This week in OncoDaily Immuno-Oncology, new discoveries and translational insights are redefining how we optimize immune-based cancer care — from lncRNAs driving therapy resistance and microbiome-derived biomarkers shaping prevention strategies, to real-world validation of Enfortumab Vedotin and landmark FDA approvals for perioperative immunotherapy in gastric cancer. Highlights include PPP2R1A mutations predicting improved ICB outcomes, IL-9–enhanced CAR-T potential, and novel CD8+ T cell niches (CRATERs) linked to melanoma response. Across tumor types, these advances reflect a rapidly evolving era of smarter, more durable, and globally accessible immunotherapy.
Wafik S. El-Deiry (Director of Legorreta Cancer Center at Brown University and Chair of the WIN Consortium in Cancer Personalized Medicine):
“New lncRNA Drives Cisplatin Resistance in Lung Cancer”
“Cracking TNBC’s Immune Code: New Clues for Smarter Treatment
A comprehensive tumor immunoprofiling study revealed distinct immune microenvironment signatures across triple-negative breast cancer (TNBC) subtypes. Differences in T-cell infiltration, myeloid cell polarization, and checkpoint expression correlate with therapeutic responsiveness to chemo-immunotherapy combinations.
The work supports biomarker-guided stratification and rational design of combination regimens. Validation across multi-omics cohorts and prospective trials will be essential for clinical translation. The findings may refine patient selection for immune checkpoint blockade and novel myeloid-targeting strategies.”
Anas Younes (Chief Medical Officer and Head of Haematology R&D at AstraZeneca):
“Engineered T cells that have been used to treat ulcerative colitis, rheumatoid arthritis and lupus show promising results.”
Giuseppe Banna (Consultant Medical Oncologist at Portsmouth Hospitals University NHS Trust):
“Our New Real-World Evidence Published! Multicenter study on Enfortumab Vedotin (EV) in metastatic urothelial carcinoma (mUC) is now out in European Urology Open Science.
After the landmark EV-301 trial established EV’s efficacy, a critical question remained: How does it perform in the diverse, real-world patient population, especially after a specific sequence of platinum-based chemotherapy (PBC) followed by pembrolizumab (PEM)?
The ARON-2EV global project set out to answer this. We analyzed data from 401 patients across 24 countries who received this exact treatment sequence.
Here are the key findings:
Consistent Efficacy: In this real-world setting, EV demonstrated robust activity.
Median Overall Survival (OS): 12.3 months
Overall Response Rate (ORR): 45%
These results strongly align with the pivotal EV-301 trial, confirming EV’s vital role in the post-platinum and post-immunotherapy space.
A Predictive Signal? We discovered a significant association between a patient’s response to prior pembrolizumab and their subsequent outcomes with EV.
Patients who achieved a Complete or Partial Response (CR/PR) to PEM had significantly better OS and Time on Treatment (ToT) with EV.
Crucially, EV was still effective even in patients refractory to PEM, with an ORR of 35%. This underscores its value across different clinical scenarios.
Why it matters:
This large, international analysis provides robust confirmation of EV’s effectiveness in everyday practice. It also offers clinicians valuable insights for patient counseling and suggests that prior immunotherapy outcomes could help inform expectations for subsequent ADC therapy.
This work would not have been possible without the incredible collaboration of all the co-authors and participating centers in the ARON-2EV project led by Matteo Santoni.”
Jorge Timoteo (Director Medical Affairs at Abbott):
“Progress against lung cancer is possible when science, policy, and collaboration align. Peru is taking an important step forward with the introduction of Abbott’s first PD-1 immunotherapy for extensive-stage small cell lung cancer (ES-SCLC) – a milestone for Latin America.
This innovative treatment, backed by global clinical evidence and regulatory approval, offers patients longer survival and improved quality of life. At Abbott, we are committed to expanding access to advanced oncology therapies and driving meaningful change for patients.
Learn more about this cutting-edge biologic treatment.
Read the full article of Business empresarial here.”
Eduardo De la Guardia (Clinical Research Coordinator – Oncology and Urology at University Clinic of Navarra):
“The FDA has approved durvalumab (Imfinzi) plus FLOT chemotherapy as a perioperative (neoadjuvant and adjuvant) treatment for resectable gastric or gastroesophageal junction cancer.”
Raffaele Di Giacomo (Director at ByronInsight AG):
Lung cancer immunotherapy has jumped from last-ditch option to reshaping survival curves in just a few years. In advanced NSCLC, pembrolizumab roughly doubles progression-free survival vs chemo and is producing 5-year survivors that used to be rare.
[1] Now IO is moving into early-stage, resectable NSCLC: perioperative/adjuvant pembrolizumab and CheckMate 77T nivolumab show event-free survival gains, forcing hospitals to redesign pathways and testing. [2][3]
Where do you see lung cancer IO most overpromised today – trial design, biomarkers, access, or something else?”
Sciqst: AI-powered literature reviews in minutes.
Generate comprehensive, up-to-date scientific reviews from the latest papers with one click.”
Mario Balsa (Medical Oncology Resident (MIR) at ICO – Bellvitge University Hospital):
“Why do some patients respond better to immunotherapy?
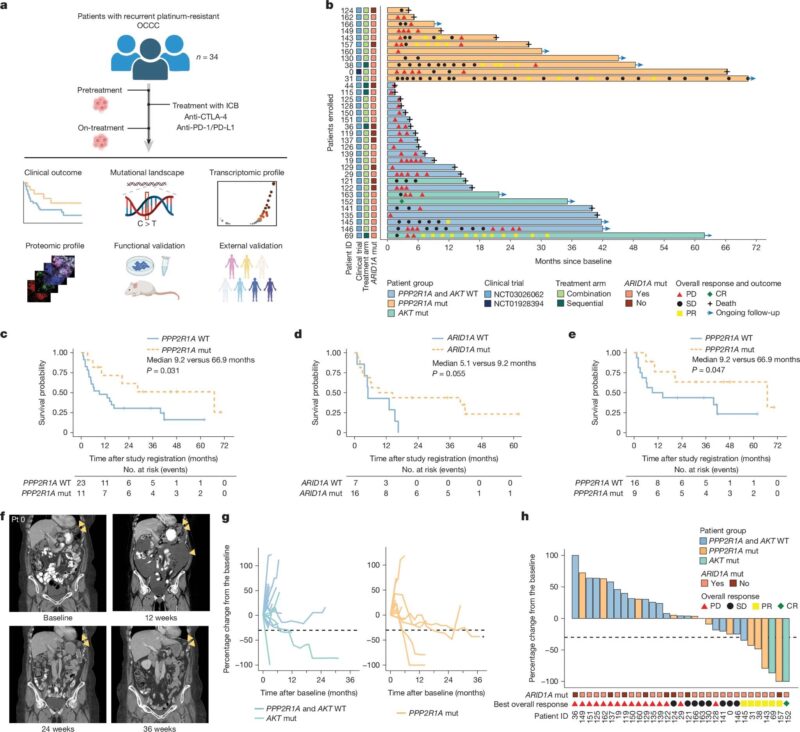
Nature: PPP2R1A mutations linked to improved outcomes with ICB! Across cancer types, PPP2R1A-mutant tumors show: increased OS and PFS after ICB ↑ IFN-γ signalling and T-cell infiltration Preclinical data: targeting PP2A boosts ICB and CAR-T efficacy If a scaffold protein becomes a signal…biology tells where therapy should go next!”
Eric Topol (Founder and Director at The Scripps Research Institute):
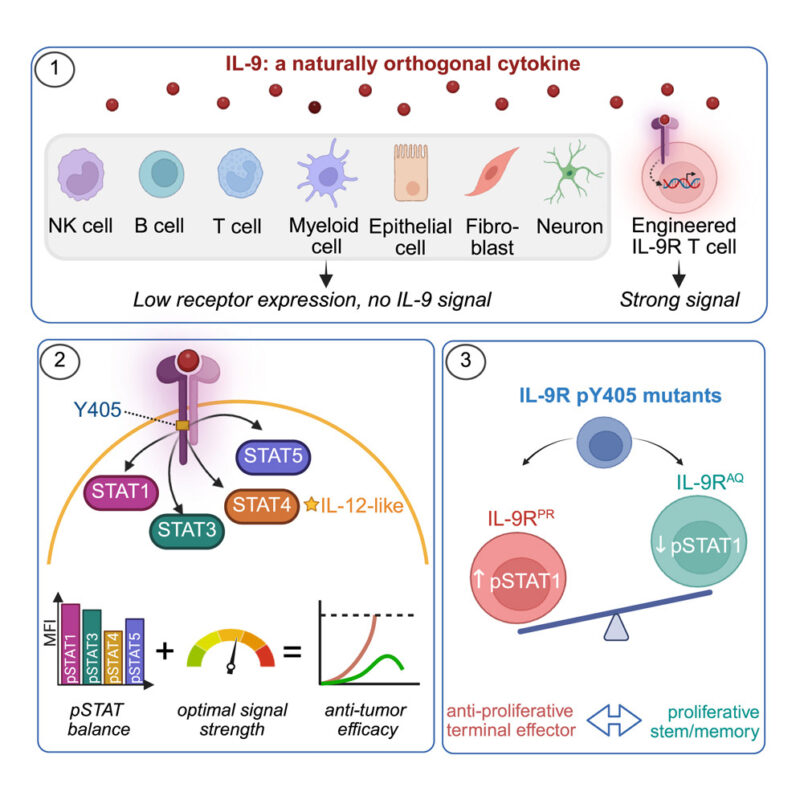
“Engineering T cells (CAR T) for treatment of solid cancers has had mixed success to date. Two reports today raise the promise of using interleukin-9 as a way to raise efficacy.”
Jose Fernando Moura (Member of the Oncology Commission at Cremepe):
“New insight in melanoma immunotherapy: CRATERs — tiny tumor niches where CD8+ T cells gather, recognize, and kill cancer cells more effectively. >CRATERs after immunotherapy = better clinical response. Small pockets. Big meaning.”
Written by Rima Grigoryan, MDc, Assistant Editor of OncoDaily IO.


