Pancreatic ductal adenocarcinoma remains one of the most difficult cancers to treat, particularly after progression on standard chemotherapy. For patients with metastatic disease, treatment options after first-line therapy are limited, and response rates with currently available second-line chemotherapy remain low.
A defining biological feature of pancreatic cancer is the high prevalence of RAS alterations. More than 90% of pancreatic ductal adenocarcinomas harbor activating RAS mutations, most commonly involving KRAS codon 12. Although KRAS G12C inhibitors have shown that direct RAS targeting is feasible, KRAS G12C mutations are rare in pancreatic cancer, limiting the applicability of these agents in this disease.
Daraxonrasib, also known as RMC-6236, is an oral RAS(ON) multiselective inhibitor designed to target the active, GTP-bound form of mutant and wild-type RAS proteins, including KRAS, NRAS, and HRAS. This broader mechanism may be particularly relevant in pancreatic cancer, where sustained RAS signaling plays a central role.
The article was published in The New England Journal of Medicine on May 6, 2026.
Title: Daraxonrasib in Previously Treated Advanced RAS-Mutated Pancreatic Cancer
Authors: Brian M. Wolpin, Wungki Park, Ignacio Garrido-Laguna, Alexander Spira, Alexander Starodub, David Sommerhalder, Salman R. Punekar, Minal Barve, Meredith Pelster, Benjamin Herzberg, Nilofer S. Azad, Joel Randolph Hecht, Sai Hong Ignatius Ou, Tong Lin, Sumit Kar, Lin Tao, Rashmi Vora, Aparna Hegde, Kyaw Aung, David S. Hong, and the RMC-6236-001 Investigators.
Daraxonrasib: Mechanism of Action
Daraxonrasib is a small-molecule inhibitor designed to target RAS in its active, GTP-bound state. This mechanism distinguishes it from earlier KRAS G12C inhibitors, which target the inactive GDP-bound form of KRAS and depend on the specific cysteine substitution found in G12C-mutant tumors.
By engaging active RAS-GTP, daraxonrasib blocks RAS effector binding and suppresses downstream signaling, including MAPK pathway activity. This is intended to reduce signaling that supports tumor-cell growth and survival. Preclinical studies showed inhibition of ERK phosphorylation across several KRAS variants, including G12D, G12V, G13D, and Q61X.
This active-state targeting strategy may be especially relevant in tumors with sustained RAS pathway activation and potential resistance to mutation-specific inhibitors.

Read more about Daraxonrasib (RMC-6236) on OncoDaily.
Methods
This was a multicenter, open-label phase 1–2 study evaluating daraxonrasib in patients with advanced solid tumors harboring activating RAS mutations. This report focused on 168 patients with previously treated advanced RAS-mutated pancreatic ductal adenocarcinoma.
Eligible patients had advanced disease with KRAS, NRAS, or HRAS mutations at codons 12, 13, or 61. Patients with pancreatic cancer were required to have disease progression or unacceptable toxicity after fluoropyrimidine- or gemcitabine-based chemotherapy, measurable disease according to RECIST 1.1, and an ECOG performance-status score of 0 or 1.
Daraxonrasib was administered orally once daily in 21-day cycles at doses ranging from 10 mg to 400 mg during dose escalation. Based on safety, exposure, and antitumor activity, 300 mg once daily was selected as the phase 3 dose.
The primary endpoint was safety. Secondary endpoints included pharmacokinetics and antitumor activity, including objective response, duration of response, and progression-free survival. Overall survival was assessed as an exploratory endpoint.
Patient Population
A total of 168 patients with RAS-mutated pancreatic ductal adenocarcinoma received daraxonrasib at doses of 300 mg or less after previous systemic therapy.
The median age was 65 years, and 45% of patients were women. All patients had stage IV disease at study entry. Liver metastases were present in 67% of patients, and lung metastases in 46%.
Most patients had RAS G12 mutations. Among all patients, 89% had RAS G12 alterations, including KRAS G12D in 39%, KRAS G12V in 31%, and KRAS G12R in 17%. Non-G12 mutations included KRAS Q61H, Q61R, Q61K, and G13D.
Patients had received prior systemic therapy, with a median of two previous systemic therapies. Forty-two percent had received one prior line of therapy for metastatic disease, while 58% had received two or more prior lines. Previous treatments included gemcitabine plus nab-paclitaxel and FOLFIRINOX or modified FOLFIRINOX.
Safety
Treatment-related adverse events were common but were mostly grade 1 or 2. Across the 168-patient safety population, treatment-related adverse events of any grade occurred in 96% of patients. Grade 3 or higher treatment-related adverse events occurred in 30%.
The most frequent treatment-related adverse events included rash, diarrhea, nausea, stomatitis or mucositis, vomiting, and fatigue. No grade 5 treatment-related adverse events were reported.
Among patients treated with the 300 mg dose, treatment-related adverse events of any grade occurred in 96%. The most common were rash, stomatitis or mucositis, diarrhea, and nausea. Grade 3 or higher treatment-related adverse events occurred in 34% of patients in this dose group.
Dose modifications were frequent but generally manageable. In the 300 mg group, treatment-related adverse events led to dose interruption in 43% and dose reduction in 30%. Among patients receiving 300 mg, no treatment-related adverse events led to treatment discontinuation.
The authors noted that rash and gastrointestinal adverse events were manageable with routine interventions, including topical or systemic treatments, antidiarrheal therapy, sun protection, and dose modifications.
Efficacy
Daraxonrasib showed antitumor activity in patients with RAS-mutated pancreatic cancer, with descriptive analyses suggesting higher response rates at the 300 mg dose and in earlier-line treatment.
In the subgroup of 26 patients with RAS G12-mutated pancreatic cancer who received daraxonrasib 300 mg as second-line therapy, the objective response rate was 35%. The disease control rate was 92%. The median time to response was 2.6 months, and the median duration of response was 8.2 months.
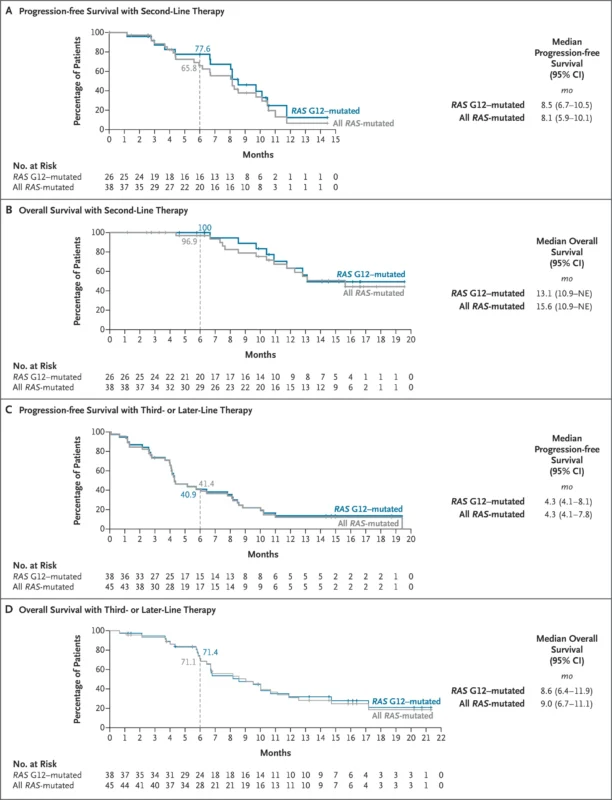
In this RAS G12 second-line subgroup, median progression-free survival was 8.5 months, and median overall survival was 13.1 months after a median follow-up of 17 months.
Among 38 patients with any RAS mutation treated with 300 mg daraxonrasib as second-line therapy, the objective response rate was 29%. Responses were observed across several RAS variants, including KRAS G12D, G12V, G12R, and Q61H. Median progression-free survival was 8.1 months, and median overall survival was 15.6 months.
These outcomes are notable in the context of previously treated metastatic pancreatic cancer, where historical second-line chemotherapy outcomes are generally modest. However, because this was a single-group phase 1–2 study, cross-trial comparisons should be interpreted cautiously.
Conclusion
In this phase 1–2 study, daraxonrasib demonstrated antitumor activity in patients with previously treated advanced RAS-mutated pancreatic ductal adenocarcinoma. The most encouraging activity was observed with 300 mg once daily in the second-line setting, particularly among patients with RAS G12-mutated disease.
The safety profile was characterized mainly by rash and gastrointestinal adverse events. Grade 3 or higher treatment-related adverse events occurred in approximately one third of patients, but treatment discontinuation due to treatment-related toxicity was uncommon, and no grade 5 treatment-related adverse events were reported.
These findings support further evaluation of daraxonrasib in randomized studies. The ongoing RASolute 302 phase 3 trial is evaluating daraxonrasib versus chemotherapy as second-line treatment for metastatic pancreatic cancer.
For a disease in which RAS mutations are highly prevalent and approved targeted options remain limited, daraxonrasib represents a promising investigational approach in the development of direct RAS-targeted therapy for pancreatic cancer.
The full article is available on NEJM.
You can also read about Phase 3 RASolute 302 Trial on OncoDaily.


