This week in OncoDaily Immuno-Oncology, expert insights highlight the expanding frontiers of immune-based cancer care — from whole-cell cancer vaccines and next-generation CAR-T strategies targeting the tumor microenvironment, to emerging approaches that turn the tumor’s own immune shield against itself.
Clinical perspectives emphasize the growing importance of immune-related toxicities, including rare but severe events like ICI-induced diabetes, alongside biomarkers such as lymphocyte counts shaping treatment response. At the same time, advances in screening and immune profiling are redefining early detection and patient selection.
Across disciplines, these developments reflect a shift toward integrated, biomarker-driven, and more inclusive immunotherapy strategies.
This Week’s Expert Highlights in Immuno-Oncology
Aditi Dubey (Research Intern at the ICMR National Institute for Implementation Research on Non-Communicable Diseases):
“I’m pleased to share my recent article exploring whole-cell cancer vaccines, a promising avenue in cancer immunotherapy that utilizes entire tumor cells to elicit a broad and potent immune response.
The article delves into :
Mechanistic insights into irradiated and genetically modified cells.
Comparative analysis of autologous vs. allogeneic vaccine approaches.
Clinical outcomes, safety profiles, and translational challenges
Emerging strategies for combination therapies and personalized immunomodulation
This article aims to contribute to ongoing discussions on optimizing vaccine design and integrating these platforms into next-generation oncology treatments.”
Ada Hang-Heng Wong (Editor-in-Chief of Advanced Oncology):
“It is my pleasure to have met all of you (and missed some ) during the AACR2026 Annual Meeting held in San Diego, California, on April 18-22, 2026. I had a wonderful time and enjoyed the talks and poster sessions – I wish I had more time to go through all of them! I hope you enjoyed our limited-edition advanced Oncology cookies illustrating IL2-expressing synthetic cells that stimulate freshly isolated and cryopreserved NK cells’ cytotoxicity
This year, the pan-KRAS inhibitor highlights the most important discovery in targeted therapy ; ADCs, bispecifics, T cell engagers, CAR-Ts remain under the spotlight of cancer immunotherapy, Biomni made into the plenary talk, opening a new era of agentic AI not only in responding to prompts but upgrading to hypothesis generation and actionable thinking . Other highlights of this year’s meeting include cancer prevention and pre-cancer studies .
Even though this meeting has come to an end, our march towards combating cancer continues . Advanced Oncology welcomes research showcasing groundbreaking discoveries from the bench to paradigm changing practices in the clinic – please visit.”
Kevin Kelley (Business Development and Project Manager at Accurant Biotech, Inc.):
“Chimeric antigen receptor (CAR) T-cell therapy is an impactful treatment for blood cancers, such as leukemia and lymphoma. However, the immunotherapy has struggled against solid tumors, as tumor cells often do not share one consistent surface target. Additionally, many solid tumors are protected by a dense network of scar tissue and immune-suppressive cells that block T-cells.
To address these challenges, researchers from Memorial Sloan Kettering (MSK) Cancer Center have developed a new CAR T-cell that attacks supportive cells in the tumor microenvironment that bear the surface protein, urokinase plasminogen activator receptor (uPAR). The work is described in a new study published in Cell titled, “A convergent uPAR-positive tumor ecosystem creates broad vulnerability to CAR T-cell therapy.””
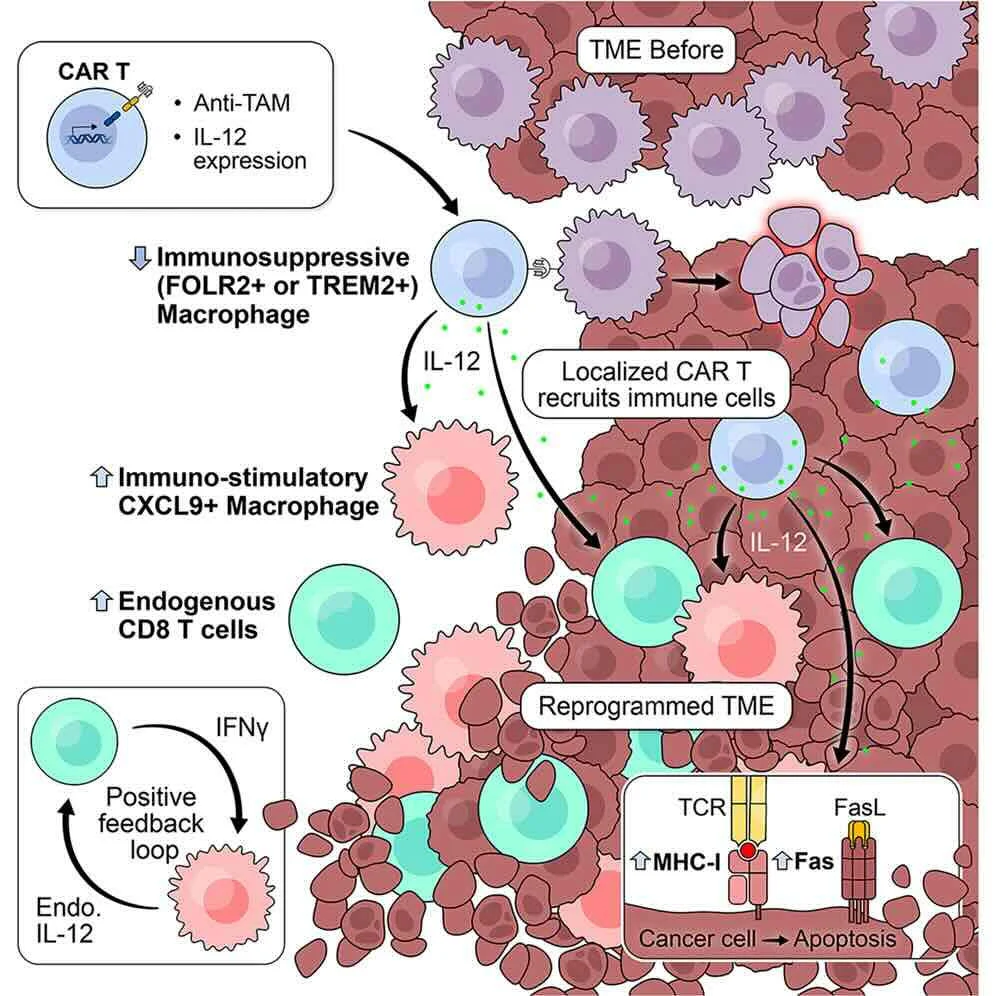
Lidiane Garcia (PhD Neuroscientist, Science Writer, and Project Manager at MolecularMente):
“When Cancer Loses Its Guards: Scientists Turn The Tumor’s Immune System Against Itself
Researchers have just revealed a promising strategy that could change the way we fight cancer: transforming the very cells that protect the tumor into weapons against it.
By eliminating this natural “shield” and activating the local immune system, the new immunotherapy has managed to destroy aggressive tumors and create a lasting defense against the disease, an advance that could pave the way for much more effective treatments against solid cancers… “
Hamdy Alkady (Endocrinology Consultant, MRCP (UK), FRCP (London & Glasgow), MSc, European Board of Endocrinology & Diabetes, Clinical Fellow of EASO):
” I am honored to share my recent scientific contribution at the AACE 2026 American Association of Clinical Endocrinology, one of the most influential and academically respected global meetings in clinical endocrinology. The AACE Congress continues to set international standards for excellence in endocrine science, clinical innovation, and multidisciplinary collaboration.
During the congress, I presented a poster titled: “Immune Checkpoint Inhibitor–Induced Diabetic Ketoacidosis in a Patient With Triple-Negative Breast Cancer.”
I am also proud that this work has been published in Endocrine Practice (April 2026), a leading Q1 journal in our field.
DOI: 10.1016/j.eprac.2026.01.170
ISSN: 1530‑891XCase Summary
Immune checkpoint inhibitors (ICIs) are increasingly used for solid tumors but can trigger immune‑related adverse events. While thyroiditis and hypophysitis are relatively common, ICI‑induced diabetes mellitus remains rare yet potentially life‑threatening.Our patient developed severe diabetic ketoacidosis (DKA) shortly after pembrolizumab therapy, with irreversible insulin deficiency, confirmed by persistently undetectable C‑peptide and negative autoantibodies. The abrupt onset and severity distinguish ICI‑induced diabetes from classic type 1 diabetes.
This case reinforces the need for high clinical vigilance, even in patients without diabetes risk factors. Early recognition, prompt DKA management, and multidisciplinary follow‑up are essential as ICIs become standard in oncology practice.”
Ankit Bharat (Professor & Chief, Executive Director, Canning Thoracic Institute):
“Eight years ago, the five-year lung cancer survival rate was 18%.
Today it stands at 28.4%.
Behind that number is a generation of progress: better screening, more precise surgery, smarter targeted therapies, and immunotherapy. The American Lung Association’s latest State of Lung Cancer report makes those gains tangible.
But the same report shows where we still fall short.
Only 18.2% of high-risk Americans get screened with low-dose CT, the single intervention proven to cut lung cancer mortality by up to 20%. Forty-three percent of patients are still diagnosed at a late stage. Black patients have a survival rate of 24.9% compared to 28.6% for white patients. Indigenous patients fare even worse at 22.8%. Twenty-one percent of cases receive no treatment at all.
And here is the part that is often missed: the current screening guidelines leave out most of the people getting lung cancer.
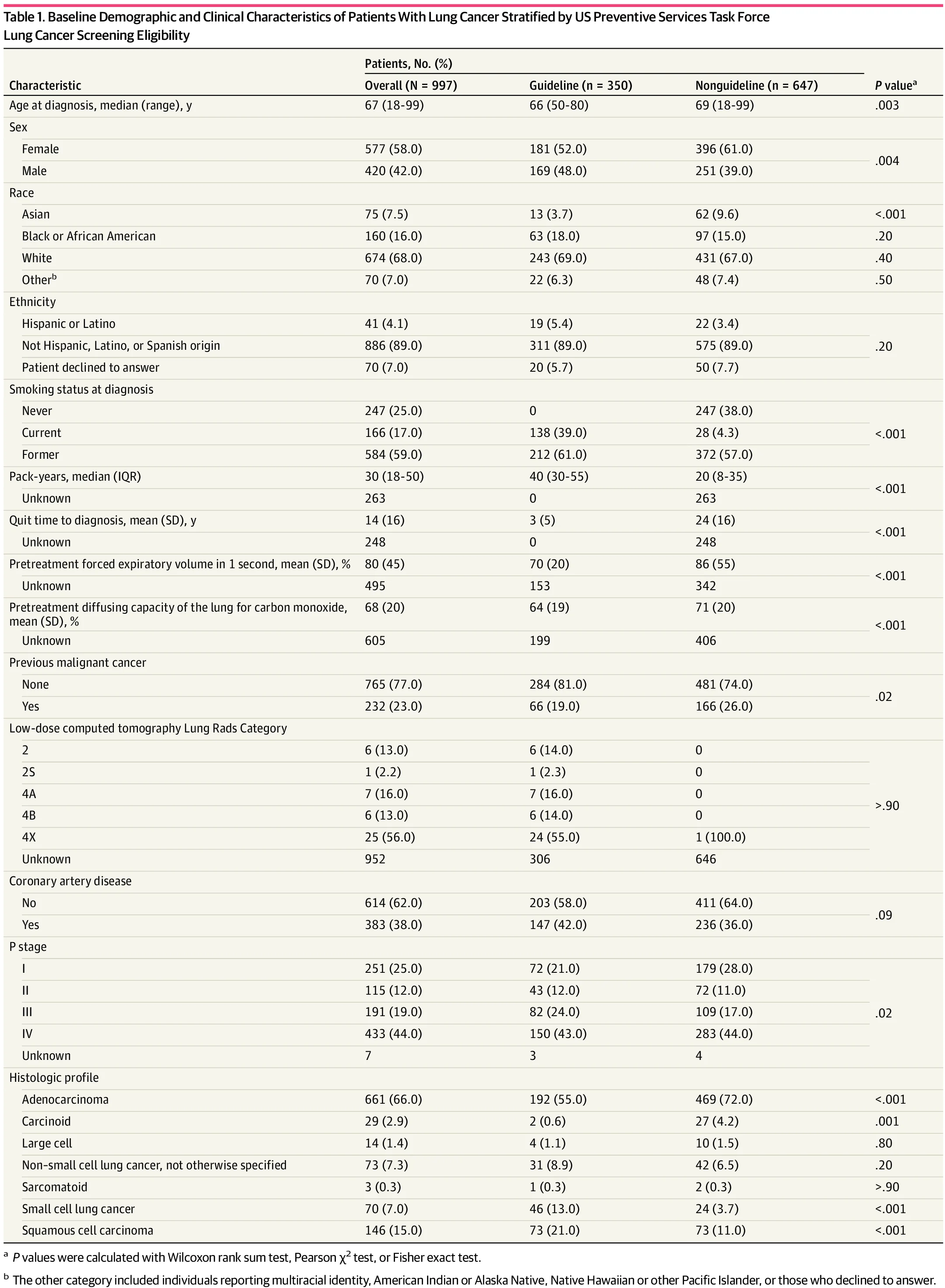
Our study published in JAMA Network Open found that only 35% of newly diagnosed lung cancer patients actually met the USPSTF screening criteria. The 65% who were missed included 38% who were never-smokers, more women, and more Asian patients. Current guidelines, built around smoking history, are structurally excluding the fastest-growing populations of lung cancer patients.
Age-based screening (ages 40-85) captured 93.9% of cases in our study and was six-fold more cost-effective than current breast and colorectal screening programs. This is not just a clinical argument. It is an equity argument.
That is why at Northwestern Medicine, we have launched universal age-based lung cancer screening. It is free for patients. It takes less than five minutes. No smoking history required.
If screening only reaches the patients we already expect to get cancer, we will keep missing the ones we do not.”
Barnini Ghosh (Consultant Medical Oncologist at Ruby General Hospital):
“Honoured to share our publication on immunotherapy in MSI-H/dMMR Early Colorectal Cancer, written under the guidance of Dr. Amol Akhade, Deeply grateful for the insights! “

Pat Soon-Shiong, (Chairman of Chan Soon-Shiong Family Foundation, Executive Chairman at ImmunityBio, and Executive Chairman of the Los Angeles Times):
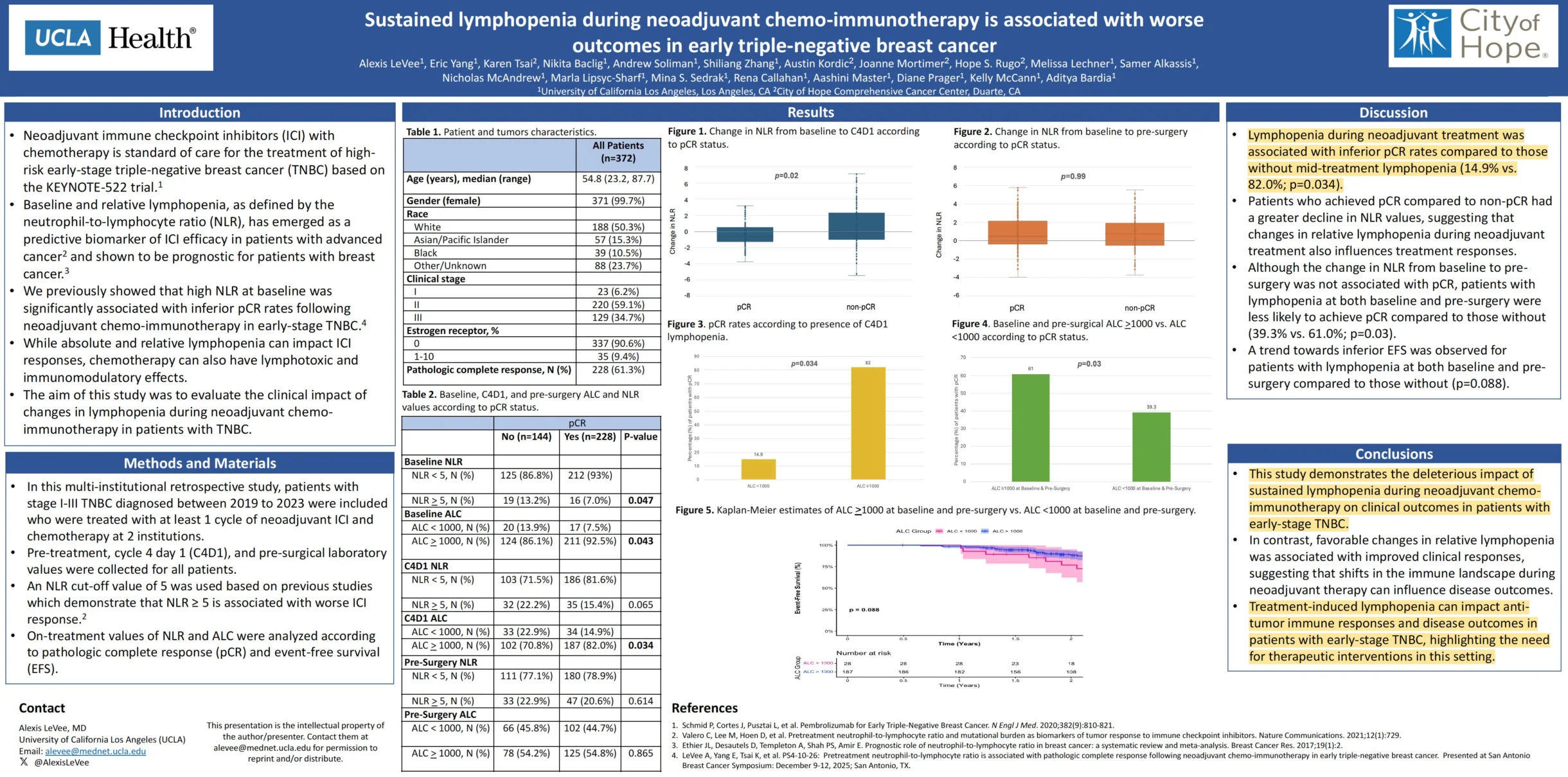
” The evidence generated by UCLA is compelling. This presentation entitled, “Sustained lymphopenia during neoadjuvant chemo-immunotherapy is associated with worse outcomes in early triple-negative breast cancer” was presented at ASCO-IO (Feb 2026).
ASCO-IO (Feb 2026): Early Stage Triple Negative Breast Cancer (TNBC)
In a 372 patient study, the data shows that only 15% of patients with early stage TNBC have a pathological complete response (pCR) when the ALC is less than 1,000 cells while 82% of patients with NK and T cells (ALC greater than 1,000 cells) have a complete response after therapy. This difference is stark and significant (p=0.034).
Dr Bardia states:”Sustained lymphopenia and the development of relative lymphopenia during neoadjuvant chemo-immunotherapy may reflect impaired anti-tumor immune responses in patients with early-stage TNBC, highlighting need for intervention therapeutics in this setting“
Conclusion: Treatment-induced lymphopenia can impact anti-tumor immune responses and disease outcomes in patients with early-stage TNBC, highlighting the need for therapeutic interventions in this setting”
Saied Froghi-Locum, (Consultant Surgeon at The Hillingdon Hospitals NHS Foundation Trust):
“Cholangiocarcinoma remains one of the most challenging cancers we face – biologically complex, immune-resistant, and clinically unforgiving.
Delighted to share our latest review in Frontiers in Immunology, where we present an integrated roadmap for overcoming these barriers through advanced cellular immunotherapy.”

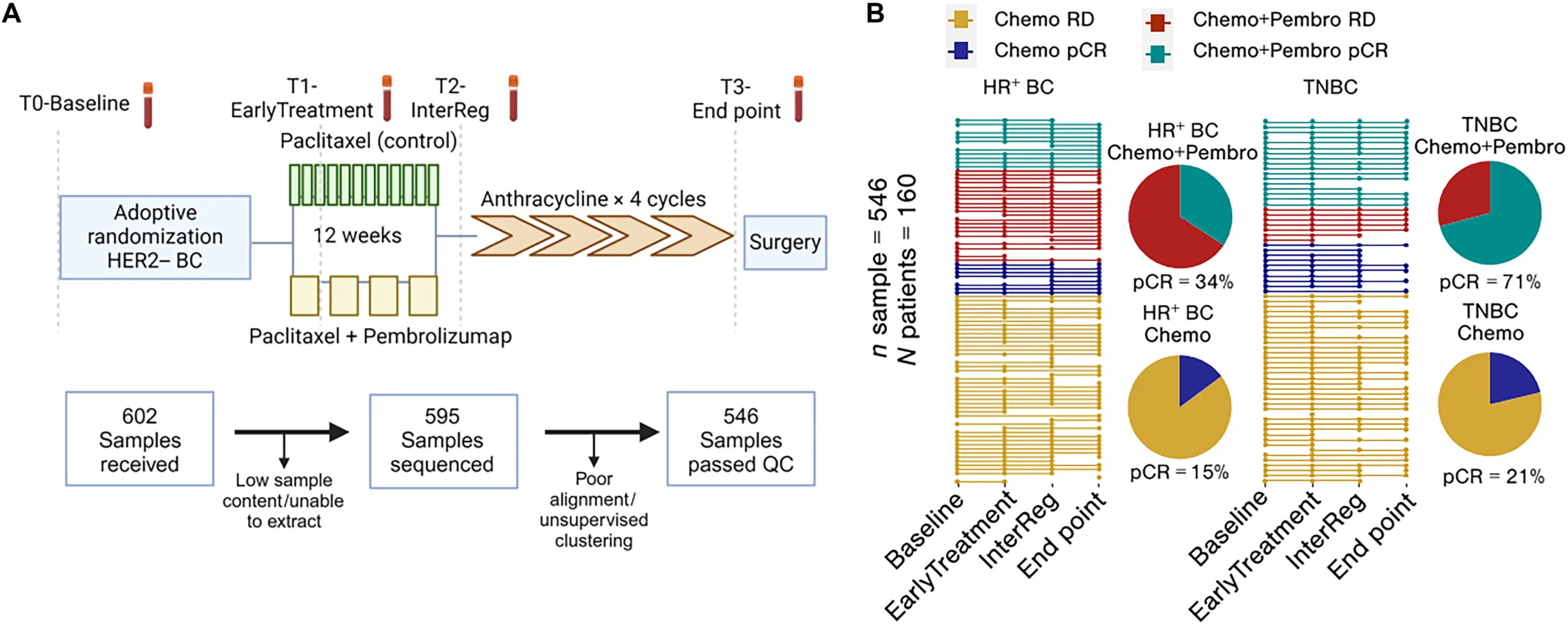
Laura Esserman, (Principal Investigator at The WISDOM Study):
” It’s exciting to see yet another way of identifying immune responsive tumors, this time from the peripheral blood in the I-SPY trial. These tumors are clearly distinct and have much better responses, strengthening our approach to adding an immune phenotype as part of standard tumor classification.
I certainly agree with the editors of Science Translational Medicine that the blood-based biomarkers for cancer are likely to be applicable across cancers regardless of organ of origin. It is also likely that the expression profiling that identifies these immune responsive tumors will be more broadly applicable.
The response predictive subtyping that we’re using now in I-SPY demonstrates the importance of an immune phenotype, which is much more important than hormone receptor in predicting response to immunotherapy. We are excited to seeing both of these types of profiling move forward.”