Locally advanced gastric and gastroesophageal junction cancers remain challenging despite curative-intent surgery and perioperative chemotherapy. Even with effective chemotherapy backbones such as SOX, CapOx, or FLOT-based regimens, relapse—particularly distant and peritoneal recurrence, continues to limit long-term survival.
The NEOSUMMIT-01 trial addresses an important question in this setting: can adding PD-1 blockade to perioperative chemotherapy improve not only pathologic response, but also long-term clinical outcomes?
Toripalimab, an anti–PD-1 antibody, was evaluated in combination with perioperative chemotherapy in patients with cT3–4aN+M0 gastric or gastroesophageal junction cancer. Earlier results showed improved pathologic regression. This 3-year follow-up now demonstrates that this pathologic benefit translated into improved survival outcomes.
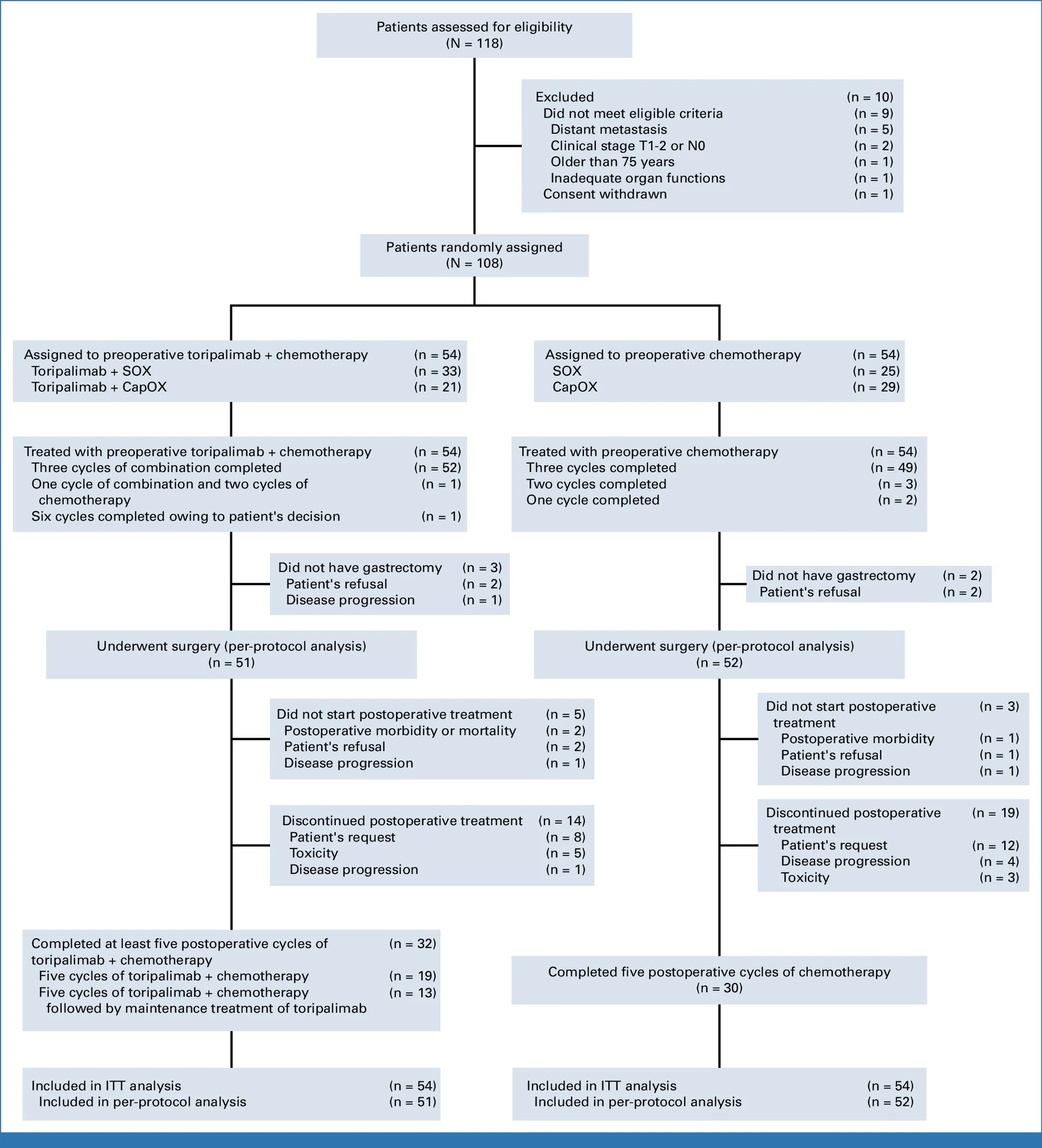
Study Design and Methods
NEOSUMMIT-01 was a randomized phase II trial enrolling 108 patients with locally advanced gastric or gastroesophageal junction cancer.
Patients were assigned to receive either perioperative toripalimab plus chemotherapy or chemotherapy alone. The chemotherapy backbone consisted of investigator-selected SOX or CapOx. Treatment included preoperative therapy, surgery, and postoperative treatment, with toripalimab continued for up to 6 months depending on patient condition.
The main long-term endpoints in this update were event-free survival and overall survival after a median follow-up of 43.2 months.
Results
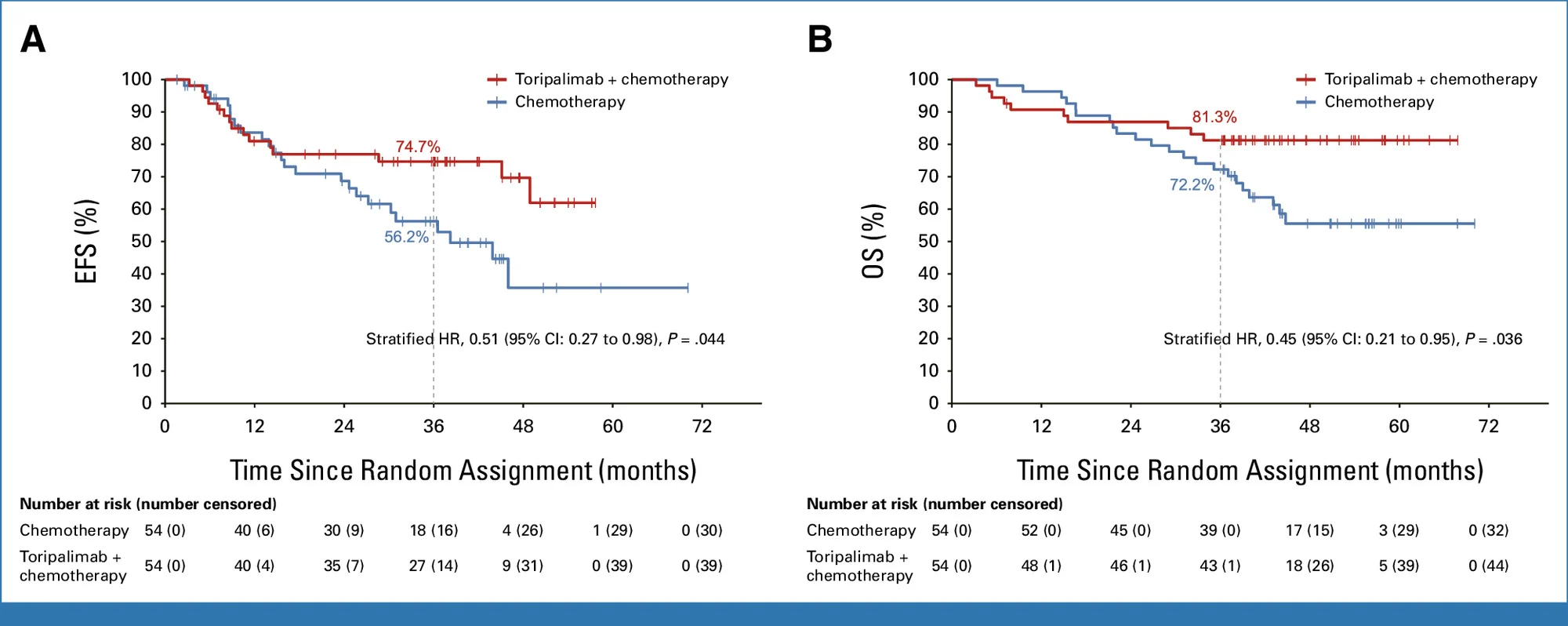
The 3-year analysis demonstrated clinically meaningful and statistically significant survival improvement with the addition of toripalimab.
- 3-year event-free survival: 74.7% with toripalimab plus chemotherapy vs 56.2% with chemotherapy alone
- Event-free survival hazard ratio: HR 0.51; P = .044
- 3-year overall survival: 81.3% vs 72.2%
- Overall survival hazard ratio: HR 0.45; P = .036
- Total documented relapse within 3 years: 20.0% vs 47.4%, with peritoneal metastasis reduced from 36.8% to 15.0%
These findings suggest that the addition of toripalimab not only improves tumor regression before surgery but may also reduce clinically relevant recurrence, particularly peritoneal relapse.
Scientific Interpretation
The most important message from NEOSUMMIT-01 is that perioperative PD-1 blockade can translate pathologic response into survival benefit in localized gastric and GEJ cancer. This is especially relevant because not all perioperative immunotherapy trials in gastric cancer have shown consistent survival improvement.
The persistence of benefit after excluding patients with dMMR tumors is also important. Since dMMR/MSI-H tumors are highly immunotherapy-sensitive, the maintained benefit in the broader population suggests that toripalimab plus chemotherapy may have activity beyond the small MSI-H subgroup.
The reduction in peritoneal metastasis is particularly interesting. Peritoneal recurrence remains one of the most difficult patterns of failure in gastric cancer, and standard chemotherapy has limited ability to fully prevent it. If confirmed, this finding may become one of the most clinically meaningful aspects of the trial.
Why This Matters
NEOSUMMIT-01 supports a growing shift in gastric cancer treatment: perioperative therapy is moving from chemotherapy alone toward immune-chemotherapy strategies designed to improve systemic control before and after surgery.
The trial also strengthens the rationale for oxaliplatin-based chemotherapy as a partner for immunotherapy in localized gastric/GEJ cancer, especially in Asian treatment settings where SOX and CapOx are widely used.
However, these data should still be interpreted carefully. This was a randomized phase II trial with a modest sample size, and the survival endpoints were part of long-term follow-up rather than the original primary pathologic endpoint. Larger phase III validation will be important before defining this as a universal standard.
Write Full Article Here


