This week in OncoDaily Immuno-Oncology, expert insights highlight how the field continues to evolve through deeper biological understanding and innovative therapeutic strategies. Discussions range from the role of circadian biology and treatment timing in immunotherapy, to emerging concepts such as tumor-intrinsic checkpoint signaling and macrophage-targeted therapies.
New research also explores T-cell temporal dynamics under checkpoint blockade, advances in tumor microenvironment and vascular biology, and updated clinical trial data including the LEAP-010 study in head and neck cancer. Additional perspectives emphasize multidisciplinary progress in bladder cancer immunotherapy, growing momentum in AI-driven precision oncology, and the development of next-generation T-cell engagers and intratumoral immunotherapy strategies.
Together, these highlights reflect an immuno-oncology landscape increasingly shaped by mechanistic insight, translational research, and more precise immune-based cancer therapies.

10 Must-Read Posts in Immuno-Oncology This Week
This Week’s Expert Highlights in Immuno-Oncology
Janet Maendel (Integrative Metabolic Oncology Consultant, Clinical Advisor at Nurture & Restore Wellness):
“Immuno-oncology has transformed the way we think about cancer treatment. Yet one of the least discussed variables in this field is time.
In clinical practice, we often focus on therapies, biomarkers, and treatment protocols. But timing – when immune activation occurs, when therapies are delivered, and how the body’s biological rhythms interact with treatment – may be just as important.
In this article, I explore what I call the quiet tension of time in immuno-oncology: the space between biological readiness and clinical intervention. It’s a reminder that the immune system operates in rhythms, not static snapshots, and that understanding these rhythms may influence how we design and sequence therapies.
As immunotherapy continues to evolve—from checkpoint inhibitors to cellular therapies—the conversation must expand beyond what we treat with, to also consider when and how treatments align with the body’s underlying biology.
This perspective is especially relevant as emerging research highlights how circadian biology and immune activity influence treatment response and outcomes.”

Vahid Vahedian (Post Doctoral Fellow in Medical Oncology and Hematology at Advanced Cancer Therapeutics and Cellular Therapy at Clinical Hospital, Faculty of Medicine, University of São Paulo):
“I am pleased to share a meaningful milestone from the “3rd Cancer Thera Congress”, held at the Faculty of Medical Sciences, University of Campinas (UNICAMP), Campinas, Brazil, on March 12–13, 2026.
During the congress, I presented my poster entitled: “Checkpoint Molecules on Tumor Cells: A Paradigm Shift in Cancer Therapy.”
I am honored that this work was recognized by the scientific evaluators and congress board as the “Best Review Poster” in the field of cancer therapeutics.
This analytic perspective work explores the emerging concept of tumor-intrinsic immune checkpoint signaling and highlights how these checkpoint molecules expressed by tumor cells may directly influence tumor biology and therapeutic responses which represents an evolving viewpoints in future estrategies in clinical application of precision oncology and immuno-oncology. The congress also provided an excellent opportunity for scientific exchange with leading researchers and clinician-scientists. I was particularly honored to meet and discuss with Prof.Giuseppe Saglio whose pioneering contributions to translational hematology and oncology have significantly shaped the field.
I would like to deeply express my sincere gratitude to my scientific mentor, Prof.Eduardo Rego, for his continuous guidance and support. I also thank my collaborator and co-author,Prof.João Agostinho Machado-Netoão, for his valuable contributions to this conceptual shift perspective work. The abstract of this study has been published in the congress supplement issue of the journal Hematology, Transfusion and Cell Therapy.
Publication link:
I am grateful for the inspiring discussions and scientific exchange during this congress and look forward to continuing close collaborative efforts advancing translational cancer research, immunotherapy, and precision oncology.”

José Luis Torres Cuadros (Associate Director of Clinical Trial Management and Professor at Aliat Universidades):
“Macrophages: Targets for next-generation cancer immunotherapy ”
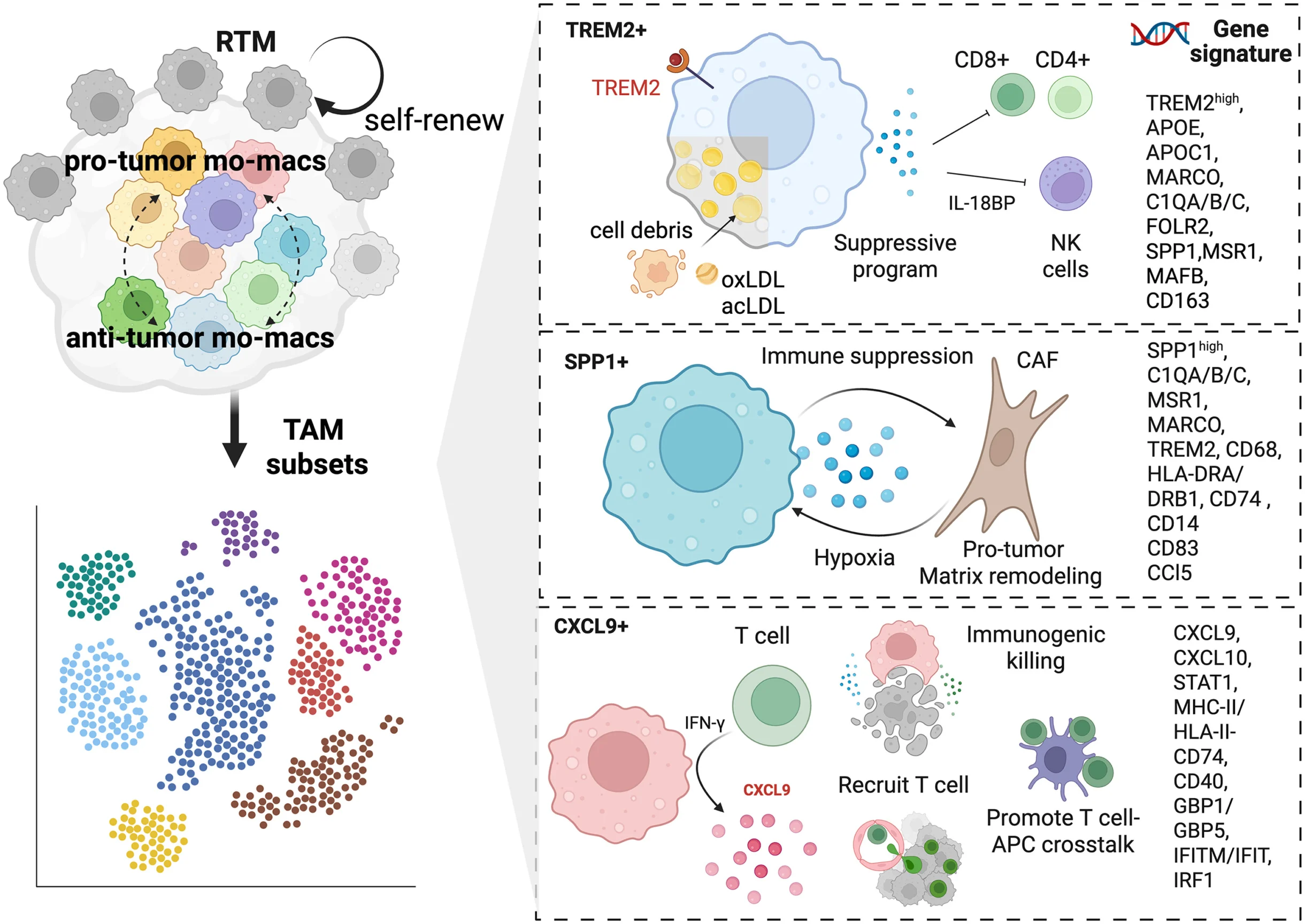
Jozafina Haj Shomaly (Postdoctoral researcher in Queen Mary University of London):
“After months of reading, discussing, and writing, I’m happy to share that our new review has been published in Journal of Cell Science!
In this Perspective, we discuss the interplay between cancer cells and immune and vascular cells, highlighting how a detailed understanding of the cell biology
of the tumour microenvironment might advance current therapies. Additionally, we describe alternative vascular modulation approaches that could potentially improve the effectiveness of immune checkpoint
inhibitors in lung cancer treatment.
Grateful to work with such great co-authors on this piece Alejandro Castillo-Kauil, Kairbaan Hodivala-Dilke and Gabriela D’Amico at the Barts Cancer Institute”

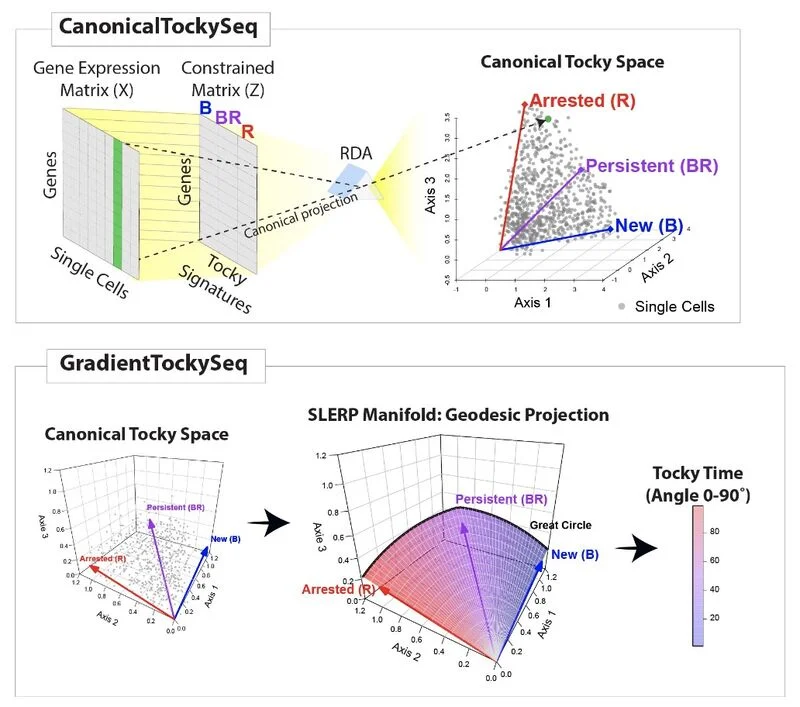
Masahiro Ono (Principal Investigator advancing cancer immunology and autoimmunity research through integrated experimentation and computation at Imperial College London):
“Delighted to share that our new bioRxiv preprint is now available: “Temporal Mechanisms of T-Cell Fate Decisions under Immune Checkpoint Blockade Resolved by CanonicalTockySeq”.
In this study, we present a new approach for resolving T cell temporal states at single-cell resolution, and apply it to cancer immunotherapy to investigate the time-dependent mechanisms through which immunotherapy promotes anti-cancer immune responses.
My sincere thanks to all co-authors and collaborators for their contributions.”
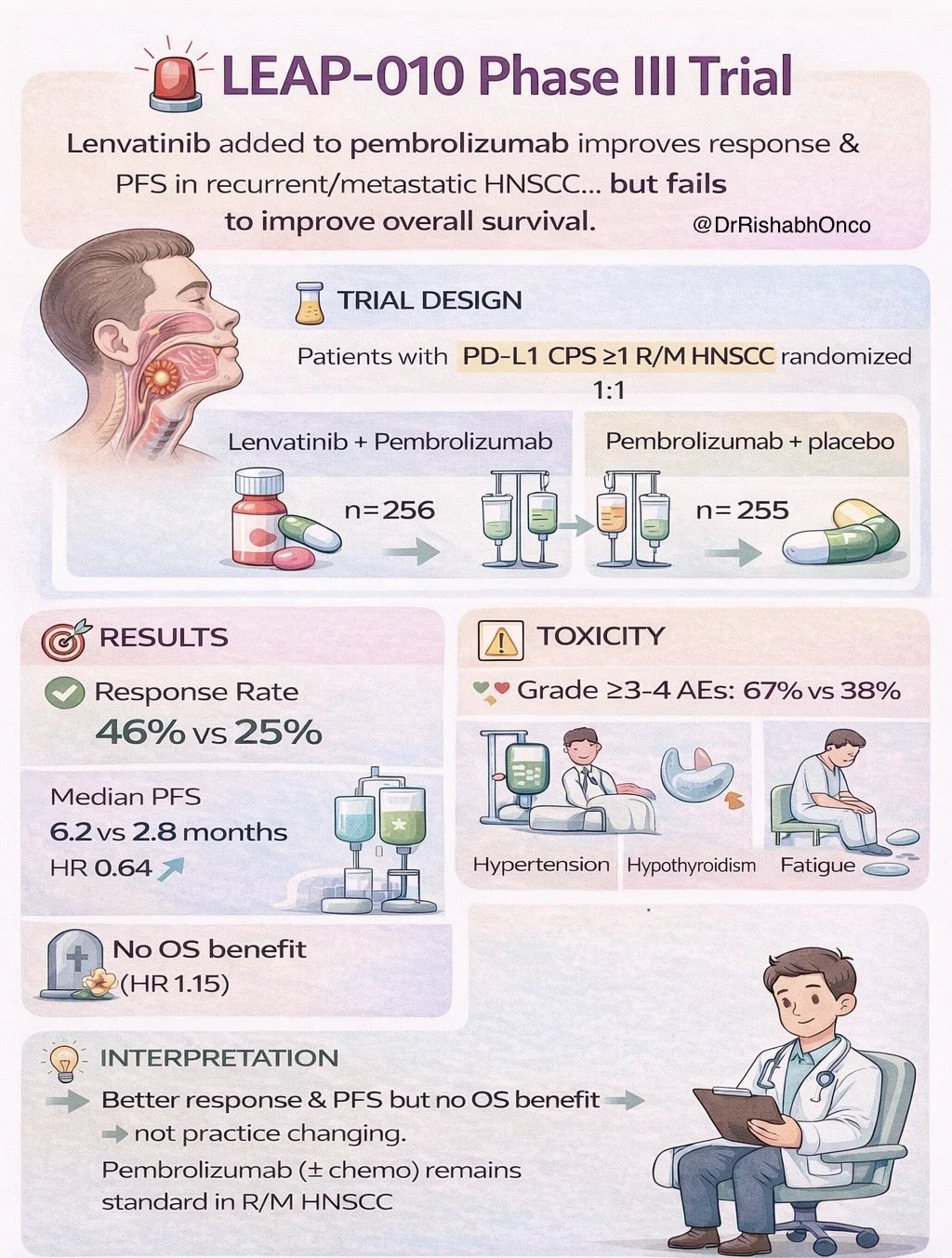
Rishabh Jain (Medical Oncologist at AIIMS):
“LEAP-010 Phase III Trial
Adding lenvatinib to pembrolizumab improves response and PFS in recurrent/metastatic HNSCC… but fails to improve overall survival. Here’s what the new JCO Phase III trial shows
Trial design
Patients with PD-L1 CPS ≥1 R/M HNSCC randomized 1:1
- Lenvatinib + Pembrolizumab
- Pembrolizumab + placebo
Primary endpoints
- ORR • PFS • OS
Key results
Response rate: 46.1% vs 25.4%
Median PFS: 6.2 vs 2.8 months
• HR 0.64
Overall survival
• 15.0 vs 17.9 months
• HR 1.15
Toxicity Grade 3–4 AEs: 66.9% vs 38.3%
Takeaway: Despite improved ORR and PFS, the combination did not improve OS. Pembrolizumab (± chemotherapy) remains the first-line standard in R/M HNSCC. Save this if you follow head & neck oncology trials
Full paper in comment ”
Enrique Grande (Director of the Medical Oncology Department at Quirónsalud, and Adjunct Professor at The University of Texas MD Anderson Cancer Center):
“Very honored and grateful to have had the opportunity to discuss at EAU26 the evolving role of immune checkpoint inhibitors combined with BCG in patients with non–muscle-invasive bladder cancer.
The emerging data from the POTOMAC, CREST, and ALBAN trials are helping us better understand how immunotherapy may complement intravesical therapy and potentially reshape the management of high-risk disease.
What truly stood out during the discussion is that progress in bladder cancer does not happen in isolation. It is the result of multidisciplinary collaboration — urologists, medical oncologists, radiation oncologists, pathologists, radiologists, and research teams working together with a shared commitment to improving patient outcomes.
This collaborative approach is exactly how we care for patients across quironsalud through One Oncology Madrid: bringing expertise together, integrating innovation, and ensuring that every patient benefits from the collective knowledge of a dedicated oncology network.
Grateful to be part of such an inspiring scientific community pushing the field forward.”

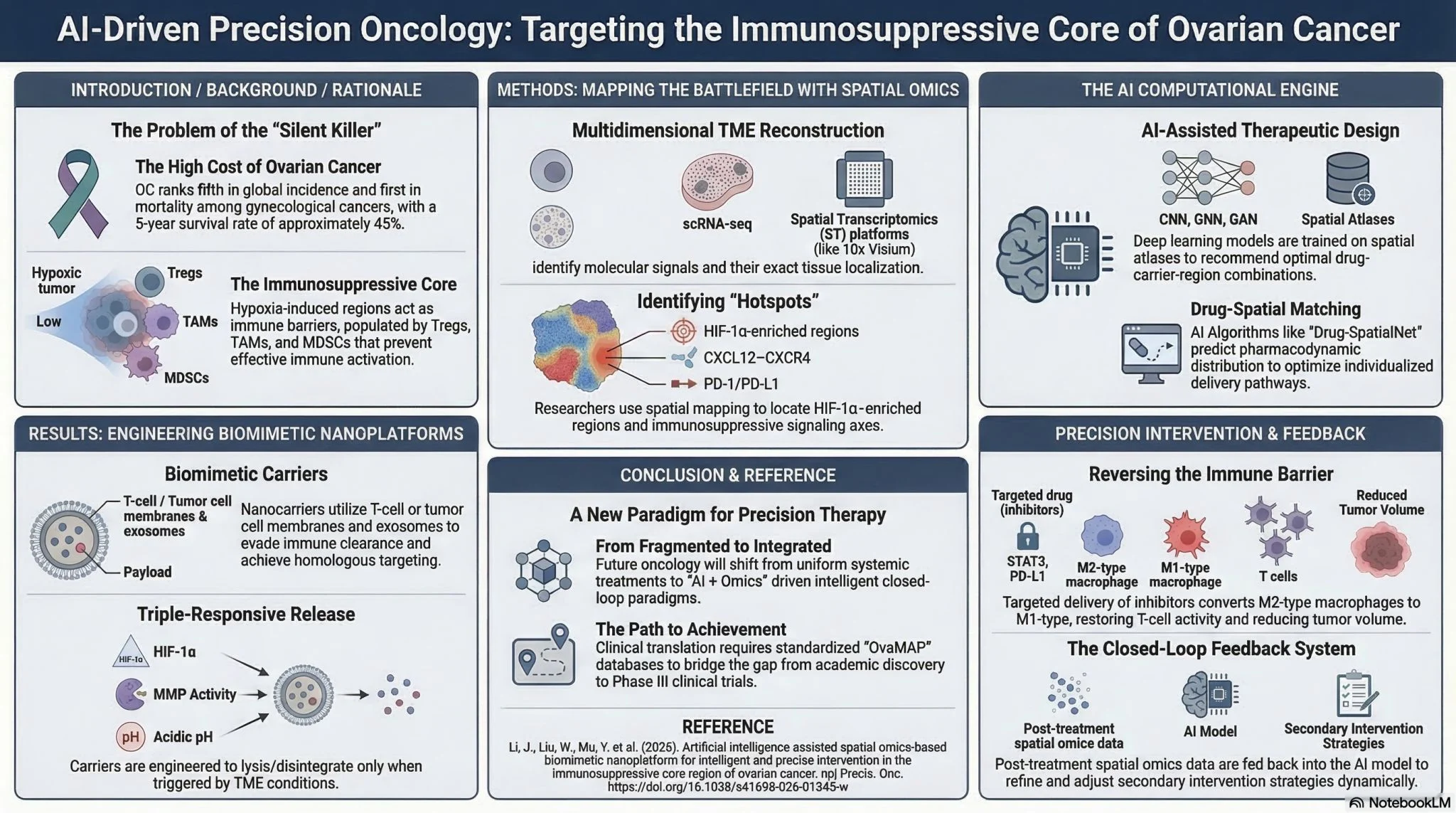
Laura Venegas (Medical Oncologist and Gynecologic Oncology and Drug Development Fellow with MSc in Translational Medicine at University Health Network):
” From Bench to Bedside B2BGynOnc:
This week’s papers highlight how translational medicine is rapidly bridging molecular discoveries and clinical decision-making in gynecologic oncology.
From mechanisms of drug resistance to AI-driven predictions and large randomized trials, the pipeline from laboratory insight to patient care continues to accelerate.
Here are a few signals worth watching:
- Overcoming PARP inhibitor resistance
New translational studies are dissecting the molecular basis of PARP inhibitor resistance. These findings open the door to combination strategies targeting BET and chromatin regulators to re-sensitize tumors. - The dual role of the glucocorticoid receptor
A fascinating mechanistic study highlights the context-dependent role of the glucocorticoid receptor in gynecologic tumors—acting as both a tumor suppressor and a driver of therapeutic resistance depending on signaling context. - AI and machine learning entering the clinic
Several groups report AI/ML models capable of predicting treatment response, survival outcomes, and recurrence risk using radiomics, genomic, and clinical data. - New immunotherapy platforms
Innovative bispecific T-cell engager therapies are showing promise by simultaneously binding tumor antigens and activating T cells, representing a sophisticated approach to immune redirection in solid tumors. - Genomics guiding chemotherapy choices
New analyses suggest that BRCA1 and BRCA2 mutations may influence the efficacy of paclitaxel monotherapy, adding another layer to how genomic context can guide chemotherapy selection. - Clinical decision-making in early disease
A new study explores whether adjuvant radiation or chemotherapy improves outcomes in low-risk endometrial cancer patients with isolated tumor cells (ITCs).
Trials in cervical cancer
• A Phase III randomized trial compares two cisplatin dosing schedules with concurrent radiation for locally advanced cervical cancer, addressing a long-standing question about optimal radiosensitization.
• The ongoing RO-PARA trial is evaluating whether prophylactic para-aortic node irradiation improves outcomes in patients with cervical cancer who already have pelvic lymph node involvement.
Takeaway:Across these studies, we see the full spectrum of translational medicine—from molecular mechanisms and epigenetics to AI-driven prediction models and large randomized clinical trials. Together, they illustrate how discoveries at the bench are increasingly shaping decisions at the bedside.”
Fabio Piccoli (Dirigente Medico in Radioterapia Oncologica at ASST Papa Giovanni XXIII):
” 𝐏r𝐢o𝐫i𝐭i𝐞s f𝐨r l𝐨c𝐚l i𝐦m𝐮n𝐨t𝐡e𝐫a𝐩y r𝐞s𝐞a𝐫c𝐡 𝐚n𝐝 𝐝r𝐮g d𝐞v𝐞l𝐨p𝐦e𝐧t
Isaacs JM, Luke J. Priorities for local immunotherapy research and drug development. Journal for ImmunoTherapy of Cancer 2026;14:e014222.
Priorities for Local Immunotherapy Research and Drug Development” analyzes the current status and future prospects of local immunotherapy, defined as the use of therapeutic agents administered directly into the tumor microenvironment to trigger an “in situ” immune response that can generate systemic antitumor immunity.
Advantages of the local approach: Intratumoral therapies allow for high drug concentrations to be achieved in the tumor while minimizing systemic toxicity. They are particularly useful for tumors that resist immune checkpoint inhibitors due to a “cold” (free of immune infiltration) or immunosuppressive environment.
Research Phases:
- Early Phase Trials: These are essential for determining the optimal dosage (which is not always the maximum tolerated dose) and measuring biological activity through biopsies of both injected and non-injected lesions.
- Clinical Validation: Efficacy is assessed through endpoints such as progression-free survival (PFS) or overall survival. (OS). New radiographic assessment criteria, such as itRECIST, are being developed to capture composite responses across all lesions and avoid biases due to pseudoprogression.
- Application contexts: Local therapies may be particularly effective in the neoadjuvant setting (before surgery), where tumor burden is lower and immune activation can prevent systemic recurrence.
Therapy classes in development:
- Oncolytic viruses (e.g., TVEC, RP1);
- Toll-like receptor (TLR) and STING pathway agonists;
- Low-dose radiotherapy to induce immunogenic cell death;
- Nucleic acid (mRNA) and cytokine therapies modified to remain confined to the tumor.
In conclusion, although there are already approved therapies (such as TVEC for melanoma or BCG for bladder cancer), the future of the field will depend on the ability to integrate these agents into combination regimens and use precise biomarkers to select patients.”

Emmanuel Amartya (Research Assistant at Edison Biotechnology Institute):
“Exciting news in prostate cancer treatment: a “cloaked” immunotherapy is showing real promise.
Prostate cancer is the most common cancer among men in many countries, including the US and UK. About 1.5 million men are diagnosed worldwide each year.
A phase I trial of VIR-5500 has shown remarkable promise for advanced prostate cancer patients. What makes this T-cell engager different? It uses a “cloaking device” that keeps the drug inactive until it reaches the tumor.
Here’s why this matters:
Traditional T-cell engagers activate the immune system throughout the entire body, causing severe inflammatory responses. This new approach solves that problem elegantly—88% of patients experienced minimal side effects.
The early results are encouraging:
• 82% of evaluable patients saw their PSA levels halved at the highest dose
• 45% experienced tumor shrinkage across multiple body sites
• The drug brings T-cells directly to cancer cells marked with PSMA, facilitating a targeted immune attack
The cloaking mechanism also extends the drug’s time in the bloodstream, potentially reducing dosing frequency for patients.
With over 75% of the 10,000 men diagnosed annually with advanced prostate cancer having PSMA-positive tumors, this could represent a significant new treatment option for a large patient population.
What excites me most is how this addresses a longstanding challenge—making immunotherapy work safely and effectively for prostate cancer, where benefits have historically remained limited compared to other cancer types.
The trial continues as VIR-5500 moves into larger studies. Hopeful that this approach brings us closer to making advanced prostate cancer curable.


