Extensive-stage small cell lung cancer remains one of the hardest diseases in thoracic oncology. The addition of immune checkpoint blockade to platinum-etoposide improved first-line outcomes and established chemoimmunotherapy as the standard of care, but long-term survival is still limited and clinicians still do not have a validated biomarker that can identify which patients are most likely to benefit. That is what makes this prospective multicenter study by Piedra and colleagues especially interesting. Rather than focusing on tumor PD-L1 alone, the investigators asked whether PD-L1 expression on circulating immune cells could provide clinically useful information before treatment even begins.
The study evaluated untreated patients with extensive-stage small cell lung cancer who received first-line durvalumab plus platinum-etoposide as part of the CANTÁBRICO trial platform. Baseline blood samples were analyzed by flow cytometry, and the authors examined the percentage of PD-L1-positive immune cell subsets in relation to progression-free survival, overall survival, and immune-related adverse events. Their central finding was clear: patients with higher baseline percentages of circulating PD-L1-positive monocytes had significantly longer progression-free survival, while higher baseline PD-L1-positive neutrophils were associated with a greater likelihood of immune-related toxicity.
Why This Question Matters
Biomarker development in extensive-stage small cell lung cancer has been frustrating. Tumor PD-L1 has not emerged as a practical first-line selection tool in the way it has for parts of non-small cell lung cancer, and other candidates, including tumor mutational burden, transcriptomic subtypes, MHC-related signals, and metastatic pattern, have not yet delivered a biomarker robust enough for routine use. This study takes a different direction. Instead of relying on tumor tissue alone, it turns to peripheral blood, a much more accessible material, and examines the immune system directly. That makes the question clinically relevant from the start.
Circulating biomarkers are attractive for another reason. They can be obtained noninvasively, repeated over time, and potentially integrated into real-world workflows faster than more complex tumor-based assays. The group also built on prior observations in non-small cell lung cancer, where PD-L1-positive circulating immune cells, especially monocytes, had already shown potential relevance. The novelty here is that this had not been studied in extensive-stage small cell lung cancer in a prospective frontline chemoimmunotherapy population.
Journal of Translational Medicine
Study Design And Patient Population
This was a prospective multicenter observational analysis embedded in the phase IIIb CANTÁBRICO trial. Eligible patients had untreated extensive-stage small cell lung cancer, ECOG performance status 0 to 1, and received first-line durvalumab with platinum-etoposide. Blood was collected before treatment initiation and centrally analyzed. Healthy donors were also included to help define reference ranges for PD-L1-positive cell populations.
The analysis included 41 patients. Their median age was 64 years, 65.9% were male, 73.2% had ECOG performance status 1, 9.8% had central nervous system metastases, and 31.7% had liver metastases. Most patients, 90.2%, completed the planned four cycles of chemotherapy plus durvalumab. At data cutoff, the median follow-up was 10.19 months, median progression-free survival was 7.0 months, and median overall survival was 10.3 months. These numbers are clinically credible for an extensive-stage small cell lung cancer cohort treated in routine-like contemporary practice.
What The Blood Showed At Baseline
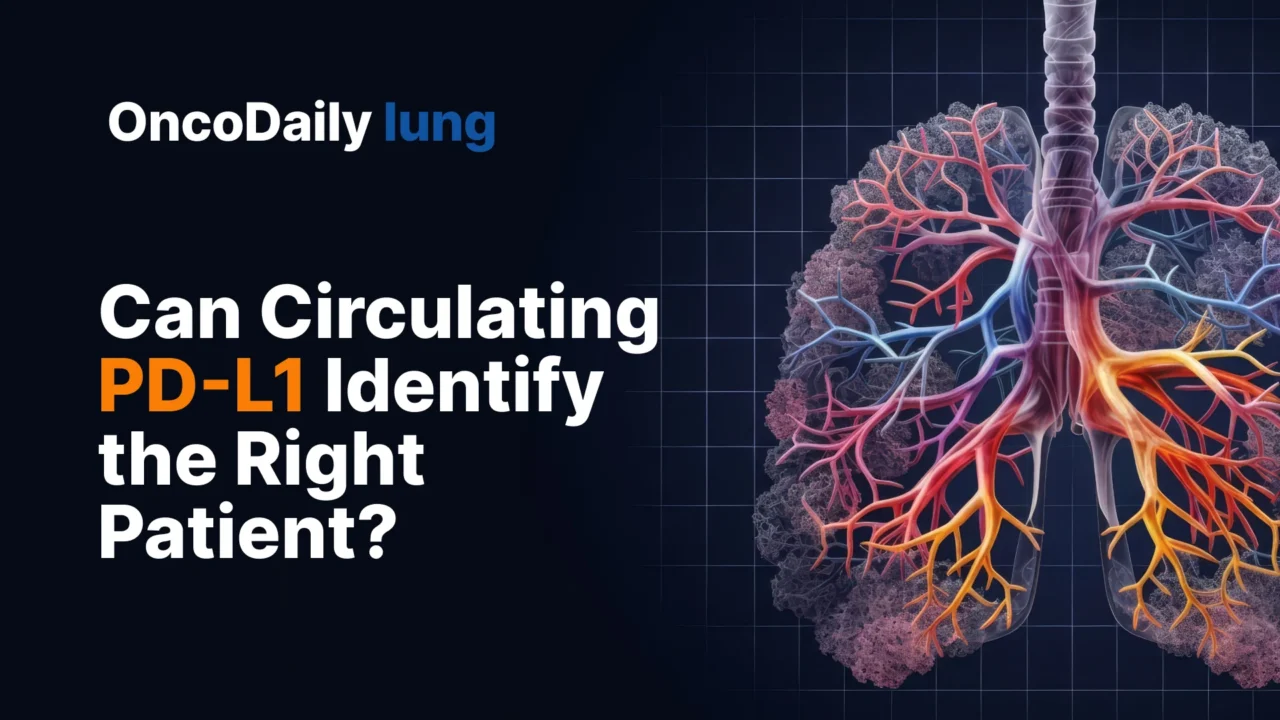
At baseline, neutrophils and T-lymphocyte subsets were the dominant circulating populations. Compared with healthy donors, patients with extensive-stage small cell lung cancer had significantly higher percentages of neutrophils, PD-L1-positive CD4-positive T cells, PD-L1-positive CD8-positive T cells, PD-L1-positive CD20-positive B cells, and PD-L1-positive platelets. There were also trends toward higher PD-L1-positive NK cells, PD-L1-positive neutrophils, and PD-L1-positive monocytes. In other words, the blood immune landscape in these patients already looked immunologically altered before treatment started.
That observation matters because it supports the biologic premise of the study. If the peripheral immune compartment is already shifted in extensive-stage disease, then measuring PD-L1 across circulating immune subsets is not a trivial exercise. It may reflect a real systemic tumor-immune interaction rather than a background signal.
Journal of Translational Medicine
The Most Important Result Was In Monocytes
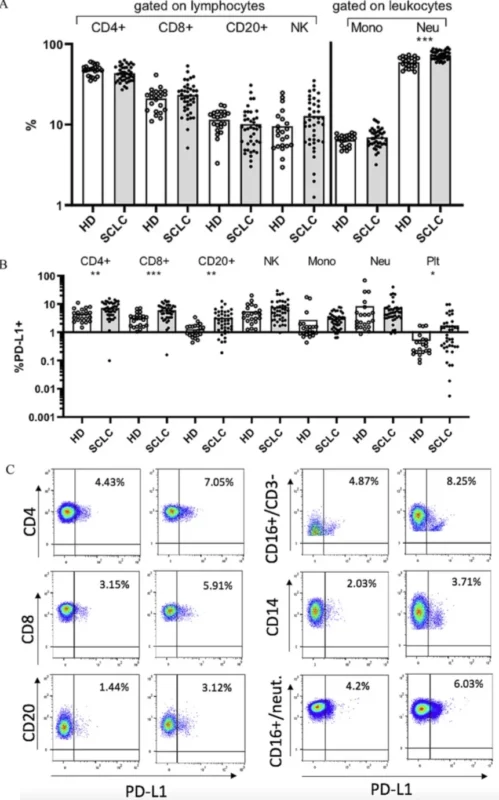
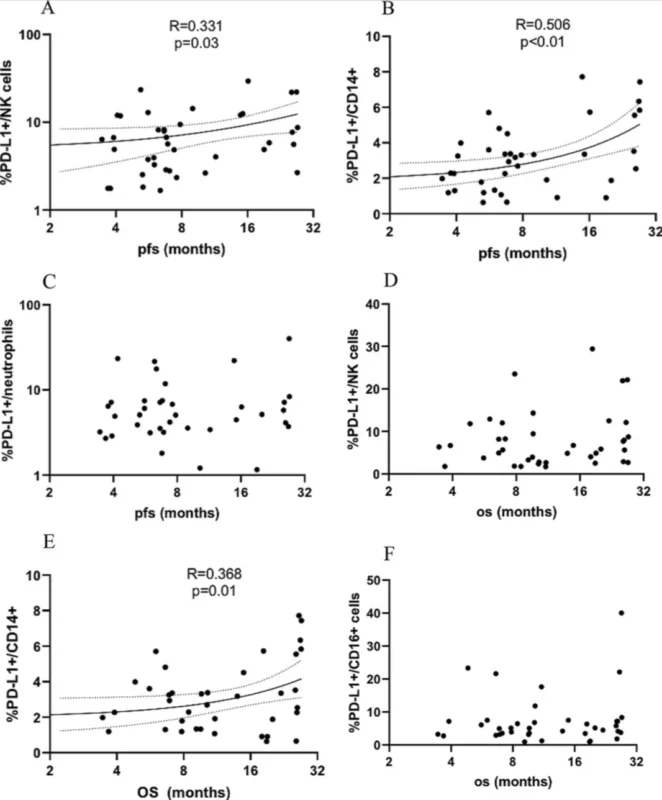
The strongest survival signal in the paper came from monocytes. The percentage of PD-L1-positive monocytes correlated with progression-free survival and also correlated with overall survival in continuous analyses. When patients were split into low and high groups using cutoffs derived from healthy donor confidence intervals, those with high baseline PD-L1-positive monocytes had significantly longer median progression-free survival: 8.97 months versus 5.97 months, with a p value of 0.007.
That difference is meaningful, especially in a disease where median frontline progression-free survival remains short. There was also a trend toward better overall survival in the high-monocyte group, although it did not reach conventional statistical significance. Median overall survival was not reached in the high group versus 9.13 months in the low group, with a p value of 0.092. In a cohort this small, that type of borderline overall survival result should not be ignored, but it should be interpreted carefully.
The study also explored clinically relevant subgroups. The progression-free survival advantage associated with high PD-L1-positive monocytes remained significant in patients without CNS metastases, without bone metastases, without liver metastases, and in those with ECOG performance status 1. These subgroup signals are intriguing because they suggest the monocyte finding was not just a random imbalance in one favorable clinical subset. Still, the numbers are small, and subgroup results in a 41-patient cohort should be read as supportive, not definitive.
Other Cell Populations Were Less Convincing
The study also looked at NK cells, neutrophils, T cells, B cells, and platelets. PD-L1-positive NK cells correlated with progression-free survival in continuous analysis, and there were trends toward longer progression-free survival in patients with high PD-L1-positive NK cells and high PD-L1-positive B cells, but these did not reach statistical significance in grouped survival analyses. PD-L1-positive neutrophils, T-cell subsets, and platelets did not show a significant survival association.
This pattern helps focus the clinical message. The paper is not claiming that all circulating PD-L1-positive immune compartments are useful. The main candidate biomarker for efficacy is PD-L1-positive monocytes.
Journal of Translational Medicine
The Toxicity Signal Pointed To Neutrophils
The other important finding involved immune-related adverse events. Sixteen of the 41 patients, or 39%, developed 25 immune-related adverse events of any grade. The most common were hypothyroidism and hyperthyroidism. Most were grade 1 or 2, but the cohort also included grade 3 events and one grade 5 event.
Patients who developed immune-related adverse events had significantly higher baseline PD-L1-positive neutrophils, both by percentage and by absolute count. Higher PD-L1-positive neutrophils were also associated with more non-dermatologic toxicities and with higher-grade immune-related adverse events. No similarly strong association was seen for monocytes, NK cells, lymphocytes, or platelets.
This is a useful distinction. The same biomarker was not doing everything. Monocytes appeared more relevant to treatment benefit, while neutrophils appeared more relevant to immune toxicity risk. That separation makes biologic sense and strengthens the paper conceptually.
What The Study Adds To The Field
The most important contribution of this paper is not that it solves biomarker selection in extensive-stage small cell lung cancer. It does not. What it does is open a realistic path toward a noninvasive baseline biomarker that may be easier to use than many tissue-based strategies that remain difficult to implement widely.
PD-L1-positive monocytes are especially interesting because they were associated with better progression-free survival in a setting where clinicians currently have no established biomarker to decide who derives the most benefit from frontline durvalumab plus chemotherapy. The authors are appropriately cautious and describe the finding as hypothesis-generating, but it is more than a weak exploratory signal. The association is statistically significant, biologically plausible, and based on a prospectively collected cohort.
The toxicity findings are also clinically relevant. Predicting immune-related adverse events before treatment remains an important unmet need, especially when immunotherapy is being moved earlier and used more broadly. If PD-L1-positive neutrophils are confirmed in larger datasets, they may eventually help identify patients who need closer toxicity monitoring.
The Limits Need To Be Taken Seriously
The study also has clear limitations, and the authors acknowledge them openly. The sample size is small. PD-L1-positive immune cells were measured only at baseline, so the study cannot describe how these populations change during treatment. There was no independent validation cohort. Multiple testing raises the risk of overinterpretation, and the overall survival analysis lacked power to confirm the trend observed for monocytes.
These limitations do not invalidate the results, but they do shape how they should be used. This is not a practice-changing biomarker paper yet. It is an important signal that deserves external validation.
Final Perspective
This study gives extensive-stage small cell lung cancer something it has badly needed: a plausible biomarker lead that is both clinically relevant and practically accessible. In a prospective cohort of patients treated with first-line durvalumab plus platinum-etoposide, high baseline PD-L1-positive monocytes were associated with significantly longer progression-free survival, while high PD-L1-positive neutrophils identified patients more likely to develop immune-related adverse events.
That does not mean PD-L1-positive circulating immune cells are ready for routine decision-making. But it does mean the field may need to look beyond tumor tissue alone when trying to understand who benefits most from immunotherapy in small cell lung cancer. Sometimes the most useful biomarker is not inside the tumor at all, but already circulating in the blood before the first cycle begins.
Read full article here