Stage III non-small cell lung cancer remains one of the most clinically challenging spaces in thoracic oncology. It sits between early-stage disease, where surgery may offer cure, and metastatic disease, where systemic treatment dominates. For patients with unresectable stage III NSCLC, definitive platinum-based chemoradiotherapy has long been the backbone of treatment. The PACIFIC trial changed the field by introducing durvalumab after chemoradiotherapy for unselected patients with unresectable stage III NSCLC, but patients with EGFR-mutated tumors remained a biologically distinct group with less clear benefit from immunotherapy-based consolidation (Antonia et al., 2017; Gray et al., 2022).
The phase III LAURA trial has now changed this treatment gap. By showing a major progression-free survival benefit with osimertinib after chemoradiotherapy, LAURA has established a targeted post-CRT strategy for patients with unresectable stage III EGFR-mutated NSCLC. This is not simply an extension of the metastatic EGFR-mutated NSCLC paradigm into earlier disease. It is a new standard for a population that previously had no clearly optimized biomarker-directed consolidation approach (Lu et al., 2024).
Why LAURA Was Needed
The PACIFIC regimen established durvalumab after chemoradiotherapy as a standard for unresectable stage III NSCLC, but the benefit in EGFR-mutated disease has remained uncertain. EGFR-mutated tumors often have biological features associated with lower responsiveness to immune checkpoint inhibition, including lower tumor mutational burden and a less inflamed tumor microenvironment. This made the need for a dedicated EGFR-targeted strategy after definitive CRT particularly important.
Osimertinib already had strong precedent in EGFR-mutated lung cancer. It improved outcomes in metastatic EGFR-mutated NSCLC and later demonstrated major benefit in resected early-stage EGFR-mutated NSCLC through the ADAURA trial (Herbst et al., 2023). LAURA asked the next logical question: can osimertinib also improve outcomes after definitive chemoradiotherapy in unresectable stage III EGFR-mutated NSCLC?
How LAURA Was Designed
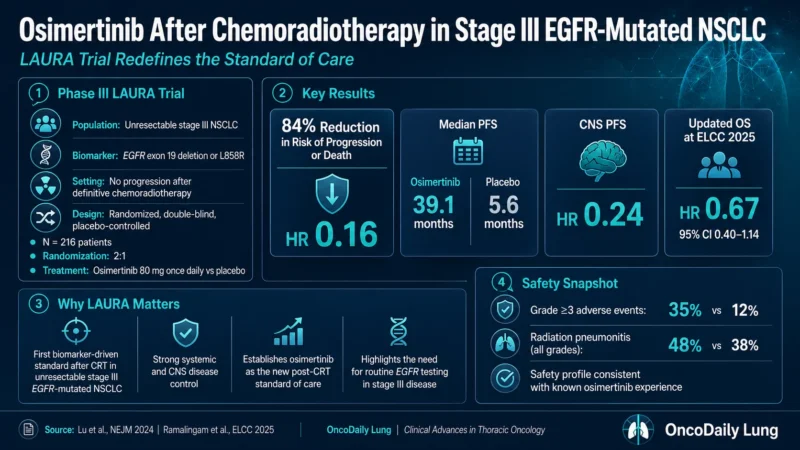
LAURA was a double-blind, placebo-controlled, randomized phase III trial enrolling patients with unresectable stage III NSCLC harboring EGFR exon 19 deletion or L858R mutation. Patients were eligible if their disease had not progressed during or after platinum-based chemoradiotherapy.
A total of 216 patients were randomized in a 2:1 ratio to receive osimertinib 80 mg once daily or placebo. Treatment continued until disease progression or unacceptable toxicity. The primary endpoint was progression-free survival by blinded independent central review. Key secondary endpoints included overall survival, central nervous system progression-free survival, and time to distant metastasis (Lu et al., 2024).
A Progression-Free Survival Result That Changed Practice
The primary LAURA results were striking. Osimertinib reduced the risk of disease progression or death by 84%, with a hazard ratio of 0.16 compared with placebo. Median progression-free survival was 39.1 months with osimertinib versus 5.6 months with placebo (Lu et al., 2024).
This magnitude of benefit is unusually large in stage III lung cancer and immediately positioned osimertinib as a defining post-CRT therapy for this molecular subgroup. The result also confirmed that EGFR mutation status is not only relevant in metastatic NSCLC or resected early-stage disease. It is now central to treatment planning in unresectable stage III disease as well.
CNS Protection Is A Core Part Of The Benefit
The CNS data from LAURA are especially important. Patients with EGFR-mutated NSCLC are at high risk for brain metastases during the course of their disease. Osimertinib is known for CNS activity, and in LAURA, osimertinib reduced the risk of CNS progression, with a reported hazard ratio of 0.24 for CNS progression-free survival in supplemental analyses (Lu et al., 2024).
This matters clinically because brain metastases affect survival, neurologic function, quality of life, and treatment complexity. In this setting, CNS protection is not a secondary advantage. It is one of the most important reasons osimertinib fits the biology of EGFR-mutated NSCLC after chemoradiotherapy.
Updated Survival Data Support The Direction Of Benefit
Updated overall survival data presented at ELCC 2025 showed a favorable survival trend, with an HR of 0.67 and a 95% confidence interval of 0.40–1.14. The OS data were still immature, with approximately 31% maturity, and interpretation was complicated by high crossover from placebo to osimertinib after progression. Around 80% of patients in the placebo arm who progressed received a third-generation EGFR TKI, mostly osimertinib, which can dilute the ability to show a statistically significant OS difference at interim analysis (Ramalingam et al., 2025).
For clinicians, the survival signal should be interpreted cautiously but reassuringly. The clearest and most mature benefit remains progression-free survival, while OS will require longer follow-up.
How LAURA Differs From PACIFIC
LAURA should not be viewed as a direct competitor to PACIFIC across all stage III NSCLC. The trials address different biological populations. PACIFIC enrolled an unselected unresectable stage III NSCLC population and established durvalumab maintenance after CRT. LAURA specifically studied patients with EGFR exon 19 deletion or L858R-mutated unresectable stage III NSCLC after CRT (Antonia et al., 2017; Lu et al., 2024).
This distinction is essential. For EGFR-mutated stage III NSCLC, LAURA provides biomarker-specific evidence that is more directly applicable than extrapolating from immunotherapy data in a broader population. The practical implication is that EGFR mutation testing must be performed early enough in unresectable stage III NSCLC to inform post-CRT treatment selection.

What This Means For Clinical Practice
The most immediate change is diagnostic. EGFR testing should now be routine in unresectable stage III nonsquamous NSCLC, and testing should not be reserved only for metastatic disease. If a patient has an EGFR exon 19 deletion or L858R mutation and does not progress after definitive chemoradiotherapy, osimertinib is now the evidence-based consolidation approach.
The second change is multidisciplinary workflow. Thoracic oncologists, radiation oncologists, pathologists, and molecular laboratories need a coordinated process so EGFR results are available before post-CRT treatment decisions are made. Delays in molecular testing can translate into delays in starting appropriate maintenance therapy.
The third issue is safety. Osimertinib’s safety profile in LAURA was generally consistent with prior experience, but the post-radiation setting requires careful attention to pneumonitis and interstitial lung disease. Radiation pneumonitis and drug-related ILD can overlap clinically and radiographically, making baseline documentation, symptom education, and rapid evaluation of new respiratory symptoms essential (Lu et al., 2024).
The Unanswered Questions After LAURA
Several questions remain. The optimal duration of osimertinib after chemoradiotherapy is still a key practical issue, especially for patients with prolonged disease control. The biology of resistance after CRT followed by osimertinib may also differ from resistance patterns seen in metastatic first-line osimertinib, and future studies will need to clarify whether MET amplification, EGFR C797S, histologic transformation, or other mechanisms dominate in this setting.
Another important question is how to manage patients who relapse after osimertinib in the stage III context. The next treatment may depend on resistance testing, site of relapse, prior radiation field, CNS involvement, and whether recurrence is oligoprogressive or systemic. Strategies combining osimertinib with MET inhibitors, antibody-drug conjugates, or other targeted agents remain important areas of investigation.
Clinical Takeaway
LAURA has changed unresectable stage III EGFR-mutated NSCLC by bringing precision oncology into a treatment setting that had long relied on a one-size-fits-most approach after chemoradiotherapy. Osimertinib after definitive CRT produced a major progression-free survival benefit, delayed CNS progression, and showed a favorable early survival trend despite substantial crossover.
The broader message is clear: molecular testing is no longer optional in stage III NSCLC. EGFR mutation status must be known before maintenance treatment decisions are made. For patients with unresectable stage III EGFR exon 19 deletion or L858R-mutated NSCLC who have not progressed after chemoradiotherapy, osimertinib is now the standard post-CRT strategy.