New patient-reported outcome (PRO) data from the phase III TROPION-Breast02 trial presented at the ESMO Breast Cancer 2026 further strengthen the clinical case for datopotamab deruxtecan (Dato-DXd) as a potential new first-line standard of care for patients with locally recurrent inoperable or metastatic triple-negative breast cancer (mTNBC) who are not candidates for immunotherapy.
Presented by Peter Schmid, the analysis showed that patients receiving Dato-DXd experienced delayed deterioration in physical functioning, global health status, and multiple symptom domains compared with investigator’s choice chemotherapy, complementing the previously reported improvements in progression-free and overall survival.
The findings are particularly important in metastatic triple-negative breast cancer, where treatment-related toxicity and quality-of-life deterioration often occur early during chemotherapy-based treatment.

TROPION-Breast02 Evaluated First-Line Dato-DXd Against Standard Chemotherapy
TROPION-Breast02 is a global, randomized, open-label phase III study evaluating first-line Dato-DXd versus investigator’s choice chemotherapy in patients with locally recurrent inoperable or metastatic TNBC for whom immunotherapy was not considered an appropriate option.
A total of 644 patients were randomized 1:1 to:
- Dato-DXd 6 mg/kg every 3 weeks (n=323)
- Investigator’s choice chemotherapy (n=321)
- Chemotherapy options included paclitaxel, nab-paclitaxel, capecitabine, eribulin, and carboplatin.
Eligible patients had not received prior chemotherapy or targeted systemic therapy in the metastatic setting and had ECOG performance status 0–1.
The dual primary endpoints were progression-free survival and overall survival, while secondary endpoints included objective response rate, duration of response, safety, and patient-reported outcomes.
Previous analyses demonstrated statistically significant and clinically meaningful improvements in progression-free survival and overall survival with Dato-DXd versus chemotherapy. Importantly, patients receiving Dato-DXd also remained on treatment for substantially longer durations than those receiving chemotherapy.
The new ESMO Breast Cancer 2026 presentation focused specifically on how treatment affected symptoms, physical functioning, and overall quality of life.

Dato-DXd Delayed Deterioration in Global Health Status and Physical Function
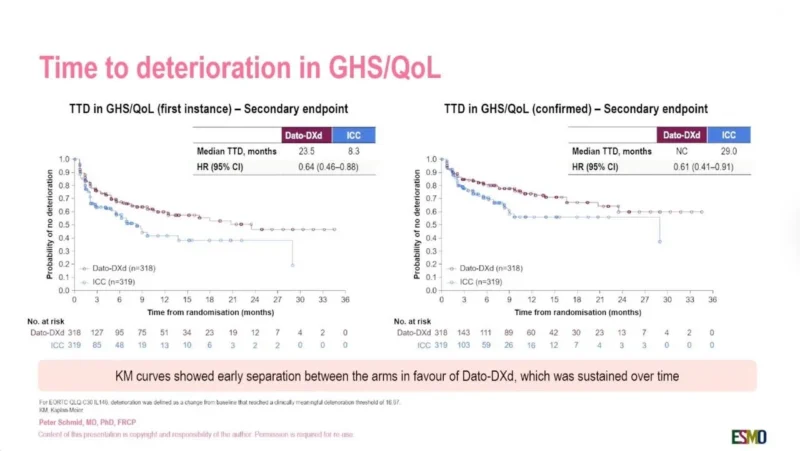
The patient-reported outcome analysis evaluated time to deterioration (TTD) using validated EORTC quality-of-life instruments.
Across multiple domains, hazard ratios numerically favored Dato-DXd, indicating delayed symptom worsening and preserved functioning compared with chemotherapy.
For global health status and quality of life:
- Median TTD (first deterioration) was 23.5 months with Dato-DXd versus 8.3 months with chemotherapy
- HR 0.64 (95% CI 0.46–0.88)
For confirmed deterioration in global health status/QoL:
- HR 0.61 (95% CI 0.41–0.91)
Kaplan-Meier curves presented during the session demonstrated early and sustained separation favoring Dato-DXd over chemotherapy.
Physical functioning also favored Dato-DXd:
- HR 0.67 for first deterioration
- HR 0.59 for confirmed deterioration
The data suggest that patients receiving Dato-DXd maintained daily functioning longer than those receiving standard chemotherapy.
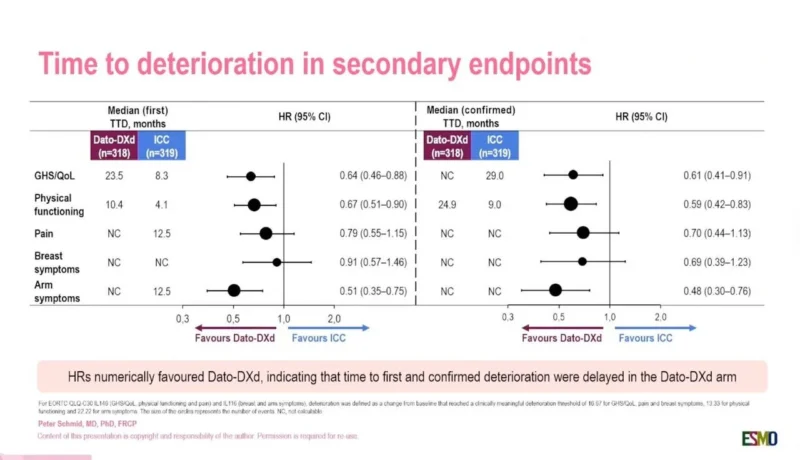
Arm Symptoms and Pain Outcomes Also Favored Dato-DXd
The analysis also assessed breast symptoms, arm symptoms, and pain.
Among these domains, arm symptoms demonstrated one of the strongest improvements:
- HR 0.51 for first deterioration
- HR 0.48 for confirmed deterioration
Pain outcomes numerically favored Dato-DXd as well:
- HR 0.79 for first deterioration
- HR 0.70 for confirmed deterioration
Although confidence intervals crossed unity for pain endpoints, the overall pattern consistently favored the antibody-drug conjugate over chemotherapy.
The investigators noted that patient-reported symptomatic adverse events and treatment tolerability remained generally consistent with clinician-reported safety findings from the primary analysis.
Why These Findings Matter
Triple-negative breast cancer remains one of the most aggressive breast cancer subtypes, and many patients who are not candidates for immunotherapy still rely heavily on conventional chemotherapy.
While extending survival remains the primary goal in metastatic disease, maintaining physical function and minimizing symptom burden are increasingly recognized as equally important clinical priorities.
The TROPION-Breast02 PRO analysis suggests that Dato-DXd may provide not only superior efficacy but also a more tolerable long-term treatment experience compared with standard chemotherapy approaches.
This distinction becomes particularly meaningful given that patients receiving Dato-DXd remained on therapy for substantially longer durations than patients receiving chemotherapy, yet still demonstrated delayed quality-of-life deterioration.
Dato-DXd Continues Expanding Across Breast Cancer
Datopotamab deruxtecan is a TROP2-directed antibody-drug conjugate jointly developed by AstraZeneca and Daiichi Sankyo.
The drug continues to generate extensive interest across multiple breast cancer settings due to its combination of efficacy, manageable toxicity, and potential quality-of-life advantages over traditional chemotherapy.
With TROPION-Breast02 now showing improvements in survival outcomes alongside favorable patient-reported outcomes, Dato-DXd is increasingly positioned as a potential first-line treatment standard for patients with metastatic TNBC who cannot receive immunotherapy.


