The rise of immune checkpoint inhibitors has transformed the treatment landscape across multiple malignancies, including gynecologic cancers. Yet despite this progress, the ability to predict which patients will derive durable benefit remains limited. In everyday practice, PD-L1 expression is still one of the most commonly used biomarkers for PD-1/PD-L1 inhibitor selection, although its predictive value is imperfect and highly disease-dependent.
At the same time, growing evidence suggests that sex-related biology may influence immunotherapy outcomes. Several meta-analyses in melanoma and non-small cell lung cancer have reported that women may derive less benefit than men from immune checkpoint inhibitor monotherapy, whereas outcomes may differ when immunotherapy is combined with chemotherapy. These observations raise an important question: are there female-specific immunologic factors that shape response to checkpoint blockade?
This is the setting in which the FECONDE study was designed.
What is FECONDE?
FECONDE is an observational, multicenter study investigating whether one or more previous pregnancies may influence the efficacy of PD-1/PD-L1 inhibitors in women with gynecologic neoplasms, specifically endometrial and cervical cancer.
The study is based on a compelling biologic premise: pregnancy is not simply a temporary physiologic state, but a profound immunologic event that may leave durable effects on the maternal immune system. These long-term changes could theoretically affect the activity of checkpoint inhibitors years later.
Why pregnancy might matter for immunotherapy
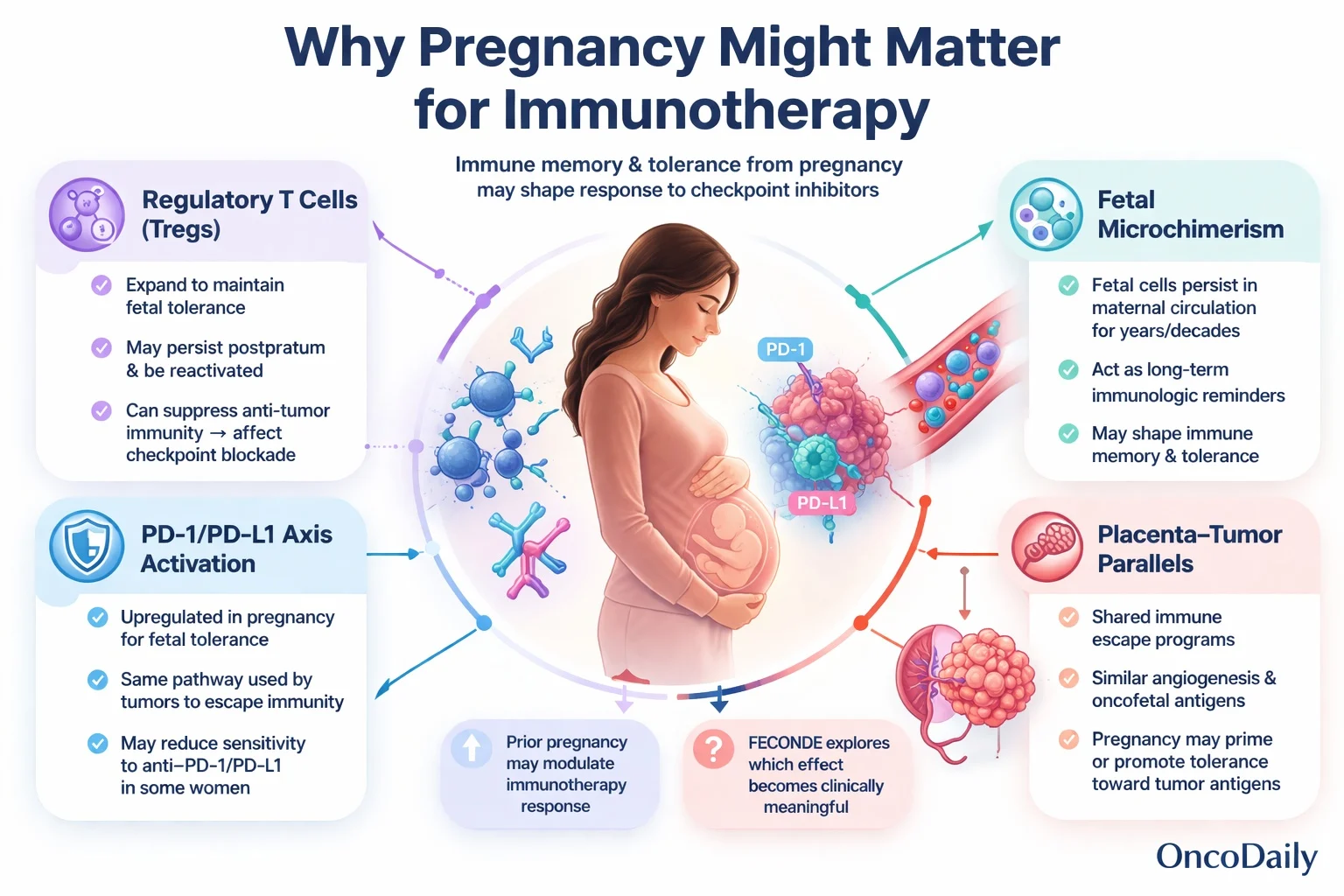
Pregnancy requires the maternal immune system to achieve a highly regulated balance. The fetus carries paternal antigens and is therefore immunologically distinct from the mother, yet successful gestation depends on the establishment of tolerance rather than rejection.
This process involves several mechanisms that are highly relevant to cancer immunology.
One important component is the expansion of regulatory T cells, which help maintain fetal tolerance. These cells do not necessarily disappear after delivery. Studies suggest that fetal-specific Tregs may persist postpartum and can be reactivated with renewed antigen exposure. Because Tregs are also central mediators of tumor immune suppression, persistent pregnancy-related immune tolerance could potentially reduce sensitivity to checkpoint blockade in some women.
Pregnancy is also associated with increased activity of the PD-1/PD-L1 axis, a pathway that plays a critical role both in fetal tolerance and in tumor immune escape. This mechanistic overlap makes it biologically plausible that prior pregnancy history could influence how a patient responds to anti–PD-1 or anti–PD-L1 therapy.
Another major concept is fetal microchimerism. During pregnancy, fetal cells can enter maternal circulation and remain detectable for years, sometimes decades. These cells may serve as long-term immunologic reminders of pregnancy and could influence maternal immune memory, either in a protective or tolerogenic direction depending on context.
The placenta adds another layer of relevance. It shares several biologic and antigenic features with tumors, including immune escape programs, angiogenic signaling, and expression of certain oncofetal antigens. This has led to two competing hypotheses. Prior pregnancy may either prime the immune system against tumor-associated antigens or, conversely, promote long-lasting tolerance toward them. FECONDE is designed to explore whether one of these effects becomes visible clinically.
Study design
FECONDE has a retrospective/prospective observational design and includes women with cervical or endometrial cancer treated with:
- PD-1/PD-L1 inhibitor monotherapy, or
- chemo-immunotherapy combinations
The investigators will collect detailed information across three major domains.
Clinical and oncologic data
This includes tumor type, treatment line, age at treatment start, body metrics, smoking history, comorbidities, performance status, sites and number of metastases, and concomitant medications.
Pathologic and molecular data
The study will also capture variables such as PD-L1 expression, molecular profile, and mutational features of the tumor.
Reproductive and hormonal history
This is the key distinguishing feature of the trial. Investigators will document:
- number of pregnancies
- age at first and last pregnancy
- menarche and menopause history
- use of hormone replacement therapy or contraception
- prior gynecologic surgery
- broader fertility history
This structure will allow the study to examine whether reproductive history has an independent association with oncologic outcomes during checkpoint inhibitor therapy.
Why this is clinically relevant
Immunotherapy is now firmly integrated into the treatment of several gynecologic malignancies.
In cervical cancer, pembrolizumab-based regimens have improved outcomes in PD-L1-positive recurrent or metastatic disease, and cemiplimab has demonstrated benefit after platinum-based therapy. In endometrial cancer, immune checkpoint blockade has become particularly important in dMMR/MSI-H disease, and combinations such as pembrolizumab plus lenvatinib have expanded options for patients with advanced disease beyond platinum.
Still, not all patients benefit. As in other tumor types, there is a clear need for better predictors of benefit and resistance. FECONDE is interesting because it shifts attention from the tumor alone to the host immune history. That is a highly contemporary concept in immuno-oncology. Response to checkpoint blockade is not determined only by PD-L1, mutational burden, or tumor histology. It may also be shaped by long-term features of the patient’s immune system, including those established through pregnancy.
Cervical Cancer: Symptoms, Causes, Stages, Diagnosis and Treatment
Scientific significance
What makes FECONDE especially compelling is that it explores a form of biologic memory that is rarely incorporated into oncology studies. Pregnancy can alter immune composition, T-cell memory, tolerance pathways, and antigen exposure in ways that may persist far beyond the postpartum period. If these changes influence immunotherapy outcomes, reproductive history could become an important contextual variable in future translational and clinical research.
The study may help address several questions:
- Does parity correlate with response, progression-free survival, or overall survival during PD-1/PD-L1 inhibitor therapy?
- Does the number of pregnancies matter?
- Is time since last pregnancy relevant?
- Are the effects different between monotherapy and chemo-immunotherapy?
- Could reproductive history partly explain previously observed sex-based differences in immunotherapy benefit?
Even a negative result would be meaningful, because it would help define which host factors are unlikely to play a major role in checkpoint inhibitor efficacy.
Key takeaway messages
- FECONDE investigates whether prior pregnancy history affects outcomes with PD-1/PD-L1 inhibitors in endometrial and cervical cancer.
- The study is biologically grounded in pregnancy-associated changes involving Tregs, PD-1/PD-L1 signaling, fetal microchimerism, and persistent immune memory.
- It addresses an important gap in immuno-oncology by asking whether a life-course immunologic event can influence modern cancer therapy.
- If positive, the study could open a new line of research into host-derived predictors of immunotherapy response in women.
- If negative, it will still provide valuable evidence on the limits of reproductive history as a clinically meaningful variable in checkpoint inhibitor outcomes.