Mycosis Fungoides (MF), despite its historic name, is neither infectious nor a fungal disease, rather, it represents a chronic lymphoproliferative malignancy with highly variable biological behavior. MF is the most common clinicopathologic subtype of Cutaneous T-Cell Lymphoma (CTCL), a heterogeneous group of extranodal non-Hodgkin lymphomas.
Because MF often mimics common skin conditions and is frequently misdiagnosed, recognizing its different stages, and knowing when management shifts from dermatology to hematology can improve early diagnosis and support more effective treatment decisions.
The Origins of MF
Genetic data support ultraviolet radiation as a potential risk factor for CTCL, while epidemiologic studies suggest an association with environmental toxin exposure. Skin barrier dysfunction and microbiome alterations are relevant contributors. Rare familial cases of MF, together with the presence of specific HLA class II alleles in both sporadic and familial disease indicate a role for host genetic susceptibility.
The term was introduced in 1806 by Jean-Louis-Marc Alibert, a French dermatologist, who described patients with advanced skin tumors resembling mushrooms – large protruding, ulcerated, fungating masses on the skin.
The pathogenesis of MF remains incompletely understood and is likely multifactorial, involving chronic antigenic stimulation, immune dysregulation, epigenetic remodeling, and progressive oncogenic alterations in mature T lymphocytes. Malignant cells in classic MF are derived from skin-resident or skin-homing CD4+ memory T cells expressing cutaneous lymphocyte antigen (CLA) and chemokine receptors (CCR4 and CCR10), which facilitate epidermal and dermal trafficking.
A defining biologic feature is epidermotropism – the migration of neoplastic lymphocytes into the epidermis without significant spongiosis. Histologically, this produces collections of atypical cerebriform lymphocytes and, in some cases, Pautrier microabscesses.
The skin microenvironment- keratinocytes, dendritic cells, macrophages, fibroblasts, and microbial (particularly Staphylococcus aureus) colonization patterns – may amplify cytokine signaling and pruritus while sustaining malignant clones.
Over time, some tumors shift from a Th1-predominant immune milieu toward Th2 polarization, associated with impaired antitumor immunity, eosinophilia, susceptibility to infection, and more aggressive behavior. Large-cell transformation represents a major biologic turning point, marked by emergence of larger atypical cells, more aggressive clinical behavior, higher relapse risk, and inferior survival outcomes (Hristov AC et al., 2025).
MF Subtypes and Clinical Patterns
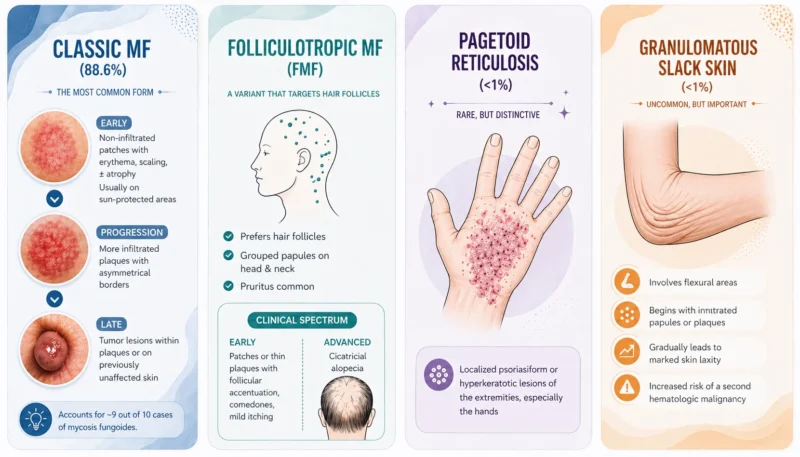
The WHO recognizes classic Mycosis Fungoides (MF) together with three distinct subtypes: folliculotropic MF (FMF), pagetoid reticulosis, and granulomatous slack skin.
Classic MF remains the predominant form (88.6%). Its course typically begins with non-infiltrated patches showing erythema, scaling, and occasional atrophy. It usually presents on sun-protected areas. Over time, lesions may evolve into more infiltrated plaques with asymmetrical borders. In some patients, often years after presentation, tumor lesions develop either within existing plaques or on previously unaffected skin.
FMF preferentially involves hair follicles, often presents with grouped papules on the head and neck, with pruritus as a frequent symptom. Earlier disease may appear as patches or thin plaques with follicular accentuation, comedones, and milder itching, whereas more advanced cases show cicatricial alopecia.
Pagetoid reticulosis and granulomatous slack skin are both rare, each accounting for <1% of MF, and usually follow a more indolent course. Pagetoid reticulosis presents with localized psoriasiform or hyperkeratotic lesions of the extremities, especially the hands. Granulomatous slack skin tends to involve flexural areas, beginning with infiltrated papules or plaques that gradually lead to marked skin laxity, and it carries an increased risk of a second hematologic malignancy (Sheern C et al., 2025).
When MF Extends Beyond the Skin
Lymph nodes are the most common and often earliest extracutaneous site of spread, with peripheral nodes affected more frequently than central nodes. Because early nodal involvement can be difficult to distinguish from benign reactive changes (dermatopathic lymphadenopathy), comparative T-cell clonality analysis of skin and lymph node specimens may help identify disease progression. Imaging modalities also play an important role.
Spleen involvement is second only to lymph nodes. It commonly presents as splenomegaly, which is classified as visceral involvement in staging systems even without histologic confirmation. Other relatively common sites include the liver and kidneys, usually in the setting of advanced systemic dissemination.
Advanced MF can clinically resemble Sézary syndrome, although both are recognized as distinct subtypes of CTCL. Sézary syndrome typically presents with diffuse erythroderma, often accompanied by intense pruritus, together with generalized lymphadenopathy and a marked burden of circulating malignant T-cells (Sézary cells) (Liu L et al., 2025).
Why MF Is Often Misdiagnosed?
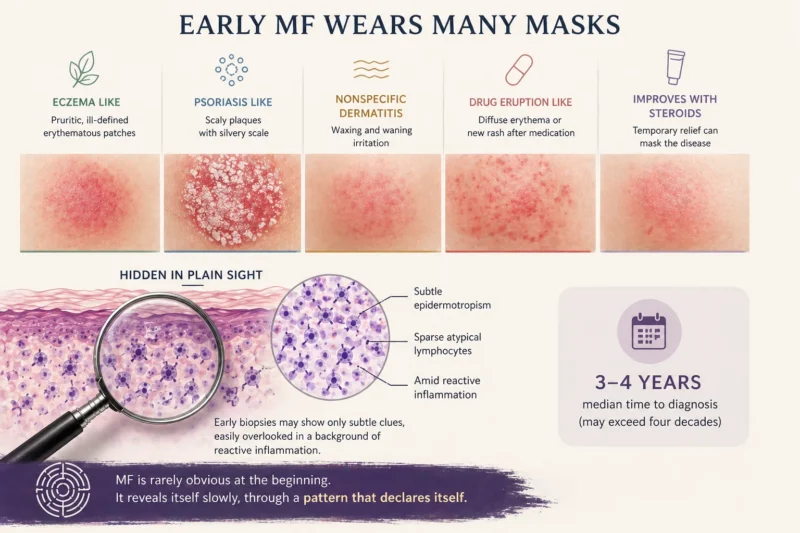
Earliest manifestations of MF often lack the classic histologic and clinical features of overt lymphoma, instead presenting as common inflammatory dermatoses.
- Patch-stage lesions may resemble eczema, presenting as pruritic, ill-defined erythematous patches
- MF may mimic psoriasis, particularly when lesions appear as scaly plaques
- It can present as nonspecific dermatitis, with waxing and waning irritation
- It may imitate a drug eruption, especially when diffuse erythema or new rashes develop after medication exposure
Early biopsies may show only subtle epidermotropism or sparse atypical lymphocytes hidden within a background of reactive inflammation. Temporary improvement with corticosteroids can also mask the disease. The median time from symptom onset to diagnosis in retrospective series is 3-4 years but may exceed four decades. In practice, MF is often recognized less by one finding than by a pattern that gradually declares itself.
How MF Is Diagnosed and Staged?
Diagnosis often requires multiple skin biopsies taken over time (at least two 6 mm punch biopsies are recommended from the most representative lesions, with topical corticosteroids avoided for at least 2 weeks). Immunohistochemical analysis most commonly demonstrates a predominance of CD4+ T cells, often accompanied by loss of CD7 and CD2 and a relative reduction in CD8+ cells.
Clonality studies using PCR or NGS are commonly used for diagnosis and for assessing relapse or progression. Yet clonality may also be detected in benign conditions such as lichen planus, pityriasis lichenoides, and lichen sclerosus as part of a reactive inflammatory process. Identification of the same T-cell clone in multiple lesions or over time increases diagnostic confidence for MF.
The staging of MF follows TNMB classification. The T category reflects the extent and type of skin lesions, including limited or extensive patches/plaques (T1-T2), tumors (T3), or erythroderma (T4), as well as lymph node involvement (N0-N3), visceral disease (M0-M1), and blood tumor burden (B0-B2).
When Dermatology Typically Leads
In early-stage MF (patch/plaque-predominant disease without extracutaneous involvement), dermatologists often coordinate frontline care. Priorities center on symptom control, lesion clearance, cosmetic preservation, quality of life, and timely escalation to systemic disease-modifying therapy. Common strategies include:
- topical corticosteroids
- topical mechlorethamine
- topical retinoids
- narrowband UVB phototherapy
- PUVA therapy
- localized radiotherapy
- management of itch, xerosis, and barrier dysfunction
- bexarotene, methotrexate, interferon
Toxicity monitoring, infection prevention, and long-term relapse surveillance are shared multidisciplinary responsibilities.
When Does Hematology-Oncology Lead?
Advanced-stage, blood-involved, transformed, or refractory MF requires systemic lymphoma-oriented management. Responses to conventional chemotherapy in CTCL are rarely durable and more than 90% of patients require additional therapy within the first year. A risk-adapted and stage-based approach, is generally preferred, incorporating biologic-response modifiers (bexarotene and interferon-alpha), histone deacetylase inhibitors (romidepsin), and monoclonal antibodies or antibody-drug conjugates (mogamulizumab, brentuximab vedotin).
Modern Systemic Therapies
The phase III ALCANZA trial established Brentuximab vedotin as an effective therapy for previously treated CTCL, showing superior PFS vs methotrexate or bexarotene (>12 vs 3.5 months), with 46-month follow-up, median PFS in MF remained 16.1 vs 3.5 months. At 1year, fewer BV-treated patients required alternative systemic therapy (34.5% vs 86.6%). ORR4 was 40% in limited-stage MF, 63% in stage IIB MF. Notably, responses occurred regardless of CD30 expression or large-cell transformation, so treatment selection should not depend solely on CD30 status.
Mogamulizumab is an approved therapy for CTCL based on the phase III MAVORIC trial, where it significantly improved PFS vs Vorinostat (7.7 vs 3.1 months). Its activity is compartment-specific, with higher response rates in blood (ORR 68%) and skin (42%) than lymph nodes (17%). Deeper and durable responses may occur in cases with increased CCR4 expression or activating CCR4 mutations.
A key toxicity is mogamulizumab-associated rash, which can mimic progression and requires dermatopathologic confirmation, notably, patients who develop this rash often have longer survival, possibly reflecting enhanced immune activation.
Durable remissions in CTCL have been observed with immunomodulatory therapies such as extracorporeal photopheresis (ECP) and interferon-α, supporting the importance of host immune control. This, together with frequent PD-L1 expression or structural variants, provides a strong rationale for checkpoint blockade. In a phase II trial of heavily pretreated advanced-stage patients, Pembrolizumab achieved an OR rate of 38%. Checkpoint inhibition may be particularly relevant in MF with large-cell transformation.
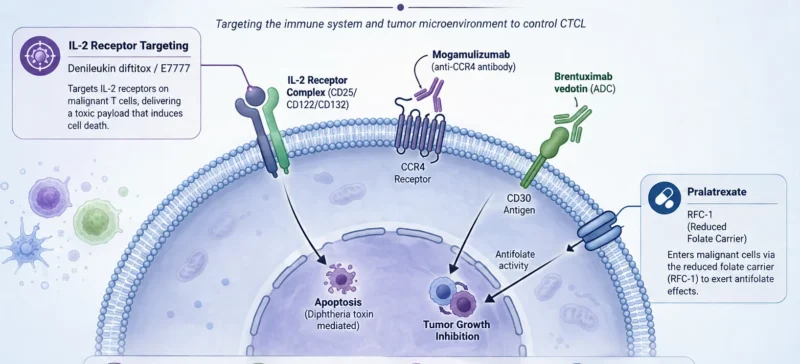
Because clonal T cells in CTCL often express the IL-2 receptor (CD25/CD122/CD132), IL-2-directed therapy became a rational target. Denileukin diftitox, an IL-2–diphtheria toxin fusion protein, demonstrated clinically meaningful activity in CTCL, with phase III data showing objective responses in 30% of patients and stable disease in an additional 32%, median time to response was 6 weeks, and median response duration was 7 months.
A later placebo-controlled phase III trial reported an ORR of 44% with median PFS exceeding 2 years. In meta-analysis, CD25-positive patients had higher response rates than CD25-negative patients (47.5% vs 30.6%), highlighting biomarker relevance. Key toxicities included vascular leak syndrome in ~25%, visual compromise, and infusion reactions, often manageable with premedication. A purified formulation, E7777, gained FDA approval in 2024 for stage I–III CTCL after at least one prior therapy.
Pralatrexate, a novel antifolate with high affinity for the reduced folate carrier (RFC-1) and a resistance profile distinct from methotrexate, demonstrated an OR rate of 29% in the PROPEL trial, which primarily enrolled patients with peripheral T-cell lymphoma, most with refractory disease.
Transplantation and Cellular Therapies
Autologous stem cell transplantation in advanced MF has generally produced only transient responses, whereas allogeneic transplantation may provide durable remission through a graft-versus-lymphoma effect. In a 60-patient series, 3-year PFS and OS were 34% and 53%, improving to 63% OS with reduced-intensity conditioning and matched-related donors. Earlier transplantation lowered 1-year relapse (25% vs 44%), and donor lymphocyte infusion induced CR in 47% of relapsed cases. Despite curative potential, non-relapse mortality remained significant (14%-40%), so allogeneic transplantation is best reserved for carefully selected patients.
Emerging cellular therapies in T-cell lymphomas demonstrate promising but variable efficacy across targets and platforms. CD70-directed allogeneic CAR T therapy with CTX130 achieved an ORR of 70% (7/10) at higher dose levels, including 80% in PTCL and 60% in CTCL, with manageable toxicity – CRS occurred in 80% and ICANS in 30%. In contrast, autologous CD5 CAR T-cell therapy showed more modest activity (ORR 44%), with responses in PTCL (57%) but none in CTCL.
AFM13 (CD30 × CD16A BsAb) combined with cord blood-derived NK cells, has shown a favorable safety profile (infusion reactions ~7%, no CRS, ICANS, or GVHD) and is under ongoing phase II evaluation for CD30-expressing transformed MF (Stuver R et al., 2024, Hristov AC et al., Roccuzzo G et al., 2025, NCI PDQ, 2025).
Strategies to Enhance Response Depth and Durability
A key modern strategy is combining time-limited local therapy with maintenance systemic treatment. Low-dose total skin electron beam therapy (TSEBT, 10-12 Gy) achieves high response rates (ORR ~90% across series) with less cumulative toxicity than historical 36 Gy regimens, allowing repeat use earlier in the disease course. Ongoing studies are pairing low-dose TSEBT with Mogamulizumab or Bexarotene to prolong remission after skin disease clearance.
A second strategy is improving tolerability of effective agents to extend treatment duration. Reduced-dose Brentuximab vedotin is under study to lessen peripheral neuropathy, interim data with 0.9 mg/kg showed ORR 42%, somewhat lower than ALCANZA, but with numerically less neuropathy and longer response duration. Similarly, optimizing dosing schedules for mogamulizumab may improve long-term practicality and durability (Stuver R et al., 2024).
Predictors of Prognosis
Prognosis in CTCL is driven primarily by stage at presentation. Increasing skin burden, lymphadenopathy, peripheral blood involvement, and visceral disease correlate with progressively worse outcomes. In a Cutaneous Lymphoma International Consortium analysis of 1,275 patients, four independent adverse prognostic factors were identified:
- stage IV disease
- age >60 years
- large-cell transformation
- elevated lactate dehydrogenase
Patients with stage IA disease often have median survival of 20+ years, with many deaths unrelated to MF, whereas more than 50% of stage III–IV patients die from disease, with median survival around 5 years. Patients with CTCL have an increased risk of secondary malignancies, including melanoma, highlighting the importance of appropriate screening and surveillance.
You can also read: Hodgkin Lymphoma (HL): Microenvironmental Biology Behind a Therapeutic Success Story
Written by Susanna Mikayelyan, MD
FAQ
Why is Mycosis Fungoides called “Mycosis” if it is not a fungal disease?
The name is historical. Early physicians described mushroom-like skin tumors in advanced disease, long before modern pathology clarified that MF is a type of cutaneous T-cell lymphoma rather than an infection.
Can Mycosis Fungoides look like eczema or psoriasis?
Yes. Early MF often mimics common inflammatory skin diseases such as eczema, psoriasis, or chronic dermatitis, which is one reason diagnosis may be delayed for years.
Is Mycosis Fungoides contagious?
No. MF is not contagious, infectious, or transmissible through touch, blood, or close contact.
Why do some patients need multiple biopsies before diagnosis?
Early lesions may show only subtle abnormalities under the microscope. Repeated biopsies over time can reveal clearer pathologic and immunologic features that support diagnosis.
Can sunlight help Mycosis Fungoides?
Controlled ultraviolet therapy, such as narrowband UVB or PUVA, is used medically in selected patients. However, unsupervised sun exposure is not a substitute for treatment and may increase skin damage risk.
What is the difference between Mycosis Fungoides and Sézary syndrome?
Both belong to cutaneous T-cell lymphomas, but Sézary syndrome usually presents with diffuse erythroderma, blood involvement, and widespread lymphadenopathy, making it biologically and clinically distinct.
Can people with early-stage MF live a normal lifespan?
Many patients with stage IA or limited early-stage disease can live for decades, especially with regular follow-up and proper management.
Why does care sometimes shift from dermatology to hematology-oncology?
When disease becomes advanced, blood-involved, transformed, or spreads beyond the skin, treatment often requires systemic lymphoma-oriented therapies best coordinated with hematology-oncology teams.
Are new treatments improving outcomes in Mycosis Fungoides?
Yes. Modern therapies such as targeted antibodies, immunotherapy, biologic agents, and cellular therapies are expanding options, particularly for relapsed or advanced disease.