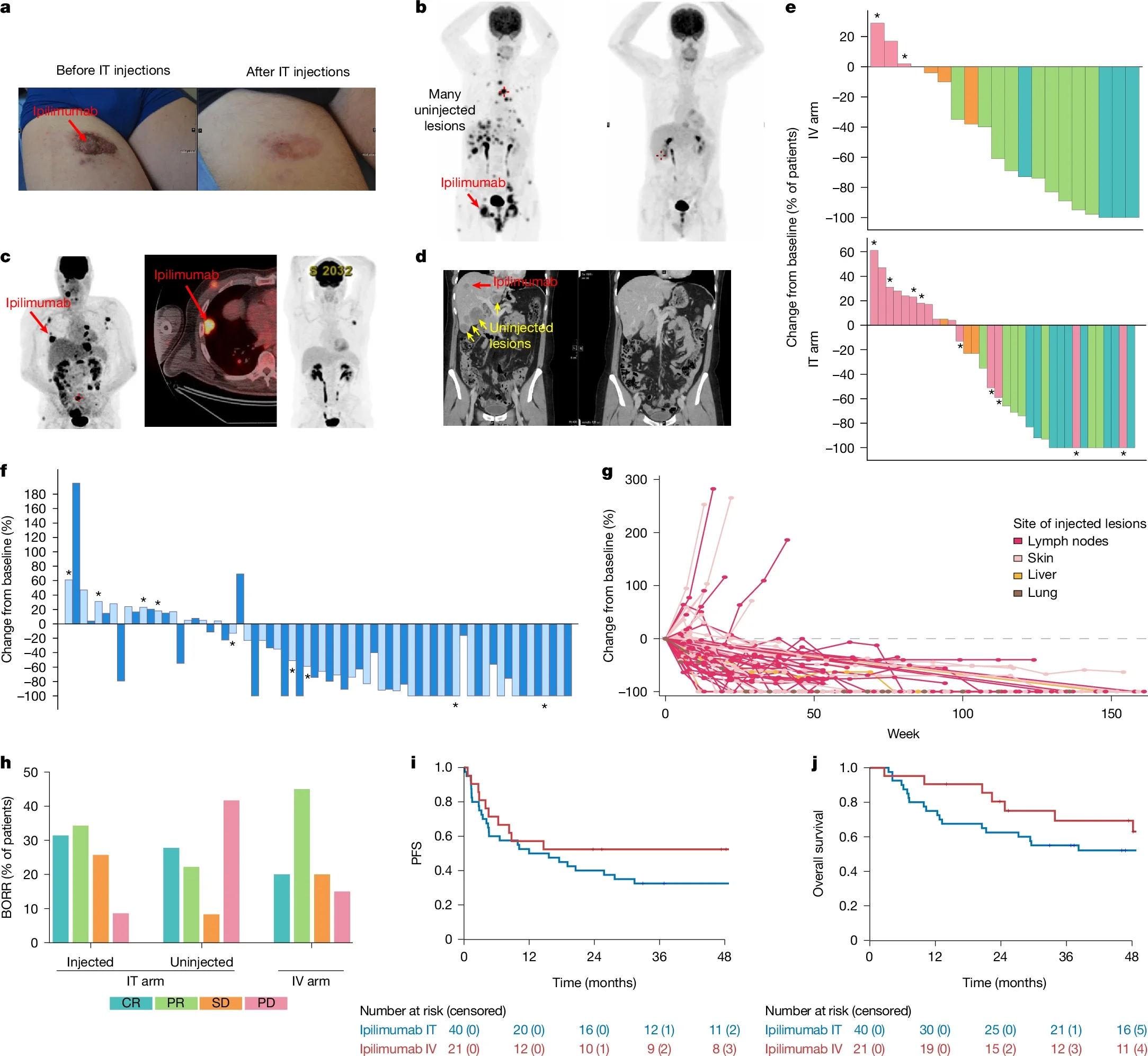
- Response rate in injected lesions: 65.7%
- Response rate in uninjected lesions: 50%
- Median follow-up: 55.5 months, with no significant PFS or OS difference between arms
Pharmacokinetic analysis confirmed markedly lower systemic ipilimumab exposure in the intratumoral arm, supporting the hypothesis that local delivery can reduce systemic immune toxicity.
Scientific Interpretation
This study is important because it shows that anti-CTLA-4 activity may not require high systemic exposure. By concentrating ipilimumab inside the tumor, the investigators achieved strong local responses and evidence of systemic antitumor activity in distant uninjected lesions.
The response in uninjected lesions suggests an abscopal-like systemic immune effect, likely mediated by local immune priming, remodeling of tumor-draining lymph nodes, and limited systemic diffusion of ipilimumab.
The translational findings are especially compelling. Durable clinical benefit was associated with a pre-existing immune-active tumor microenvironment, including MHC-I and MHC-II expression, CD8+PD-1+ T cells, TH1 CD4+ signatures, B-cell involvement, and tertiary lymphoid structure–like features.
Unexpectedly, tumors that benefited also had higher baseline activated Treg cells and M2 macrophages. Rather than being purely negative prognostic markers, these populations may indicate an immune microenvironment actively shaped by immunoediting and therefore vulnerable to CTLA-4–mediated remodeling.
Why This Matters
The NIVIPIT trial offers a clinically relevant strategy to separate the efficacy of CTLA-4 blockade from its systemic toxicity. This is particularly important because severe immune-related adverse events remain one of the main barriers to broader use of nivolumab–ipilimumab combinations.
The study suggests that intratumoral anti-CTLA-4 may be especially valuable in settings where local disease control and immune priming are central, such as:
- oligometastatic disease
- neoadjuvant immunotherapy
- injectable melanoma lesions
- tumors with immune-active but suppressive microenvironments
This approach may also provide a model for future immunotherapy design: not only choosing the right drug, but delivering it to the right immune compartment.



