For more than two decades, cisplatin-based neoadjuvant chemotherapy followed by radical cystectomy has represented the standard treatment for muscle-invasive bladder cancer (MIBC).
Despite this approach, nearly 50% of patients ultimately develop metastatic recurrence, highlighting persistent unmet need for more effective perioperative strategies.
Antibody–drug conjugates (ADCs) and immune checkpoint inhibitors have independently demonstrated substantial activity in advanced urothelial carcinoma. The phase III KEYNOTE-B15 trial evaluated whether integrating enfortumab vedotin (EV) — a Nectin-4–directed ADC — with pembrolizumab could improve outcomes compared with platinum-based chemotherapy in the curative-intent setting.
Study Design
KEYNOTE-B15 was a randomized, open-label phase III trial enrolling 808 patients with cisplatin-eligible MIBC scheduled for radical cystectomy.
Patients were randomized to receive:
- Enfortumab vedotin + pembrolizumab administered both before and after cystectomy, or
- Standard gemcitabine + cisplatin neoadjuvant chemotherapy, followed by cystectomy and observation.
The primary endpoint was event-free survival (EFS) assessed by blinded independent central review.
Key secondary endpoints included overall survival (OS) and pathologic complete response (pCR).
Results
Event-Free Survival
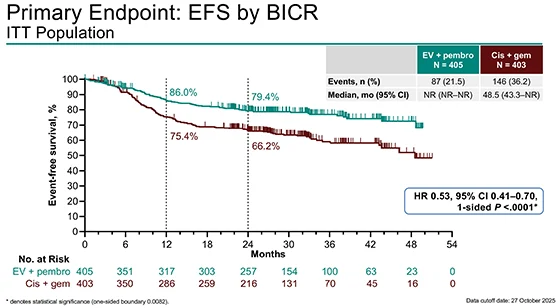
The trial met its primary endpoint, demonstrating a significant improvement in EFS with EV plus pembrolizumab.
- Median EFS: Not reached with EV + pembrolizumab
- Median EFS: 48.5 months with cisplatin + gemcitabine
- HR 0.53 (95% CI 0.41–0.70; P < 0.0001)
This corresponds to a 47% reduction in recurrence, progression, or death.
Landmark EFS rates further supported durable benefit:
- 12 months: 86.0% vs 75.4%
- 24 months: 79.4% vs 66.2%
Benefit was consistent across major clinical subgroups, including PD-L1 status, age, sex, and geographic region.
Pathologic Complete Response
A major improvement was observed in surgical outcomes:
pCR rate:
- 55.8% with EV + pembrolizumab
- 32.5% with chemotherapy (P < 0.0001)
Among patients undergoing surgery:
- 64.4% vs 36.3%, respectively.
These findings suggest substantially deeper tumor eradication prior to cystectomy.
Safety
Treatment-emergent adverse events occurred in nearly all patients in both groups.
Grade ≥3 adverse events:
- 75.7% with EV + pembrolizumab
- 67.2% with chemotherapy
Key toxicities included:
- EV-related skin reactions (63.5%)
- Peripheral neuropathy (36%)
- Hyperglycemia (13.2%)
- Immune-related hypothyroidism (12.2%)
- Pneumonitis (6.9%)
No unexpected safety signals were identified, confirming feasibility of prolonged perioperative therapy.
Clinical Interpretation — What Changes Compared With Prior Practice
KEYNOTE-B15 represents the first phase III trial demonstrating superiority of a non-platinum regimen over cisplatin-based neoadjuvant chemotherapy in MIBC.
Historically:
- Treatment relied on cytotoxic chemotherapy alone
- Many patients remained ineligible for cisplatin
- Recurrence risk remained high despite surgery
This study introduces a perioperative immune-ADC strategy that:
- Improves EFS and OS,
- Nearly doubles pCR rates,
- Moves treatment beyond chemotherapy dependence.
Importantly, the results support the emergence of a cisplatin-chemotherapy–free curative pathway, integrating systemic immune activation and targeted cytotoxic delivery.
Key Takeaway Messages
- EV + pembrolizumab significantly improves EFS and OS versus standard cisplatin-based therapy in MIBC.
- The regimen achieves markedly higher pathologic complete response rates, indicating deeper preoperative tumor clearance.
- This is the first successful non-platinum perioperative strategy to outperform historical standard therapy.
- Findings position EV + pembrolizumab as a potential new standard of care for eligible patients undergoing radical cystectomy.
- Future work will likely integrate ctDNA-guided postoperative decision-making to optimize adjuvant treatment intensity.
Read Full Article Here.


