Hodgkin lymphoma (HL) is a B-cell-derived hematologic malignancy that presents a unique paradox: it is defined less by its malignant cells than by the immune microenvironment it subverts, making it both biologically intriguing and highly amenable to therapeutic targeting.
This article unpacks the core biology of Hodgkin lymphoma, bridges it to clinical practice, and – most importantly – outlines the future directions shaping patient care.
Risk Factors and Epidemiologic Patterns
There is no clearly defined etiology for HL, but there are risk factors that may predispose individuals to develop the malignancy:
- Epstein-Barr virus positivity – associated with approximately 1% of all cancers and one-third of all HL cases. Distinct mechanisms include loss of immune surveillance and provision of alternative survival signaling pathways, particularly in cases lacking extensive genetic lesions.
- Immunosuppression secondary to a solid organ or hematopoietic cell transplantation, immunosuppressive drugs, and HIV.
- Familial predisposition – studies found that there is a ten-fold increase in developing HL in same-sex siblings of patients with HL.
Every year, HL accounts for approximately 10% of newly diagnosed lymphoma cases (2.6 cases per 100,000) in the United States, or 2.5% of total cancer diagnoses. It is distinguished by a bimodal age distribution, with incidence peaks in adolescents and young adults (15-35 years) and again in older adults (>55 years). It affects males more than females, especially in the pediatric population, where 85% of cases occur in boys (Munir F et al., 2023, Kaseb H et al., 2023).
The 1% Tumor Paradox
The pathogenesis of HL can be conceptualized as a multistep process:
1. Emergence of a crippled GC B cell with nonfunctional BCR
2. Epigenetic reprogramming and loss of B-cell identity
3. Rescue from apoptosis via NF-κB/JAK-STAT activation
4. Recruitment and reprogramming of a supportive microenvironment
5. Establishment of immune evasion through checkpoint activation and antigen presentation defects
The disease is characterized by unique, mononucleated Hodgkin cells, and large, multinucleated Reed-Sternberg cells (HRS cells). Genetic analyses confirm that this cells are transformed B cells, evidenced by clonally rearranged immunoglobulin heavy- and light-chain variable (IgV) genes with somatic hypermutation, including crippling mutations that indicate origin from germinal center B cells that escaped apoptosis.
A hallmark feature is the loss of the canonical B-cell transcriptional program, characterized by downregulation of key factors such as OCT2, BOB.1, and PU.1, alongside acquisition of a distinctive immunophenotype (classically CD30⁺, CD15⁺, with weak or absent CD20), reflecting profound epigenetic reprogramming and lineage infidelity.
Constitutive activation of the NF-κB signaling pathway is a defining molecular event in HL that drives transcription of anti-apoptotic genes (BCL-XL, c-FLIP), cytokines, and chemokines. This arises through multiple, often redundant mechanisms:
- genetic alterations in negative regulators (TNFAIP3/A20 loss)
- amplifications or mutations in REL and other pathway components
- upstream activation via CD30 and CD40
Aberrant activation of the JAK-STAT pathway is another critical axis. Mechanisms include:
- Copy number gains at chromosome 9p24.1
- Autocrine/paracrine cytokine loops (IL-13, IL-6)
Genetic amplification of 9p24.1 also upregulates PD-L1/PD-L2 on HRS cells, driving PD-1-mediated T-cell exhaustion and immune tolerance. In parallel, HRS cells frequently downregulate MHC class I and II expression, impairing antigen presentation and further promoting immune escape.
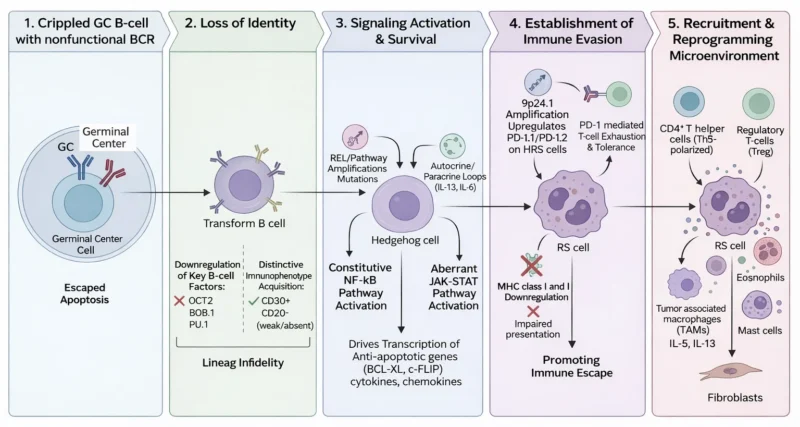
Unlike most malignancies, HRS cells constitute <1% of the tumor mass, with the remainder composed of:
- CD4⁺ T helper cells (often Th2-polarized)
- Regulatory T cells (Tregs)
- Tumor-associated macrophages (TAMs)
- Eosinophils, mast cells, and fibroblasts
HRS cells actively orchestrate this microenvironment through secretion of CCL5, CCL17/TARC, IL-5, IL-13, which recruit immune cells, polarize them toward tumor-supportive phenotypes and inhibit effective cytotoxic responses. Macrophages and Tregs contribute to immune suppression, while fibroblasts participate in extracellular matrix remodeling and fibrosis (Weniger M et al., 2021, Munir F et al., 2023).
HL Under the Microscope
The WHO classifies Hodgkin lymphoma into two main categories – classical Hodgkin lymphoma (cHL) and nodular lymphocyte-predominant Hodgkin lymphoma (NLPHL) – based on morphological and immunohistochemical features.
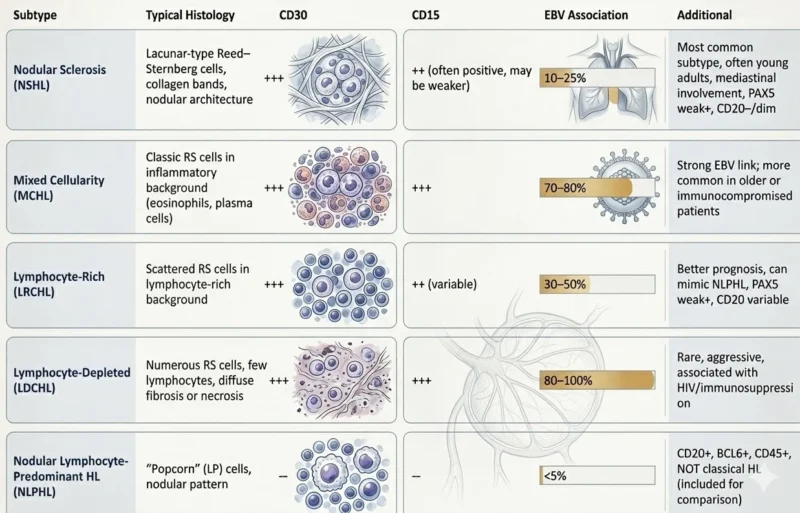
cHL accounts for 90-95% of Hodgkin lymphoma cases and typically presents as an aggressive disease. Its defining feature is the presence of HRS cells, which are characteristically CD30⁺ (100%), CD15⁺ (85%), CD45⁻ and PAX5⁺. cHL is subdivided into four histologic subtypes:
1. Nodular sclerosis cHL – the most common subtype (70%), characterized by lacunar-type HRS cells and fibrotic (sclerosing) bands, often presenting with mediastinal involvement and bulky disease, prognosis is generally favorable.
2. Mixed cellularity cHL – more frequent in developing regions and associated with HIV and Epstein-Barr virus, shows a diffuse inflammatory background without sclerosis and carries a relatively poorer prognosis.
3. Lymphocyte-rich cHL – comprising 5% of cases, this subtype may have a nodular or diffuse cellular background of small lymphocytes, without neutrophils or eosinophils, presents early, lacks bulky disease, and is associated with an excellent prognosis.
4. Lymphocyte-depleted cHL – a rare (1%) and aggressive form marked by abundant HRS cells and minimal inflammatory background, with poor clinical outcomes.
NLPHL is characterized by a nodular architecture with LP (“popcorn”) cells in a background of small B lymphocytes, follicular dendritic cells, and follicular T lymphocytes. Representing 5-10% of Hodgkin lymphoma cases, it is a distinct entity with features overlapping B-cell non-Hodgkin lymphomas. The malignant cells are CD20⁺ and lack CD30 expression, in contrast to HRS cells (Mamotov J et al., 2021, Kaseb H et al., 2023).
Clinical Spectrum of HL
HL most commonly presents with painless lymphadenopathy, observed in 80% of pediatric cases, typically involving the cervical, supraclavicular, or axillary regions. Affected nodes are firm and rubbery. Mediastinal disease is also prominent, particularly in older children and adolescents, with up to 75% presenting with a mediastinal mass. This may manifest clinically as dyspnea, orthopnea, dysphagia, or chest discomfort, and in severe cases can lead to respiratory compromise or superior vena cava syndrome.
Systemic “B symptoms”- fever, drenching night sweats and unintentional weight loss (≥10% over six months) – are more frequent in advanced stages (III-IV), in mixed cellularity or lymphocyte-depleted subtypes. A high ESR is a non-specific marker of inflammation that often accompanies these conditions. Increased susceptibility to infections may also occur.
Additional features include fatigue, anorexia, pruritus, and the classic but uncommon alcohol-induced lymph node pain (possibly mediated by vasodilation or cytokine-driven neural sensitization within the tumor microenvironment). Extranodal involvement is less common but may produce site-specific manifestations when present.
NLPHL predominantly affects males and presents with localized, painless peripheral lymphadenopathy – most commonly in the cervical region – while typically sparing the mediastinum. It follows an indolent course but is notable for a tendency toward late relapses.
Distinguishing HL from Its Mimics
Definitive diagnosis of HL requires histopathologic evaluation of an involved lymph node or affected tissue, with excisional biopsy preferred, as fine-needle aspiration or core biopsy often yields non-specific results due to the low proportion of malignant cells and loss of nodal architecture.
Differential diagnosis of HL includes both reactive and malignant conditions. Key considerations include Infectious mononucleosis (can mimic HL clinically and histologically), Peripheral T-cell lymphoma and Anaplastic large cell lymphoma (may resemble HL due to overlapping immunophenotypic features such as CD30 expression), Diffuse large B-cell lymphoma, which can present with similar clinical and radiologic findings but differs in morphology and immunohistochemistry.
In addition, inflammatory and autoimmune conditions should also be considered, including Systemic lupus erythematosus, Rheumatoid arthritis, Sarcoidosis, and Hemophagocytic lymphohistiocytosis, all of which may present with lymphadenopathy and systemic features that can mimic HL.
Risk-Adapted Treatment Strategies
Following confirmation, comprehensive staging is performed using imaging modalities such as chest radiography, cross-sectional CT imaging and FDG-PET/CT, which has become the standard for both staging and response assessment. Additionally, metabolic tumor burden and ctDNA, are being actively explored for prognostic capability.
The modified Ann Arbor staging system is the most common and is based on the location of lymphadenopathy, the number and size of the lymph node, and whether the extranodal lymph node involvement is shown systemic. The Lugano Classification modernized the Ann Arbor/Cotswolds system by incorporating PET/CT into initial staging, removing the “X” bulky disease suffix while retaining “A” and “B” designations for systemic symptoms.
Clinical disease risk stratification is performed based on disease stage, bulk status, and presence or absence of B symptoms. In adult Hodgkin lymphoma, there are 3 subgroups:
- Early-stage favorable
- Early-stage/ limited-stage unfavorable (bulky mediastinum, elevated ESR, multiple nodal sites)
- Advanced-stage disease (stage III–IV), often further assessed using the IPS
The 5-year OS in stage I or IIa is approximately 90%, on the other hand, stage IV disease has a 5-year OS of approximately 60%.
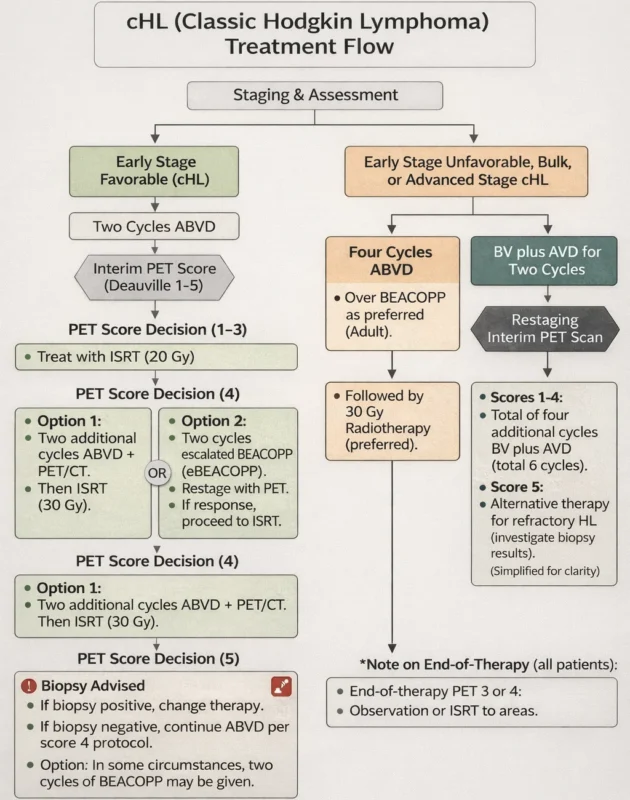
A multicenter phase II trial evaluated the efficacy and safety of BV-AVD versus ABVD followed by 30 Gy of radiotherapy. In the BV-AVD arm, 82.3% achieved PET negativity, compared with 75.4% in the ABVD arm. Among patients with high total metabolic tumor volume, 2-year PFS was 90.9% with BV-AVD versus 70.7% with ABVD, indicating that the addition of brentuximab vedotin improves both response rates and survival outcomes. (Munir F et al., 2023, Zhang S et al.,2023, Evens AM, 2025).
Early-stage NLPHL is often managed with radiotherapy, while advanced cases are treated with anti-CD20-based therapy (such as Rituximab, either alone or in combination with chemotherapy). Relapse is typically manageable, although transformation to DLBCL necessitates more intensive treatment.
Can PET- Guided Therapy Be Truly Global?
While PET/CT-guided decision-making has become central to modern HL management – it raises important questions regarding its real-world applicability across diverse healthcare settings. The global distribution of PET/CT technology remains profoundly unequal: high-income countries have approximately 3.5 scanners per million inhabitants, compared to as little as 0.004 per million in low-income settings, representing a nearly 1000-fold disparity.
Even when available, access is constrained by infrastructure, workforce, and geographic limitations, with one PET/CT scanner serving roughly 600,000 individuals in high-income regions versus up to 10 million in lower-resource countries. Cost further compounds this inequity. These disparities fundamentally challenge the external validity of PET-adapted treatment paradigms, which are often developed and validated in resource-rich environments, highlighting the need for resource-stratified treatment approaches (EANM, 2021, WHO, 2022).
What Is the Role of Transplantation?
R/R disease develops in approximately 10-25% of patients. The conventional treatment for chemosensitive relapsed cHL remains salvage chemotherapy followed by HDC and ASCT. According to the CIBMTR, outcomes after ASCT in HL have improved markedly over the past two decades, with 3-year survival rising from 72% to 92%. While this paradigm has been the backbone of management for decades, outcomes – particularly in high-risk relapse – remain challenging, and no single gold-standard salvage regimen has emerged.
Newer combinations such as ifosfamide-vinorelbine and gemcitabine-vinorelbine offer RR of 76% with improved tolerability, reflecting a gradual shift toward more balanced efficacy-toxicity profiles. For patients who fail salvage therapy, outcomes with HDC/ASCT are generally poor, necessitating consideration of alternative strategies such as allogeneic transplantation or novel targeted and immunotherapeutic approaches (Munir F et al., 2023).
Is HL at a Therapeutic Inflection Point?
The treatment story in cHL is no longer just about whether PD-1 blockade works. That question has largely been answered. The more interesting question is where checkpoint inhibition belongs in the treatment sequence and how long its benefit persists.
Pembrolizumab remains anchored by the phase 3 KEYNOTE-204 result, which established superior PFS of 13.2 months vs 8.3 months with brentuximab vedotin in R/R cHL and solidified the role of PD-1 blockade as a preferred option after relapse in many patients. Long-term activity from KEYNOTE-087 has reinforced that the responses are not merely transient, with an ORR of 69% (CR 22%) and MDR approaching 17 months, with durable antitumor activity maintained on extended follow-up (Kuruvilla J et al., 2021, Armand P et al., 2024).
At ASH 2025, a phase 2 PET-adapted de-escalation regimen using brentuximab vedotin, pembrolizumab, doxorubicin, and dacarbazine reported that all 25 treated patients achieved a PET response sufficient to stop chemotherapy after cycle 3, with an 88% complete remission rate at end of treatment and no reported progression at a median follow-up of 11.2 months. These data are early and conference-level, but they are conceptually important: pembrolizumab is increasingly being studied not only as salvage therapy, but as a tool to reduce cumulative chemo exposure in a predominantly young patient population (Lee H et al., 2025).
For nivolumab, the long tail of benefit remains visible in CheckMate 205. 5-year follow-up confirmed durable efficacy and overall survival in R/R cHL after autologous transplant failure, with no major new safety signal and with the intriguing observation that some patients who stopped therapy in persistent complete remission could respond again when retreated at relapse (Stephen M et al., 2023).
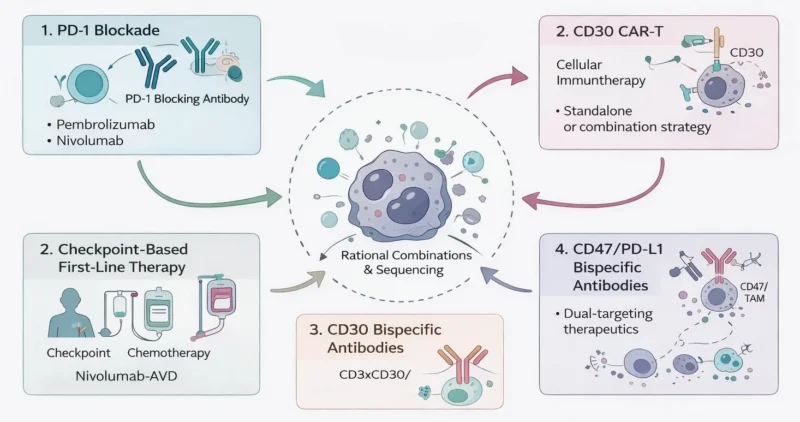
The bigger practice-shaping story, the 3-year update of SWOG S1826, showed that nivolumab-AVD maintained a PFS advantage over BV-AVD, with approximately 91% 3-year PFS vs 82% for the BV arm, and benefit observed across multiple subgroups, including adolescents and older adults. No major new safety concern emerged in the update. This reinforces the evolving view that PD-1-based therapy is transitioning from a salvage option to a foundational component of frontline treatment (Herrera A et al., 2025).
CD30-directed bispecific antibodies (CD3- and CD16-engaging) are emerging as promising “bridge” between checkpoint inhibitors and cellular therapy, aiming to overcome microenvironment-driven resistance. At the EHA 2023 Congress, Yu et al. reported early results of the CD47/PD-L1 bispecific antibody IBI322 in patients with cHL resistant to prior anti-PD-1 or PD-L1 therapy. In this heavily pretreated population, IBI322 demonstrated an ORR of 47.8% and a disease control rate of 91.3%, indicating encouraging antitumor activity alongside a manageable safety profile.
CD30 CAR-T: Still Investigational, But No Longer Hypothetical
The most compelling “next-step” story is CD30 CAR-T. A 2025 long-term update of the CHARIOT reported that autologous CD30 CAR-T was well tolerated, with no treatment-related deaths, no dose-limiting toxicities, and no high-grade CRS or neurotoxicity. In that update, with median follow-up around 45 months, the median PFS was 7.1 months, the 36-month PFS was 17%, and the 36-month OS was 91%. That profile suggests that single-agent CD30 CAR-T can induce meaningful responses with good tolerability, but durability remains a key hurdle. A 2025 ASH abstract on CD30 CAR-T + nivolumab in R/R cHL after frontline failure reported particularly strong activity, with ORR 92% and CR rate 77% (Ahmed S et al., 2025).
Combination data with CD30 CAR-T plus camrelizumab demonstrated a favorable safety profile alongside clinically meaningful activity. In the overall efficacy-evaluable population, the ORR was 63.6% with a CR rate of 36.3%. Notably, outcomes were more pronounced in the cHL subset, where the ORR reached 100% and the CR rate 57.1%, with 2-year PFS and OS of 57.1% and 100%, respectively. Although based on small numbers, these findings reinforce the direction: CD30 CAR-T may be most effective not as a standalone strategy, but as part of a rational immunotherapy sequence or combination (Yu M et al., 2026)
In Hodgkin lymphoma, success now depends less on eliciting responses and more on strategically sequencing therapies to make them durable.
You can also read Immunotherapy for Lymphoma: Types, Success Rate, Side Effects and More on OncoDaily.
Written by Susanna Mikayelyan, MD
FAQ
What makes Hodgkin lymphoma different from other cancers?
Hodgkin lymphoma is unique because the malignant cells make up less than 1% of the tumor mass, while the majority consists of immune cells that are actively manipulated by the tumor microenvironment.
What are the most common early signs of Hodgkin lymphoma?
The most common presentation is painless lymphadenopathy, often in the cervical region, along with possible systemic “B symptoms” such as fever, night sweats, and weight loss.
Why do Hodgkin lymphoma lymph nodes sometimes hurt after drinking alcohol?
Although rare, alcohol-induced lymph node pain is a classic but poorly understood feature of Hodgkin lymphoma. It is thought to result from alcohol-triggered vasodilation or cytokine-mediated nerve sensitization within affected lymph nodes.
If the immune system recognizes the tumor, why doesn’t it destroy it?
In Hodgkin lymphoma, the immune system is present - but functionally suppressed. Malignant cells exploit immune checkpoints (such as PD-1 signaling) and reprogram surrounding immune cells, effectively turning off the body’s natural anti-tumor response.
What role does PET/CT play in Hodgkin lymphoma management?
PET/CT is central for staging and treatment response assessment, allowing clinicians to adapt therapy based on metabolic response, although access remains unequal globally.
What is the difference between classical Hodgkin lymphoma and NLPHL?
Classical Hodgkin lymphoma is characterized by CD30⁺ Reed–Sternberg cells and an inflammatory background, while nodular lymphocyte-predominant Hodgkin lymphoma (NLPHL) features CD20⁺ LP (“popcorn”) cells and behaves more like an indolent B-cell lymphoma.
How are immunotherapies changing Hodgkin lymphoma treatment?
Checkpoint inhibitors such as pembrolizumab and nivolumab have significantly improved outcomes in relapsed disease and are increasingly being integrated into earlier lines of therapy.
What are the future directions in Hodgkin lymphoma treatment?
Emerging approaches include CAR-T cell therapy, bispecific antibodies, and combination immunotherapy strategies aimed at improving durability of response and reducing treatment toxicity.