For patients with unresectable stage III non-small cell lung cancer (NSCLC), concurrent chemoradiotherapy (CCRT) followed by durvalumab remains the established standard of care after the landmark PACIFIC trial study. However, despite this major advance, more than half of patients still experience progression or death within 18 months, highlighting the persistent unmet need for more effective curative-intent strategies.
The phase III CheckMate 73L trial explored whether earlier and more intensive immunotherapy integration could improve outcomes. Investigators evaluated concurrent nivolumab with chemoradiotherapy followed by consolidation nivolumab with or without ipilimumab, compared with standard durvalumab consolidation after CCRT. The rationale was based on the hypothesis that radiotherapy may enhance tumor antigen presentation and T-cell priming, while PD-1 and CTLA-4 blockade could amplify and sustain anti-tumor immunity.
Study Design and Treatment Strategy
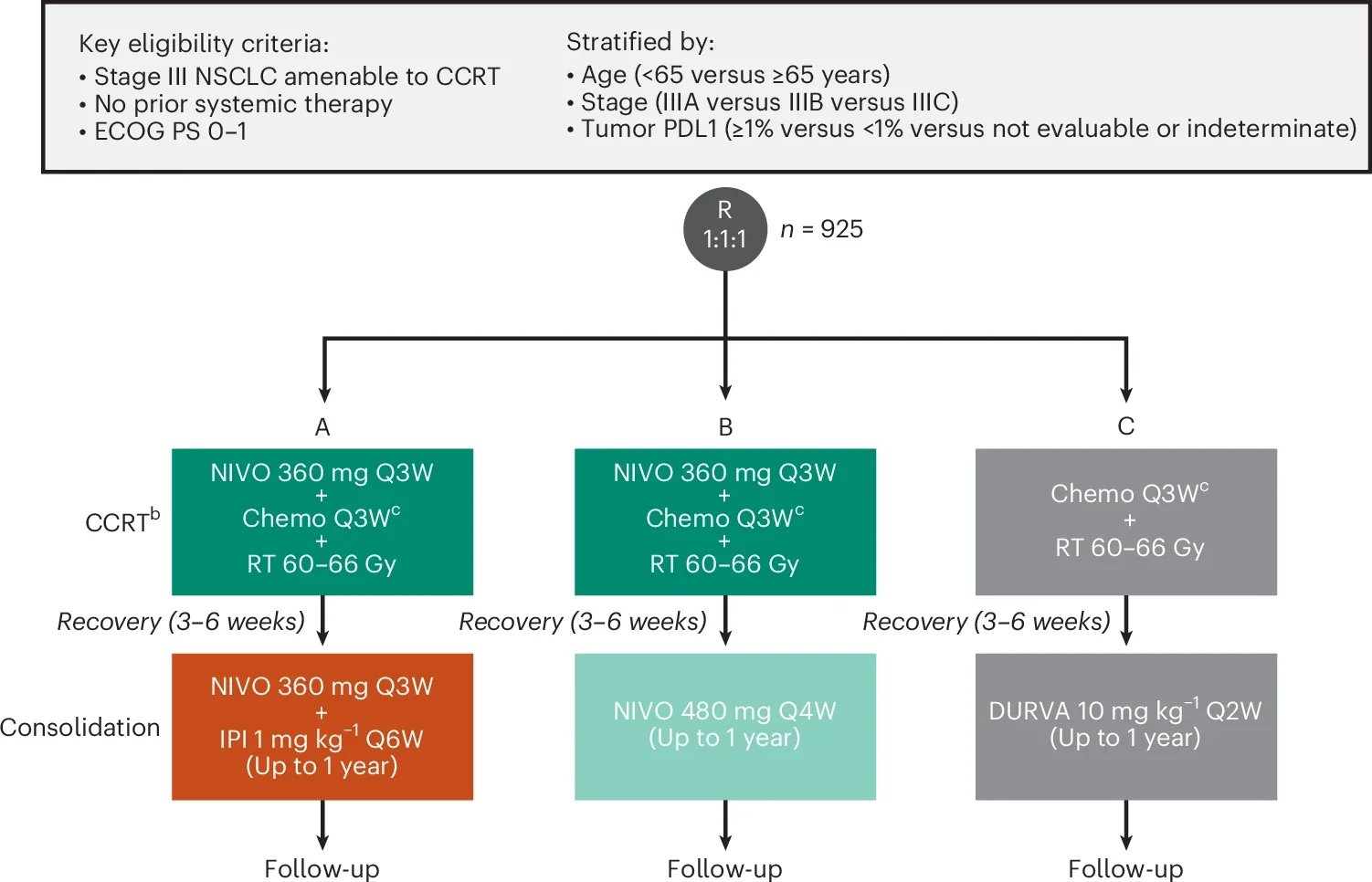
CheckMate 73L was a global randomized phase III trial enrolling 925 patients with previously untreated, unresectable stage III NSCLC across 159 sites in 26 countries. Participants were randomized into three treatment arms:
- Nivolumab + concurrent chemoradiotherapy followed by nivolumab + ipilimumab consolidation
- Nivolumab + concurrent chemoradiotherapy followed by nivolumab consolidation alone
- Standard concurrent chemoradiotherapy followed by durvalumab consolidation
Patients received platinum-doublet chemotherapy with thoracic radiotherapy to 60–66 Gy, followed by up to one year of consolidation immunotherapy. Most patients had stage IIIA or IIIB disease, approximately 58% had squamous histology, and more than 60% had PD-L1 expression ≥1%.
Median follow-up reached 30.5 months.
Key Results
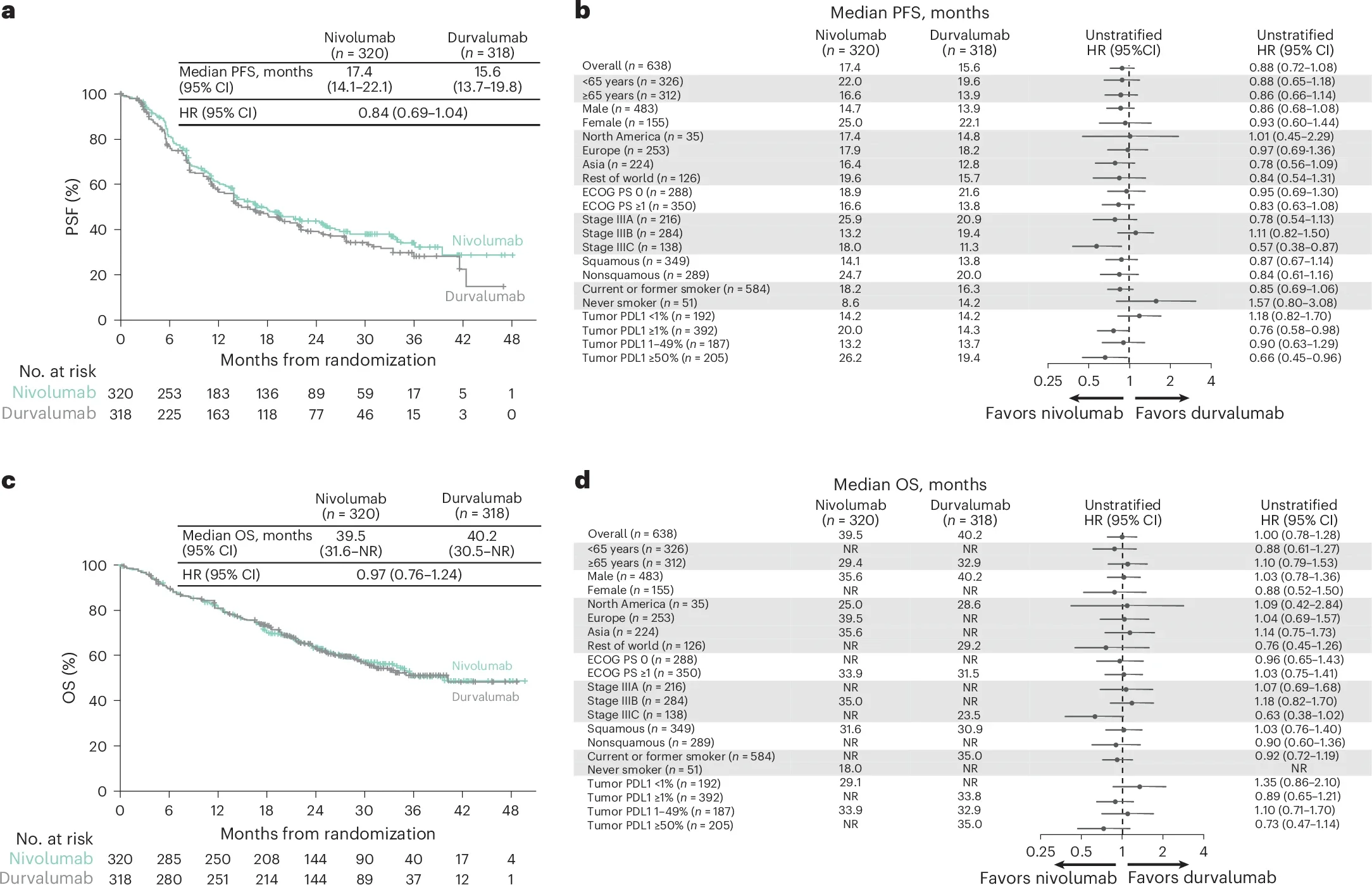
- The primary endpoint was not met. Median progression-free survival was 16.7 months with nivolumab + ipilimumab versus 15.6 months with durvalumab consolidation (HR 0.95; P=0.65).
- Overall survival also did not improve with intensified nivolumab-based strategies. Median OS was 34.6 months in the nivolumab + ipilimumab arm versus 40.2 months in the durvalumab arm (HR 1.12).
- Nivolumab alone combined with CCRT similarly failed to improve survival outcomes compared with durvalumab, with PFS HR 0.84 and OS HR 0.97.
- Objective response rates were numerically similar across treatment groups: 68% for nivolumab + ipilimumab, 72% for nivolumab alone, and 64% for durvalumab. Median duration of response ranged from 25.1 to 31.1 months.
- Exploratory subgroup analysis suggested a numerical PFS improvement in tumors with PD-L1 ≥50%, where median PFS reached 25.5 months with nivolumab + ipilimumab versus 19.4 months with durvalumab, although this was not formally powered for significance.
Safety and Pneumonitis Signal
A major finding of the study was the increased pulmonary toxicity observed with concurrent immunotherapy and chemoradiation. Pneumonitis emerged as the dominant safety concern across nivolumab-containing arms.
Grade 3–4 treatment-related pneumonitis occurred more frequently with nivolumab-based therapy:
- 4% with nivolumab + ipilimumab
- 3% with nivolumab alone
- <1% with durvalumab
Immune-mediated pneumonitis rates were also substantially higher with nivolumab-containing regimens (23% in both nivolumab arms versus 10% with durvalumab).
Importantly, 15 treatment-related deaths across the study were attributed to pneumonitis, emphasizing the clinical challenge of combining concurrent checkpoint blockade with thoracic radiation.
Clinical Interpretation
The CheckMate 73L trial delivers an important negative phase III result in stage III NSCLC immunotherapy. Despite strong biologic rationale and encouraging earlier-phase data, adding concurrent nivolumab during chemoradiation — with or without CTLA-4 blockade, did not improve progression-free or overall survival compared with the established PACIFIC approach.
The findings suggest that simply intensifying checkpoint inhibition during chemoradiotherapy may not overcome resistance mechanisms in locally advanced NSCLC and may substantially increase pulmonary toxicity. The study also raises critical questions regarding the optimal timing of immunotherapy relative to radiation exposure, the immunologic impact of irradiating draining lymph nodes, and whether immunotherapy may be more effective in minimal residual disease settings rather than during active bulky disease.
At the same time, the exploratory signal in PD-L1–high tumors suggests that biomarker-selected populations could still derive benefit from more intensified immune approaches and may warrant future investigation.
Conclusion
CheckMate 73L demonstrated that nivolumab-based concurrent chemoradiotherapy strategies failed to improve survival outcomes over standard durvalumab consolidation in unresectable stage III NSCLC, while increasing pneumonitis risk. The study reinforces concurrent chemoradiotherapy followed by durvalumab as the current standard of care and highlights the need for more precise, biomarker-driven immunotherapy strategies in locally advanced NSCLC.
Read Full Article Here