Clear cell sarcoma (CCS) is an ultra-rare and aggressive sarcoma with very limited sensitivity to conventional systemic therapy. Most cases are driven by EWSR1 rearrangements, usually EWSR1::ATF1, and outcomes in advanced disease have historically been poor. Against this background, the IMMUNOSARC II phase II trial evaluated whether combining the antiangiogenic TKI sunitinib with the PD-1 inhibitor nivolumab could offer more meaningful disease control.
FDA Fast Track for CLD-201: Stem Cell–Based Oncolytic Virus for Soft Tissue Sarcoma
Study overview
Patients aged 12–80 years with advanced, progressive, measurable CCS were enrolled after central pathology review, with molecular confirmation of EWSR1 rearrangement required. Treatment consisted of:
Sunitinib 37.5 mg daily for the first 2 weeks, then 25 mg daily
Nivolumab 240 mg every 2 weeks, starting on day 15
The primary endpoint was the 6-month progression-free survival rate.
Patient population
A total of 26 patients received treatment, and 23 were evaluable for the primary endpoint. This was a clinically aggressive population:
- median age: 42 years
- 92.3% had metastatic disease at baseline
- median metastasis-free interval: 4.1 months
The most common fusion identified was EWSR1::ATF1.
Efficacy results
The trial met its primary endpoint and showed a clinically meaningful signal of activity.
Key efficacy findings included:
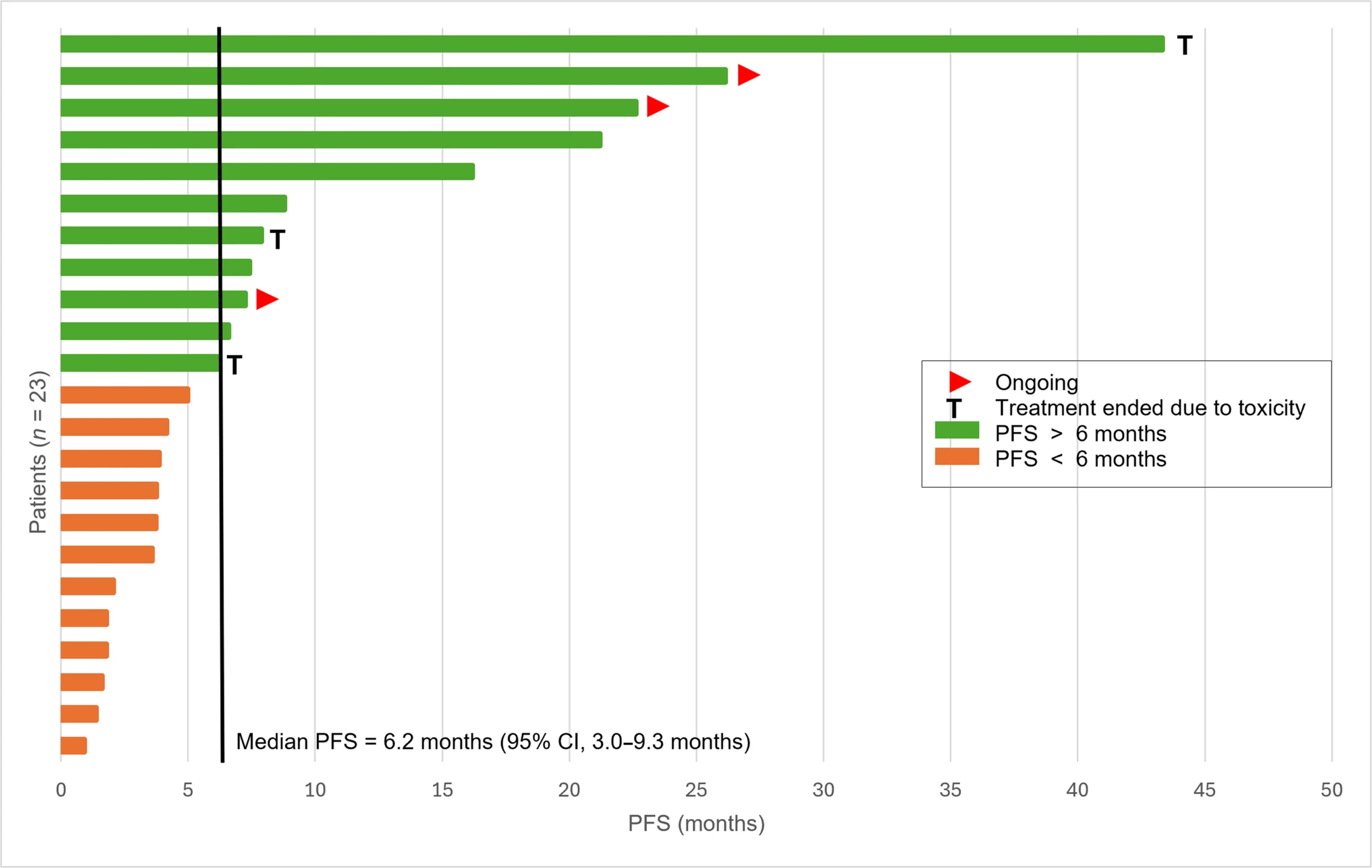
- 6-month PFS rate: 50.1%
- Median PFS: 6.2 months
- Median OS: 17.0 months
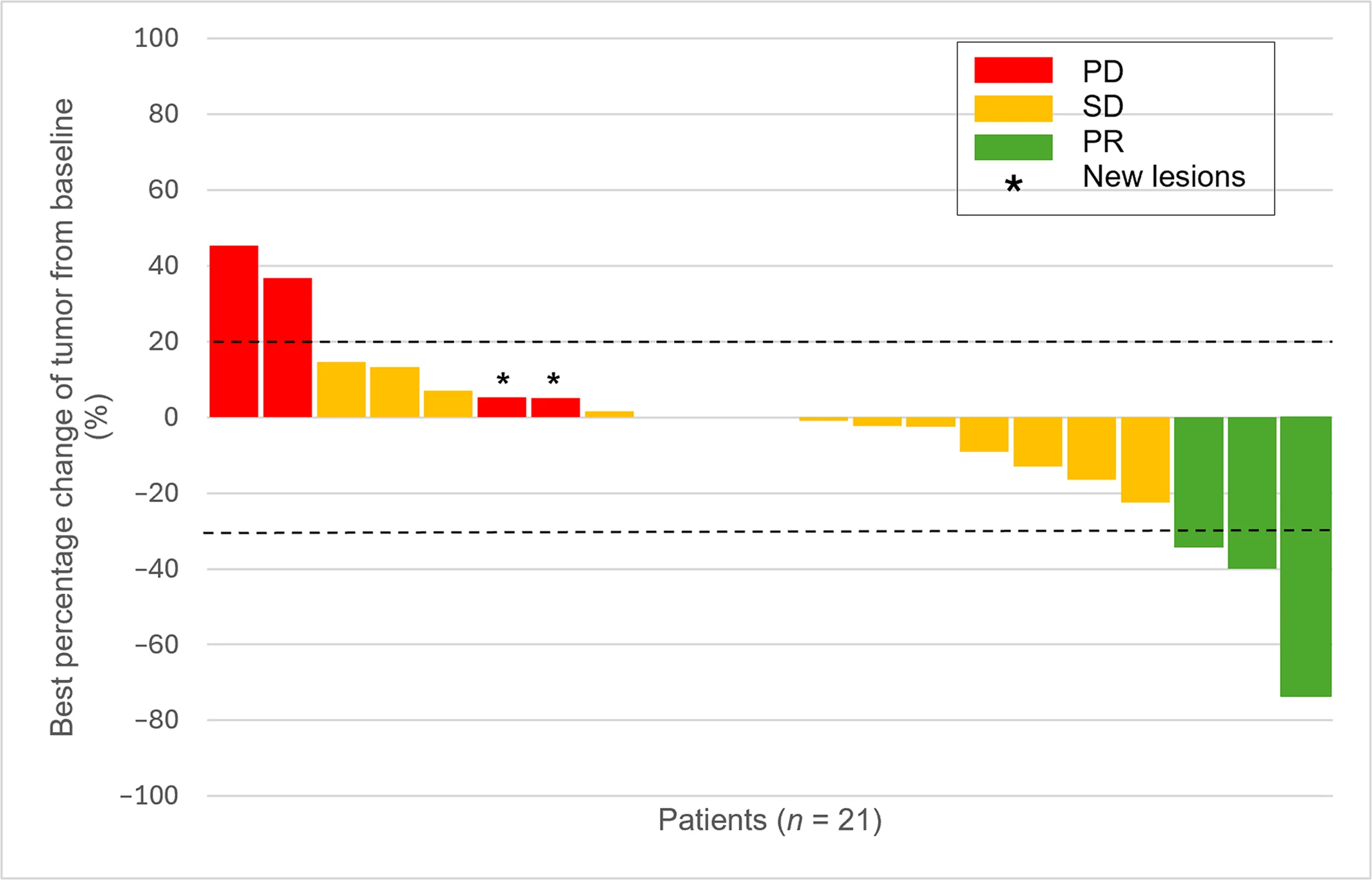
- Objective response rate: 14.3%
- Stable disease: 66.7%
Among the 21 patients with at least one radiologic assessment, 3 achieved a partial response, while 14 achieved stable disease. This is particularly relevant in CCS, where objective responses are rare and durable disease stabilization can itself represent meaningful benefit.
Another important point is that some responses appeared durable. The median duration of response was not reached, and several patients experienced prolonged disease control, suggesting that this regimen may be doing more than producing short-lived tumor shrinkage.
Why this matters
These findings compare favorably with previously reported prospective CCS studies, where median PFS has generally remained short and response rates have been low. Although cross-trial comparisons must be made carefully, the combination of sunitinib plus nivolumab appears to offer a stronger signal than earlier MET inhibitor-based approaches.
Biologically, this also makes sense. CCS is a fusion-driven sarcoma with evidence of angiogenic signaling and possible immune vulnerability. Sunitinib may help reverse vascular-driven immune suppression, while nivolumab may restore antitumor T-cell activity. Together, this creates a rational combined strategy in a disease where very few systemic options have shown real promise.
Safety
Toxicity was significant but overall consistent with the known profiles of both agents.
The most frequent all-grade toxicities included:
- lymphocytopenia
- leukopenia
- anemia
- neutropenia
Grade 3 toxicities were not uncommon, particularly anemia and lymphocytopenia, and two grade 4 toxicities were reported. Immune-related adverse events included diarrhea, rash, hypothyroidism, and uveitis. Importantly, no grade 5 toxicity was observed.
So while the regimen is clearly not toxicity-free, the safety profile appears manageable and broadly in line with expectations for this type of combination.
Biomarker signal
One of the most interesting exploratory findings was the association between higher PD-L1 composite score and improved progression-free survival. Patients with higher PD-L1 scores had substantially longer PFS than those with lower scores, and PD-L1 also correlated with response.
This remains exploratory, but in such a rare and biologically homogeneous disease, it raises the possibility that PD-L1 may have biomarker relevance in CCS, even though checkpoint biomarkers have often been disappointing across broader sarcoma populations.
Key takeaway
IMMUNOSARC II suggests that sunitinib plus nivolumab may represent a meaningful therapeutic option in advanced clear cell sarcoma, a disease with a major unmet need and very limited effective systemic therapies.
The study showed:
- meaningful 6-month disease control
- a median PFS of 6.2 months
- a median OS of 17 months
- durable benefit in a subset of patients
For an ultra-rare sarcoma with historically poor outcomes, this is an important signal and supports further exploration of immunotherapy-based strategies in CCS.
You Can Read Full Article Here