Carcinoembryonic antigen, or CEA, has long been one of the most familiar serum biomarkers in lung cancer. In daily practice, it is often interpreted as a rough signal of tumor burden, especially in adenocarcinoma, but its place in the era of molecularly defined metastatic non-small cell lung cancer has remained less clear.
As comprehensive genomic profiling reshaped the treatment of advanced NSCLC, the question became more relevant: does baseline CEA simply reflect disease extent, or can it also offer clues about the underlying molecular biology of the tumor?
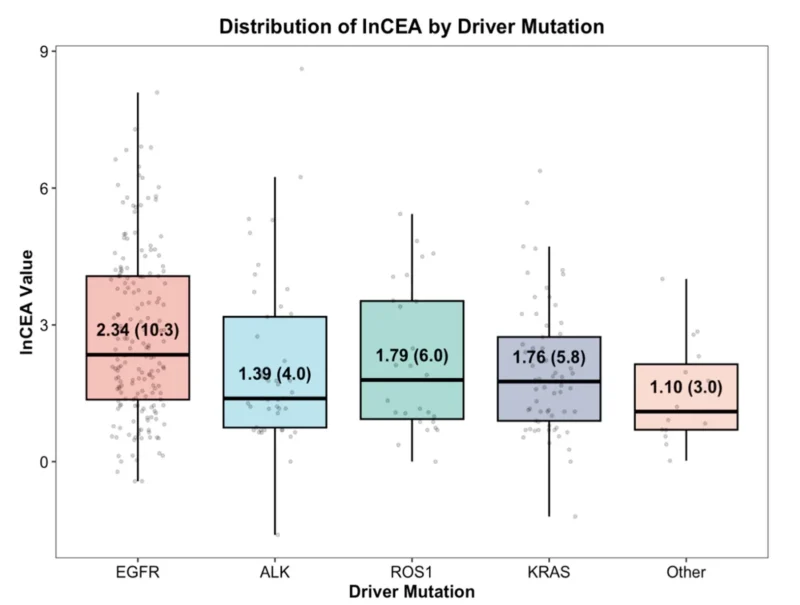
A new retrospective multicenter study brings this question back into focus. In a cohort of 332 patients with metastatic NSCLC harboring oncogenic driver alterations, the investigators found that baseline serum CEA levels differed significantly across molecular subtypes, with the highest median values seen in EGFR-mutant disease.
Higher baseline serum CEA was also independently associated with worse overall survival, even after adjustment for driver subtype and metastatic burden (Aytac et al., 2026).
This is not the kind of study that immediately changes treatment selection. Molecular testing remains the standard, and no serum marker can replace next-generation sequencing. Still, the findings are clinically interesting because they suggest that baseline serum CEA may capture more than just advanced disease burden. It may also reflect differences in tumor biology across driver-defined metastatic NSCLC.
Why CEA Still Matters in Modern NSCLC
Lung cancer remains the leading cause of cancer-related death worldwide, and NSCLC accounts for the large majority of cases (Sung et al., 2021). Over the past two decades, the discovery of actionable oncogenic alterations such as EGFR, ALK, ROS1, KRAS, BRAF, and MET has transformed metastatic NSCLC from a single disease category into a collection of biologically distinct subgroups, each with different treatment options and different prognostic expectations (Soria et al., 2018; Solomon et al., 2014).
In that context, tissue-based or plasma-based molecular profiling is now central to treatment planning. But real-world practice is still full of delays, limited tissue, incomplete testing, and unequal access to broad molecular platforms. That is exactly why interest in simple and inexpensive biomarkers such as cerum CEA has persisted. CEA is far from specific, but it is widely available, easy to measure, and already part of routine oncology workflows in many centers.
Historically, elevated baseline serum CEA has been associated with more advanced disease and poorer outcomes in NSCLC, particularly in adenocarcinoma (Grunnet and Sorensen, 2011; Wang et al., 2014). What remained less well defined was whether CEA behaves differently across driver mutation subgroups in metastatic disease.
Copyright: © 2026 by the authors.
Published by MDPI on behalf of the
Lithuanian University of Health
Sciences.
How the Study Was Designed
The study by Aytac et al. was a retrospective multicenter analysis including eight oncology centers across Türkiye. The investigators reviewed patients diagnosed with metastatic NSCLC between 2010 and 2025 and selected a molecularly defined cohort of 332 patients with oncogenic alterations. These were grouped as EGFR-mutant, ALK-positive, ROS1-positive, KRAS-mutant, and a smaller group with other rare alterations. Baseline serum CEA values were collected at the time of metastatic diagnosis, before systemic therapy started.
Because serum CEA values were markedly right-skewed, the investigators analyzed them on a natural logarithmic scale, which is a reasonable methodological choice for this kind of biomarker distribution. They then explored associations between baseline serum CEA, molecular subtype, clinical characteristics, and overall survival using multivariable generalized linear models and Cox proportional hazards regression.
This is important because the study was not simply describing raw serum CEA values. It was testing whether the association remained meaningful after adjusting for the clinical variables that also influence biomarker levels and prognosis.
EGFR-Mutant Tumors Had the Highest Median CEA Levels
The most immediate finding was that baseline CEA differed significantly across driver-defined subgroups. EGFR-mutant tumors had the highest median CEA levels, while lower values were seen in ALK, ROS1, KRAS, and especially the rare alteration group. In multivariable analysis, driver subtype remained independently associated with CEA levels, even after adjustment for histology and metastatic burden.
This aligns with earlier literature suggesting a link between elevated CEA and EGFR-mutant adenocarcinoma. Prior studies reported that patients with EGFR-mutated NSCLC often have higher baseline CEA levels than those with wild-type tumors, and a meta-analysis by Gan et al. found that CEA-positive NSCLC cases were more likely to harbor EGFR mutations (Gao et al., 2017; Gu et al., 2018; Gan et al., 2023).
The current study does not prove that CEA can predict EGFR status in clinical practice, and it should not be used that way in isolation. But the consistency of the signal is notable. It suggests that CEA may partly reflect the biological characteristics of EGFR-driven adenocarcinoma, not only the size or spread of disease.
Histology and Metastatic Burden Also Shaped Baseline CEA
The study also confirmed that CEA is not primarily a molecular marker. It still behaves as a marker influenced by broader clinical context.
In multivariable analysis, squamous histology was associated with lower CEA compared with adenocarcinoma, which is not surprising and fits prior experience with CEA being more informative in adenocarcinoma-predominant disease. More importantly, increasing metastatic site count was independently associated with higher baseline CEA. In other words, each additional metastatic site was linked to a rise in CEA, reinforcing the idea that CEA still tracks tumor burden.
This dual signal is probably the most interesting part of the paper. Serum CEA seems to sit at the intersection of disease extent and disease biology. That makes it potentially useful as an adjunctive clinical clue, even if it remains far too nonspecific to act as a surrogate for molecular testing.
Higher Baseline CEA Was Linked to Worse Survival
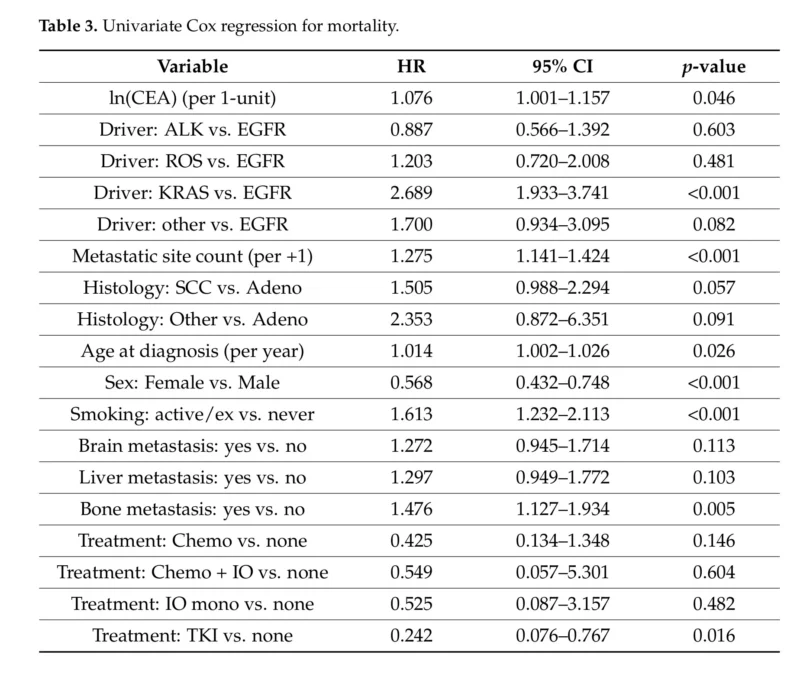
The prognostic analysis is where the study becomes most clinically relevant. Median overall survival for the full cohort was 26.2 months. In multivariable Cox regression, higher baseline serum CEA remained independently associated with increased mortality risk. The same was true for greater metastatic burden. Female sex appeared protective, while KRAS mutation was associated with significantly worse survival compared with EGFR-mutant disease.
These results fit well with the broader NSCLC literature. Multiple earlier studies and meta-analyses have already shown that higher pretreatment CEA is associated with poorer survival in NSCLC (Wang et al., 2014; Zhang et al., 2015). What this new study adds is that the association remains visible within a molecularly selected metastatic population, even after accounting for driver subtype.
That matters because targeted therapy has changed the natural history of several molecular subsets. One could argue that once driver status is known, a crude serum biomarker such as CEA may lose much of its relevance. This analysis suggests that is not entirely true. CEA still appears to carry prognostic information beyond genotype alone.
Copyright: © 2026 by the authors.
Published by MDPI on behalf of the
Lithuanian University of Health
Sciences.
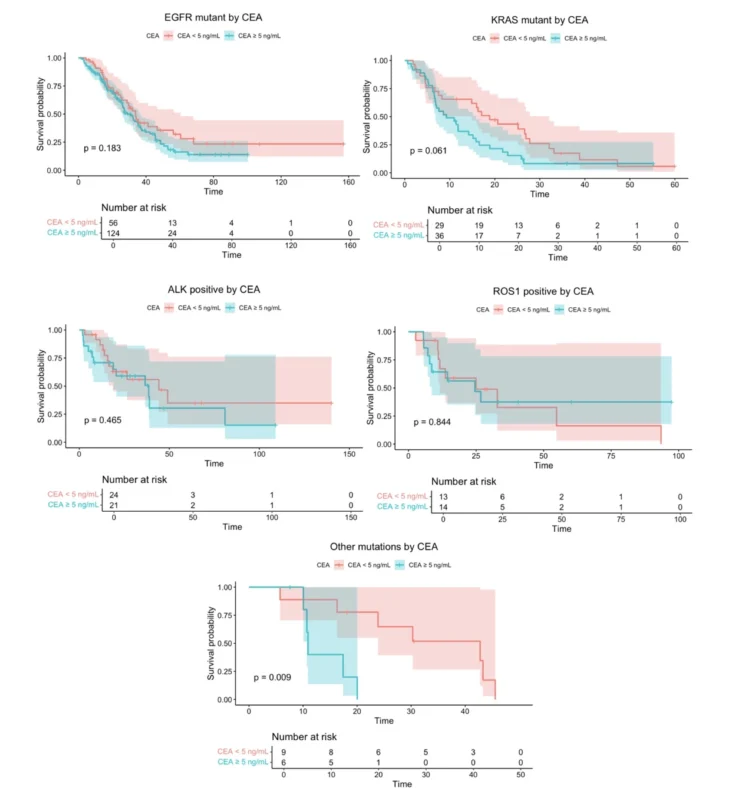
The CEA Cutoff Story Was More Uneven
The investigators also looked at a clinically intuitive cutoff of 5 ng/mL. Across most driver-defined subgroups, patients with CEA below 5 ng/mL tended to have longer survival than those with higher CEA. However, this reached statistical significance only in the small rare-alteration group.
That result deserves caution. The overall trend was directionally consistent, but the subgroup sizes were limited, especially for ROS1 and the rare-alteration cohort. This likely reduced statistical power and makes it difficult to draw firm subtype-specific conclusions from the Kaplan–Meier comparisons alone.
Still, the broader message holds. Higher baseline CEA generally moved with worse outcome, even if the simple 5 ng/mL binary split was not equally informative in every molecular subgroup.
KRAS Remained the Poor-Prognosis Comparator
One of the clearest survival findings in the study was the poorer outcome associated with KRAS-mutant disease compared with EGFR-mutant disease. In multivariable analysis, KRAS mutations carried a substantially higher mortality risk, while ALK and ROS1 were not significantly different from EGFR.
This is not unexpected. EGFR-mutant and ALK-positive NSCLC often derive major benefit from effective targeted therapy, which has historically translated into longer survival. KRAS-mutant disease, although now increasingly targetable in select settings, still tends to show more variable outcomes and often lacks the same degree of durable disease control seen with EGFR-directed therapy.
That context is important because it reminds us that CEA is not operating in a vacuum. Prognosis in metastatic NSCLC is still shaped primarily by tumor genotype, metastatic burden, treatment access, and treatment efficacy. CEA may add information, but it does not replace the dominant role of molecular classification.
What This Study Adds to Practice
The main practical message is not that oncologists should use CEA to guess the mutation before molecular results return. That would be too simplistic and too unreliable.
Instead, the value of this study is more subtle. It suggests that baseline CEA may function as an easily accessible biomarker that reflects both tumor burden and molecularly influenced biology in metastatic NSCLC.
In resource-limited settings, or during the interval before full molecular results are available, an unexpectedly high baseline CEA in a patient with metastatic adenocarcinoma might reinforce clinical suspicion of certain biologic patterns, especially EGFR-driven disease. That does not justify treatment decisions without molecular confirmation, but it may add context.
The second contribution is prognostic. Even in a driver-positive metastatic cohort, higher baseline CEA remained independently associated with worse survival. That makes it a potentially useful marker for baseline risk stratification and perhaps later for disease monitoring, although this study did not address dynamic CEA changes during treatment.
Copyright: © 2026 by the authors.
Published by MDPI on behalf of the
Lithuanian University of Health
Sciences.
The Limits of the Evidence
This remains a retrospective study, and its limitations are real. The cohort was molecularly selected, not an unselected NSCLC population, so the results cannot automatically be generalized to all metastatic cases. Molecular testing methods varied across centers and over time, PD-L1 data were incomplete, ECOG performance status was unavailable, and some subgroups were small.
Treatment patterns were also heavily tied to driver subtype, which makes it difficult to completely separate biomarker effects from therapy effects.
The authors also appropriately note that only baseline CEA was studied. Longitudinal CEA kinetics may ultimately be more clinically informative than a single baseline value, especially in patients receiving targeted therapy, where early biomarker changes may reflect treatment response or resistance patterns.
The Bottom Line
The study by Aytac and colleagues does not redefine CEA as a precision oncology biomarker. But it does sharpen its relevance.
In metastatic NSCLC with oncogenic driver alterations, baseline CEA levels were not uniformly distributed. They were highest in EGFR-mutant tumors, lower in several other molecular subgroups, and independently associated with metastatic burden, histology, and overall survival. Higher baseline CEA remained a marker of worse prognosis even after adjustment for driver subtype.
For clinicians, that means CEA may still have a role as an adjunctive clinical signal in metastatic NSCLC, especially when interpreted in context rather than isolation. It reflects disease burden, but it may also reflect something deeper about tumor biology. In the molecular era, that makes it more interesting than it first appears.
You can read full article here


