Metastasis remains the central reason lung cancer is so difficult to cure. Although modern imaging, molecular profiling, immunotherapy, and targeted therapy have changed outcomes for many patients with non-small cell lung cancer (NSCLC), the biological steps that allow cancer cells to leave the primary tumor, establish distant disease, and continue spreading remain incompletely understood.
A new Nature study from the TRACERx lung study and the PEACE research autopsy programme offers one of the most detailed evolutionary maps of NSCLC metastasis to date. By analyzing 501 longitudinally collected primary and metastatic tumor samples from 24 patients, the investigators reconstructed how lung cancer spread from diagnosis to death, revealing that metastatic disease is often far more genetically diverse and spatially dynamic than single-biopsy sampling suggests (Hessey et al., 2026).
A Rare View Across The Full Disease Course
The study combined two major research efforts. TRACERx follows patients with operable NSCLC using multi-region tumor sampling, while PEACE enables research autopsy sampling after death. Together, they allowed investigators to compare paired primary tumors, relapse samples, progression samples, and post-mortem metastases across the same patients.
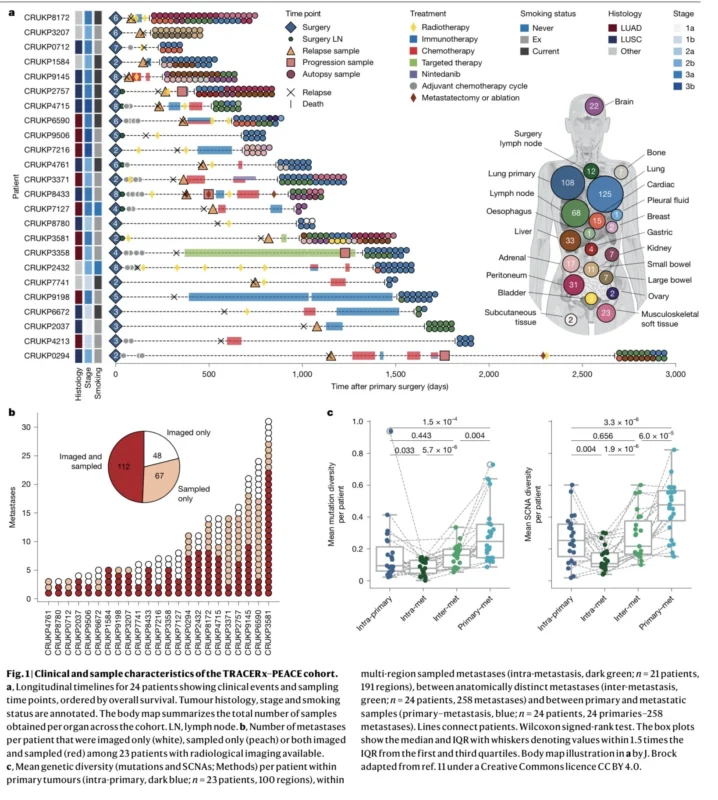
The final cohort included patients with common NSCLC histologies, including lung adenocarcinoma and squamous cell carcinoma. In total, the researchers analyzed 108 regions from 24 primary tumors, 41 regions from 35 pre-mortem metastases, and 352 regions from 233 anatomically distinct metastases collected at autopsy. Importantly, sequencing data covered approximately 70% of metastases that had been radiologically detected before death, giving the analysis unusual depth compared with standard metastatic biopsy studies (Hessey et al., 2026).
This level of sampling matters because metastatic disease is rarely represented by a single lesion. The study showed that individual metastases and anatomically distinct metastases can contain different subclones, meaning that one metastatic biopsy may underestimate the true evolutionary complexity of advanced NSCLC.
© The Author(s) 2026
Metastases Continue To Evolve After They Spread
One of the central findings was that metastases often diverged substantially from their ancestral primary tumors. The genomes of metastases accumulated additional driver alterations, whole-genome doubling events, and site-specific subclonal changes after dissemination.
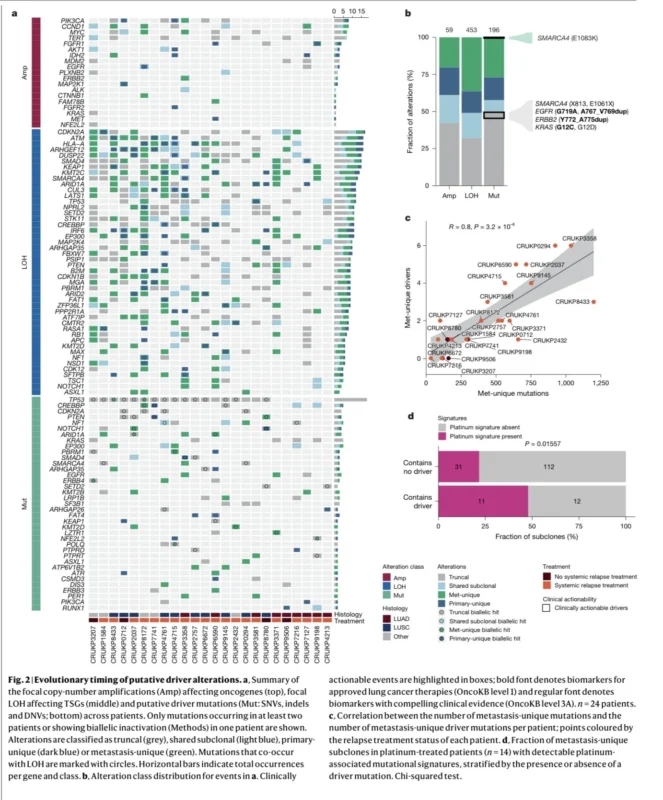
Most driver alterations were still found in primary tumors, but metastases acquired additional mutations with potential biological consequences. Approximately 83% of patients had at least one metastasis-unique driver mutation. The study also found that whole-genome doubling occurred in 92% of patients, sometimes in primary disease and sometimes later in metastases. These events were associated with greater copy-number heterogeneity between metastases and between primary and metastatic disease (Hessey et al., 2026).
This supports a clinically important idea: metastatic NSCLC is not simply a late copy of the primary tumor. It is an evolving ecosystem. Treatment pressure, time, anatomical location, and chromosomal instability all appear to shape how metastatic clones develop.
Multiple Primary Subclones Can Seed Metastases
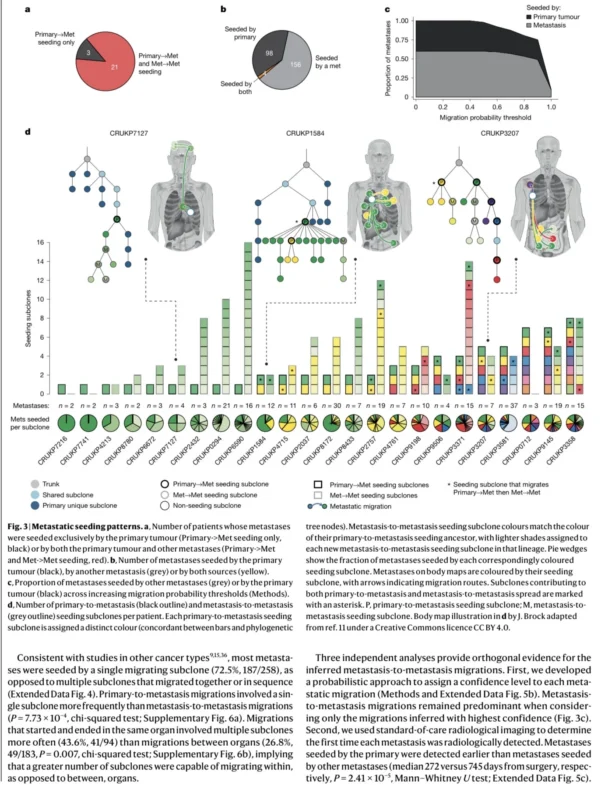
In 62.5% of patients, more than one subclone from the primary tumor disseminated and founded distinct metastases. This challenges a simplified model in which one dominant clone leaves the primary tumor and drives all subsequent metastatic disease.
Instead, the study suggests that several genetically distinct primary tumor subclones may independently acquire the ability to spread. Most metastases were founded by a single migrating subclone, but different metastases within the same patient could originate from different primary subclones (Hessey et al., 2026).
This finding has practical implications for early-stage NSCLC. It reinforces why intratumoral heterogeneity matters before relapse and why multi-region profiling has been so central to the TRACERx programme. A single region of the primary tumor may not capture all subclones with metastatic potential.
© The Author(s) 2026
Metastasis-To-Metastasis Spread Was Common
Perhaps the most striking result was the extent of metastasis-to-metastasis seeding. The investigators found that metastases were not only end-points of spread; they frequently became sources of further dissemination.
Overall, 60% of sampled metastases were seeded by other metastases, while 38% were seeded by the primary tumor and 2% by both. In most patients, seeding subclones were found in both the primary tumor and established metastases (Hessey et al., 2026).
This creates a more dynamic model of NSCLC progression. After the primary tumor seeds early metastatic deposits, those metastases may continue the cascade by founding additional lesions. The authors also observed that metastases seeded by the primary tumor were detected earlier on imaging than metastases seeded by other metastases, supporting the temporal logic of this metastatic cascade.
Time In Situ May Increase Seeding Potential
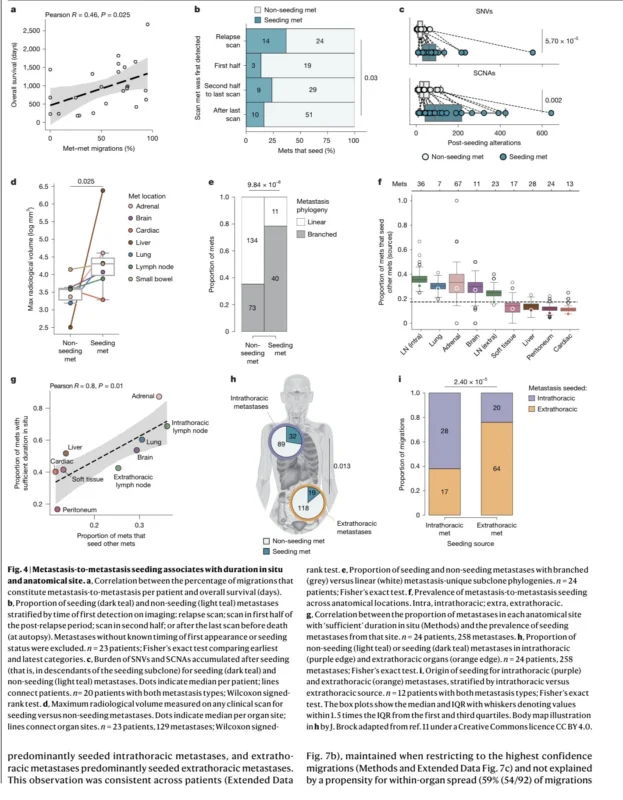
The study found that metastases with longer time in the body were more likely to seed additional metastases. Metastases that seeded other lesions were detected earlier on imaging, had more accumulated somatic alterations, were larger on radiology, and contained more subclonal diversity than non-seeding metastases (Hessey et al., 2026).
This does not mean every older metastasis will inevitably spread. But it suggests that time creates opportunity: larger tumor volume, more cancer cells, and greater subclonal diversity may increase the probability that a metastasis generates a subclone capable of onward dissemination.
This finding offers a biological rationale for why controlling existing metastases may matter in selected patients. It aligns with the concept behind local consolidative therapy in oligometastatic NSCLC, where radiotherapy or surgery to persistent metastatic sites has improved outcomes in some phase II studies, although more recent immunotherapy-era data have been mixed (Gomez et al., 2019; Iyengar et al., 2024).
© The Author(s) 2026
Spread Was Often Anatomically Constrained
Metastatic migration was not random. Most metastasis-to-metastasis migrations began and ended within the same anatomical cavity. Intrathoracic metastases tended to seed other intrathoracic metastases, while extrathoracic metastases more often seeded other extrathoracic sites.
The study found that 71.3% of metastasis-to-metastasis migrations remained within the anatomical cavity where they originated. Intrathoracic metastases also appeared earlier and were more likely to seed other metastases than extrathoracic lesions, partly because they had more time in situ (Hessey et al., 2026).
This anatomical pattern suggests that the route of spread matters. Local thoracic spread, lymphatic movement, airspace spread, vascular dissemination, and distant colonization may require different biological capabilities. NSCLC metastasis may therefore be better understood as several related but distinct dissemination processes rather than one uniform event.
Chromosomal Instability May Help Tumors Escape The Thorax
A key biological signal involved chromosomal instability. Subclones that seeded metastases had higher somatic copy-number alteration burdens than non-seeding subclones. This was especially clear for subclones that seeded extrathoracic metastases.
The authors found that primary subclones capable of leaving the thorax and seeding distant disease were enriched for copy-number alterations. Intrathoracic metastases that seeded extrathoracic metastases also had higher copy-number alteration burdens than those that seeded only intrathoracic lesions (Hessey et al., 2026).
This supports previous TRACERx findings linking chromosomal instability with relapse, metastatic spread, and poorer outcomes in lung cancer (Frankell et al., 2023; Pawlik et al., 2025). The new study adds spatial resolution, suggesting that chromosomal instability may be particularly relevant to the ability of cancer cells to colonize outside the thoracic cavity.
What This Means For Lung Cancer Care
This work is not immediately practice-changing in the sense of introducing a new treatment. Its value is biological and strategic. It shows that metastatic NSCLC is often shaped by multiple seeding events, ongoing evolution after dissemination, and frequent metastasis-to-metastasis spread.
For clinicians and researchers, this has several implications. First, single metastatic biopsies may miss clinically relevant diversity. Second, metastatic lesions may continue evolving under treatment pressure, potentially generating new resistance mechanisms. Third, some metastases may function as reservoirs for further spread, supporting continued interest in local treatment strategies for carefully selected patients. Fourth, chromosomal instability may become an important biomarker for identifying tumors with higher metastatic potential, particularly for extrathoracic dissemination.
The authors appropriately note limitations. The cohort was small, most patients received systemic therapy, and the findings may not fully apply to untreated metastatic NSCLC or patients who present with de novo stage IV disease. Bulk whole-exome sequencing also cannot capture all structural variants, single-cell diversity, microenvironmental drivers, or non-genetic mechanisms of metastasis (Hessey et al., 2026).
Still, the depth of sampling makes this one of the clearest views yet of how lung cancer metastases evolve over time.
© The Author(s) 2026
Key Takeaway
The TRACERx–PEACE study shows that NSCLC metastasis is not a one-way journey from primary tumor to distant site. In many patients, multiple primary subclones seed distinct metastases, and established metastases then seed further metastases. Time in situ, anatomical location, and chromosomal instability appear to shape this metastatic cascade.
For lung oncology, the message is clear: metastatic disease is biologically diverse, spatially organized, and continuously evolving. Understanding that complexity is essential for designing better strategies to prevent relapse, monitor progression, and treat advanced NSCLC more effectively.
Read full article here


