The antibody-drug conjugate era, which has already transformed breast oncology, is now firmly entering thoracic oncology. Datopotamab deruxtecan (Dato-DXd) received FDA accelerated approval in June 2025 for patients with locally advanced or metastatic EGFR-mutated non-small cell lung cancer following prior EGFR-targeted therapy and platinum-based chemotherapy. This approval represents the first TROP2-directed ADC indication in lung cancer and reflects a meaningful shift in the post-TKI treatment landscape, where options have historically been limited and largely chemotherapy-based.
Supported by the phase II TROPION-Lung05 and the phase III TROPION-Lung01 trials, this approval establishes Dato-DXd as a new mechanism-driven therapy in a population with persistent unmet need. At the same time, emerging first-line data suggest that this is only the beginning of a broader clinical role.

Read About FDA Approval on OncoDaily
A Mechanism Designed for Broad Targeting
Dato-DXd is built on a now familiar ADC platform but applied to a distinct biological target. It consists of a humanized anti-TROP2 monoclonal antibody linked via a cleavable tetrapeptide-based linker to DXd, a potent topoisomerase I inhibitor. This same payload has already demonstrated clinical success in other ADCs, most notably trastuzumab deruxtecan.
TROP2 is widely expressed across epithelial tumors, including most NSCLC histologies. Unlike many modern targets, its expression is not driven by specific genomic alterations, making it broadly applicable across patient populations. This feature allows Dato-DXd to function independently of driver mutation status, although its clinical activity varies by histology and molecular context.
The cleavable linker enables intracellular release of the cytotoxic payload and contributes to a bystander effect, allowing neighboring tumor cells to be affected even if TROP2 expression is heterogeneous. This design is particularly relevant in lung cancer, where tumor heterogeneity remains a major therapeutic challenge.
TROPION-Lung01: Establishing Activity in Pretreated NSCLC
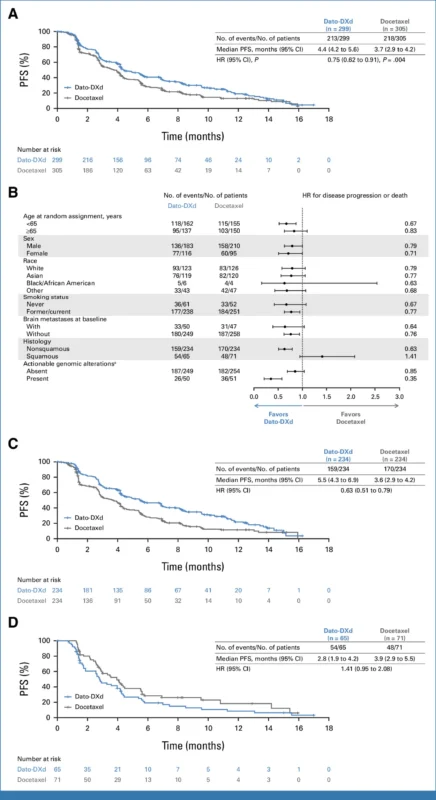
The phase III TROPION-Lung01 trial evaluated Dato-DXd versus docetaxel in patients with previously treated advanced NSCLC. The study demonstrated a statistically significant improvement in progression-free survival, with a median of 4.4 months compared to 3.7 months for docetaxel (HR 0.75; p = 0.004).
While the overall survival endpoint was not statistically significant, this finding must be interpreted cautiously. High rates of subsequent therapy and crossover, along with immature data at the time of analysis, limit the ability to detect OS differences in this setting.
The benefit was more pronounced in patients with nonsquamous histology, where median PFS reached 5.5 months compared to 3.6 months with docetaxel (HR 0.63), alongside a higher response rate. In contrast, patients with squamous NSCLC did not derive a meaningful benefit, highlighting an important limitation and reinforcing the need for careful patient selection.
EGFR-Mutated NSCLC: Where the Signal Becomes Clear
The strongest and most clinically meaningful activity of Dato-DXd emerged in patients with EGFR-mutated NSCLC. This subgroup ultimately drove regulatory approval.
In the phase II TROPION-Lung05 study, Dato-DXd demonstrated an objective response rate of 43.6% in EGFR-mutated patients who had progressed on targeted therapy and chemotherapy. Disease control approached 80%, and pooled analyses across studies reported a median PFS of 5.8 months and overall survival of 15.6 months.
These outcomes compare favorably to historical docetaxel data in this setting, where responses are typically limited and durability is modest. For clinicians managing post-osimertinib progression, Dato-DXd introduces a non-TKI, non-chemotherapy option with meaningful activity.
The biological rationale for this preferential efficacy remains under investigation. EGFR-mutated tumors are typically adenocarcinomas with relatively uniform TROP2 expression, potentially enhancing ADC binding and internalization. At the same time, these tumors tend to be immunologically “cold,” limiting the effectiveness of checkpoint inhibitors and leaving space for alternative mechanisms such as ADCs.
CNS Activity: A Clinically Relevant Signal
Brain metastases remain a major challenge in EGFR-mutated NSCLC, and any signal of intracranial activity is clinically meaningful.
Exploratory analyses from TROPION-Lung01 suggest that Dato-DXd may provide benefit in this setting. In patients with brain metastases and nonsquamous histology, the risk of CNS progression was substantially reduced compared to docetaxel. The effect appeared even stronger in the EGFR-mutated subgroup.
Although these findings are based on post-hoc analyses and require prospective validation, they align with an emerging pattern seen with other ADCs and suggest that Dato-DXd may contribute to CNS disease control.
Moving Into the First-Line Setting
The most forward-looking data for Dato-DXd come from the TROPION-Lung02 study, which explored its use in combination with pembrolizumab, with or without chemotherapy, in the first-line setting.
The study demonstrated encouraging activity across treatment arms, with durable responses and manageable toxicity. Interestingly, the chemotherapy-free combination of Dato-DXd plus pembrolizumab showed longer treatment duration compared to the triplet regimen, suggesting that ADC–immunotherapy combinations may offer a viable strategy to reduce chemotherapy exposure.
These findings are now being tested in the phase III TROPION-Lung08 trial, which will determine whether Dato-DXd can challenge or even replace chemotherapy-containing regimens in selected patients.
Safety Profile: Familiar but Not Benign
The safety profile of Dato-DXd is broadly consistent with other DXd-based ADCs but requires careful management.
Common adverse events include nausea, stomatitis, and fatigue, which are generally manageable. Importantly, rates of grade 3 or higher adverse events were lower compared to docetaxel, supporting a more favorable tolerability profile overall.
However, interstitial lung disease remains the most critical toxicity. Although severe cases are relatively uncommon, ILD can be life-threatening if not recognized early. Routine monitoring, prompt evaluation of respiratory symptoms, and early corticosteroid intervention are essential components of safe administration.
Where Does Dato-DXd Fit Today?
The current role of Dato-DXd is clearest in EGFR-mutated nonsquamous NSCLC after progression on targeted therapy and chemotherapy, where it now represents an FDA-approved option with demonstrated efficacy.
In the broader nonsquamous population without driver mutations, the drug has shown meaningful activity and is under further regulatory evaluation. In squamous NSCLC, current evidence does not support its use.
Looking ahead, the most important question is whether Dato-DXd can move earlier in the treatment paradigm. If ongoing phase III trials confirm the first-line activity seen in early studies, ADC-based strategies may begin to reshape initial treatment approaches in NSCLC.

Read About Datopotamab Deruxtecan on OncoDaily
A Structural Shift in Thoracic Oncology
The arrival of Dato-DXd is not an isolated event. It signals a broader transition in thoracic oncology toward ADC-based therapies targeting surface antigens rather than genomic alterations alone.
For years, lung cancer treatment has been defined by targeted therapy and immunotherapy. ADCs introduce a third pillar, one that leverages tumor biology in a different way and may bridge gaps where other strategies fall short.
The approval of a TROP2-directed ADC in lung cancer marks the beginning of this shift. The next few years will determine how far it extends.


