For older women with early-stage estrogen receptor-positive breast cancer, treatment decisions are often more complex than standard algorithms suggest. Tumor biology matters, but so do frailty, multimorbidity, life expectancy, functional status, caregiver support, and patient preference. In this setting, the question is not simply how to control the cancer, but how to do so without causing unnecessary burden. A new prospective trial now suggests that circulating tumor DNA may help identify which older patients can safely avoid surgery and remain on primary endocrine therapy alone.
The study, titled “Use of ctDNA in Older Women with ER+ Breast Cancer to Facilitate Surgical De-Escalation: A Prospective, Hybrid-Decentralized Trial with Correlative Studies,” was published in Clinical Cancer Research by Carleton et al. It evaluated whether longitudinal ctDNA monitoring using the personalized tumor-informed Signatera assay could distinguish patients at low versus high risk of progression among women aged 70 years and older with stage I–III ER-positive/HER2-negative breast cancer who chose to forgo surgery in favor of primary endocrine therapy (Carleton et al., Clin Cancer Res, 2026).
Why This Question Matters
De-escalation in older adults with breast cancer has become an increasingly important area of research. Prior studies have shown that carefully selected older women with ER-positive disease may avoid certain locoregional interventions, particularly when competing causes of mortality are substantial and tumor behavior is indolent (Biganzoli et al., 2021; Wyld et al., 2021; Chadha et al., 2023). Yet one of the major unresolved issues is how to identify which patients can safely remain on endocrine therapy alone and which are at risk of local or distant progression.
This is especially relevant because primary endocrine therapy can be clinically effective in some older patients, but it is not without limitations. Earlier retrospective work by the same group found that while breast cancer-specific mortality remained low in this setting, locoregional progression still occurred in about 20% of patients over time, suggesting that better biological selection is needed (Carleton et al., 2024). ctDNA offered a possible solution: a minimally invasive biomarker capable of dynamic, real-time assessment of tumor behavior.
Study Design and Patient Population
This was a prospective, single-center, observational, non-randomized hybrid-decentralized study conducted at the University of Pittsburgh and registered as NCT05914792. Eligible patients were women aged 70 years or older with histologically confirmed stage I–III ER-positive/HER2-negative non-metastatic breast cancer who elected to forgo surgery and begin primary endocrine therapy. Patients needed adequate tumor tissue for assay development and the ability to provide serial blood samples.
A total of 43 patients were enrolled between April 2022 and April 2024, and 34 had a baseline pre-treatment ctDNA blood draw. Median age at diagnosis was 86 years with a range of 75 to 94 years. Most patients had ECOG performance status 1 or 2, tumors larger than 2 cm, no clinical nodal involvement, and received a non-steroidal aromatase inhibitor such as letrozole or anastrozole as first-line endocrine therapy (Carleton et al., Clin Cancer Res, 2026).
The study was notable for its decentralized design. 72.1% of patients were enrolled through community oncology sites, and many depended on caregivers for transport. Reflecting that reality, 70% of participants chose mobile phlebotomy rather than returning to the clinic for serial blood draws. This is an important practical detail because it shows that biomarker-guided surveillance in older adults may be feasible outside major academic centers.
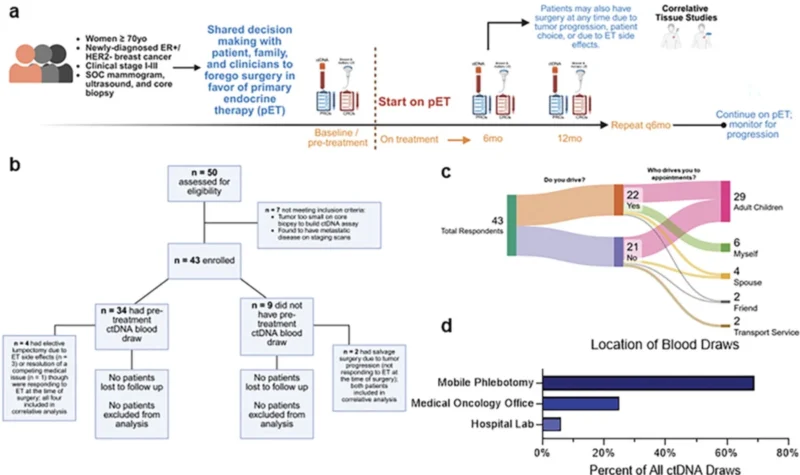
©2026 American Association for Cancer Research.
How ctDNA Was Measured
The ctDNA assay used was Signatera, a personalized tumor-informed multiplex PCR next-generation sequencing platform. For each patient, tumor tissue and matched normal blood were used to identify up to 16 somatic single-nucleotide variants by whole-exome sequencing. A patient-specific assay was then created to longitudinally quantify ctDNA in plasma. Samples with more than 2 variants detected above the predefined threshold were considered ctDNA-positive, and ctDNA concentration was reported as mean tumor molecules per milliliter of plasma (MTM/mL).
Blood was collected every 3 to 6 months, aligned with standard-of-care imaging, most commonly breast and axillary ultrasound, with optional mammography. Tumor progression was defined by RECIST 1.1 criteria.
What the Trial Found
The central finding was clear: ctDNA status strongly tracked with clinical outcomes.
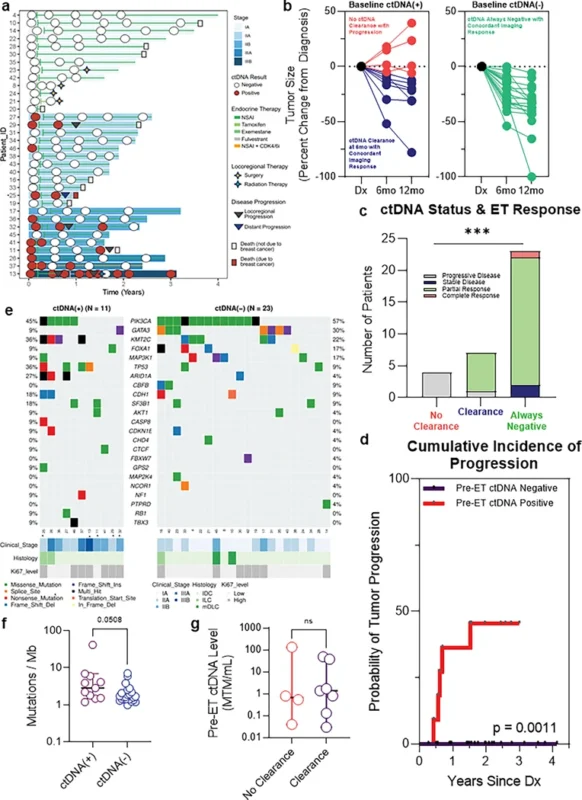
Among the 34 patients with baseline ctDNA assessment, 32% were ctDNA-positive before treatment. Over a median follow-up of 2.75 years (range 0.25 to 4.25 years), 5 of 34 patients (15%) experienced tumor progression. Only 1 of 34 patients (2.9%) died from breast cancer, while 8 of 34 (24%) died from non-breast cancer causes, underlining the importance of competing mortality in this age group (Carleton et al., Clin Cancer Res, 2026).
The most striking result was that no patient with pre-treatment ctDNA negativity experienced tumor progression. In contrast, pre-treatment ctDNA positivity was strongly associated with increased risk. In a competing risk regression adjusted for age, tumor stage, tumor grade, and Ki-67, baseline ctDNA positivity was associated with a hazard ratio of 30 for tumor progression with a 95% confidence interval of 4.4 to 209 and a p value of 0.0005.
This is a remarkably large effect size, although it should be interpreted in the context of a small sample.
The Six-Month ctDNA Landmark Was Especially Informative
The trial also showed that early ctDNA dynamics on endocrine therapy mattered greatly.
Patients with baseline ctDNA positivity who cleared ctDNA by 6 months generally showed response to endocrine therapy and remained free of distant progression. By contrast, 100% of patients with persistent ctDNA positivity at 6 months experienced tumor progression. At the same time, 100% of patients with baseline and persistent ctDNA negativity remained progression-free throughout follow-up, with a statistically significant association (p = 0.002).
This suggests that the 6-month ctDNA assessment may represent a clinically actionable checkpoint. If ctDNA remains detectable despite endocrine therapy, it may indicate that endocrine monotherapy is not sufficient and that escalation should be considered.
That escalation might involve surgery, palliative radiation, switching endocrine therapy, or potentially future biomarker-guided strategies using oral SERDs or CDK4/6 inhibitors, although those approaches were not formally tested in this study.
©2026 American Association for Cancer Research.
Stage I Tumors Appeared Especially Indolent
Another noteworthy observation was that all clinical stage I (cT1N0) tumors were ctDNA-negative at baseline, regardless of grade or Ki-67, and all remained ctDNA-negative during follow-up. None of those patients progressed.
This may reflect two overlapping possibilities. First, very low-volume tumors may fall near or below the technical limit of plasma detection. Second, these tumors may represent a biologically indolent subgroup in older women, which would support a less aggressive treatment strategy in carefully selected cases.
How Closely Did ctDNA Match Imaging?
The concordance between ctDNA and imaging was one of the study’s strongest practical findings. Longitudinal ctDNA patterns paralleled serial ultrasound changes in tumor size. Tumors that were ctDNA-negative throughout, or that became negative by 6 months, generally showed tumor stability or shrinkage. Tumors with ongoing ctDNA positivity showed growth and eventual progression.
In one patient who had more than 10 longitudinal blood draws, ctDNA closely tracked serial CT imaging across multiple lines of endocrine therapy and palliative radiation. This level of concordance supports ctDNA not just as a prognostic biomarker, but as a real-time surveillance tool.
Genomic Features of ctDNA-Positive Versus ctDNA-Negative Tumors
The authors also explored genomic correlates using whole-exome sequencing from diagnostic core biopsies.
Tumors with baseline ctDNA negativity were enriched for mutations commonly associated with luminal, endocrine-responsive biology, including PIK3CA, GATA3, and MAP3K1. In contrast, tumors with baseline ctDNA positivity were more often associated with genomic alterations linked to endocrine resistance or more aggressive biology, including TP53 and ARID1A.
Baseline ctDNA-positive tumors also tended to have higher tumor mutational burden, with a borderline association (p = 0.0508). Interestingly, the quantitative baseline ctDNA level itself did not distinguish those who would later clear ctDNA from those who would not (p = 0.79). This suggests that the simple presence or persistence of ctDNA may be more clinically useful than the starting ctDNA quantity alone.
The investigators also assessed the Magee Equations, which act as a surrogate for Oncotype DX recurrence score, and found that these scores did not predict ctDNA status or ctDNA clearance. That is important because it suggests ctDNA may provide distinct information not captured by traditional clinicopathologic or genomic risk models.
©2026 American Association for Cancer Research.
Patient-Reported Outcomes Add Important Context
This trial is especially valuable because it did not focus solely on biomarker kinetics. It also captured patient- and caregiver-reported outcomes, which are essential in geriatric oncology.
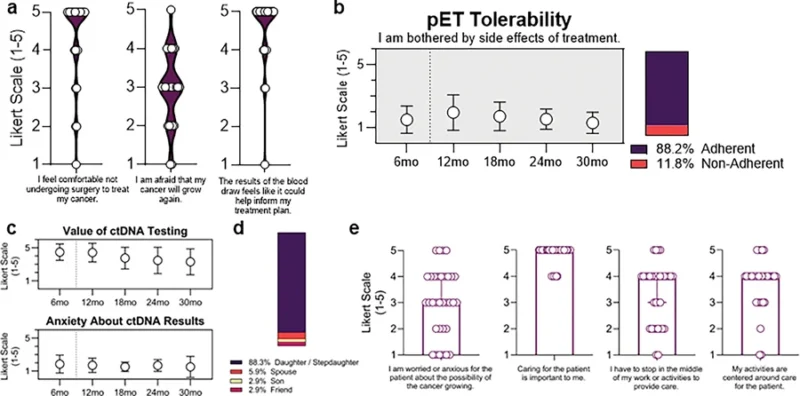
Among patients, 93% completed at least one survey. Overall, 82% reported feeling comfortable not undergoing surgery, though 92% were at least a little concerned about the possibility of tumor growth or progression. More than 80% felt the ctDNA blood draws could help inform their treatment plan, and importantly, patients generally did not report increased anxiety related to ctDNA testing.
Adherence to primary endocrine therapy was 88.2%, which is consistent with prior data in this setting (Hubbard et al., 2024). Many patients reported being only “a little bit” bothered by side effects.
This matters because one concern with repeated biomarker testing is that it could worsen distress. In this study, that did not appear to happen.
Caregiver Burden Was Also Measured
The caregiver data were just as revealing. 86% of patients had caregivers present during visits, and 89% of those caregivers completed at least one survey. Most caregivers were adult daughters, stepdaughters, or daughters-in-law.
While caregivers felt that caring for the patient was very important, many reported interruptions in work and daily activities, indicating a significant caregiver burden. This is one of the reasons the decentralized structure of the trial is so relevant. Home-based blood collection may reduce some of the treatment burden not only on patients but also on families.
What the Correlative Biology Showed
One of the most scientifically interesting aspects of the study was its correlative analysis of tumor microenvironment changes in a subset of patients who underwent surgery after receiving long-term primary endocrine therapy.
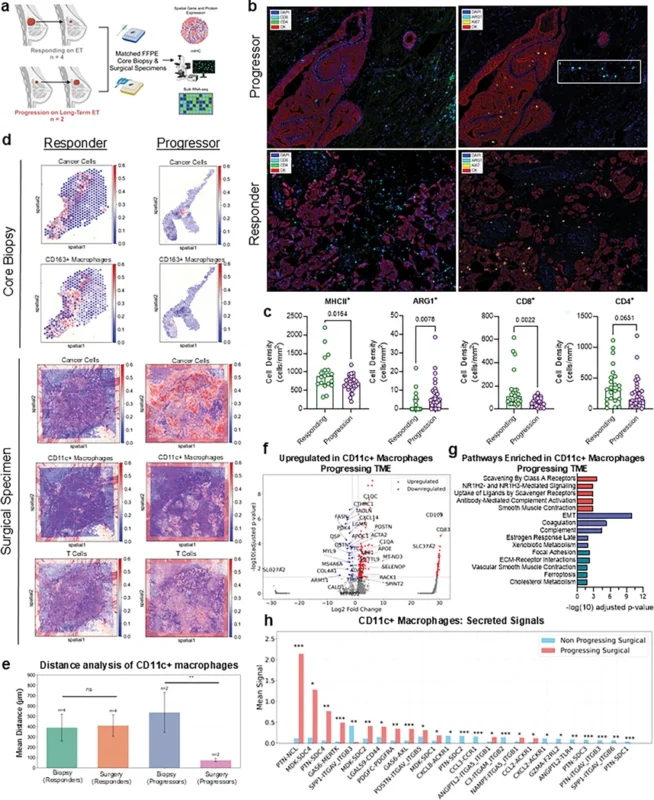
Six patients contributed matched biopsy and surgical specimens. Four were responding to endocrine therapy at the time of surgery but stopped treatment because of side effects or resolution of another medical issue. Two underwent surgery because of tumor progression. All responding tumors were ctDNA-negative, while both progressing tumors were ctDNA-positive.
Spatial transcriptomic and multiplex immunohistochemistry analyses showed that progressing tumors had a distinctly immunosuppressive microenvironment. Compared with responding tumors, progressing tumors contained fewer CD8+ T cells, fewer CD4+ T cells, fewer MHC II+ macrophages, and more ARG1+ macrophages. The progressing tumor microenvironment also showed CD11c+ macrophages in closer proximity to proliferating cancer epithelial cells.
These macrophages upregulated CD109 and CD89, and secreted molecules including CCL2, pleiotrophin, midkine, and LGALS9, all of which are associated with immunosuppressive signaling, T-cell exclusion, and tumor-supportive behavior. Pathway analyses also suggested activation of MYC, oxidative phosphorylation, and mTORC signaling in progressing cancer cells, while one progressing sample harbored an ESR1 D538G mutation, a known mechanism of endocrine resistance.
Taken together, these data suggest that ctDNA-positive progression on primary endocrine therapy is not merely a radiographic phenomenon. It reflects underlying biological evolution involving endocrine resistance and macrophage-driven remodeling of the tumor microenvironment.
©2026 American Association for Cancer Research.
What This Means Clinically
This trial does not establish a new standard of care yet, but it does provide one of the clearest prospective demonstrations that ctDNA may help guide surgical de-escalation in older women with ER-positive breast cancer.
The clinical message is not that surgery should be omitted broadly. Rather, it is that a biologically selected subgroup may exist in whom long-term endocrine therapy alone is sufficient, and ctDNA may help identify them.
At the same time, the study highlights a group at clearly higher risk: patients with baseline ctDNA positivity, especially if ctDNA remains positive after 6 months. These patients may need a different strategy and should not necessarily be reassured by endocrine therapy alone.
This type of approach fits well with the idea of “right-sizing” therapy in older adults, where both overtreatment and undertreatment carry real harms.
Limitations
The limitations are important. This was a small, single-center, non-randomized study. Only 34 patients had baseline ctDNA data, and only 5 progression events occurred. That means the hazard ratio estimates, while striking, are based on limited numbers. The follow-up, although reasonable for a frail older population, is still relatively short for ER-positive breast cancer, where late events are common.
In addition, treating physicians and patients were not blinded to imaging or ctDNA results, which may have influenced management decisions. And while the correlative biology is intriguing, it is hypothesis-generating rather than definitive.
The Bottom Line
This study provides compelling pilot evidence that longitudinal ctDNA monitoring may help identify older women with ER-positive/HER2-negative early breast cancer who can safely avoid surgery and remain on primary endocrine therapy. It also suggests that persistent ctDNA positivity, especially at 6 months, identifies patients at high risk of progression and may define a window for treatment escalation.
Just as importantly, the study shows that this type of monitoring can be delivered in a patient-centered, decentralized way, with strong patient acceptance and without major added anxiety.
The next step is clear: larger, randomized biomarker-guided trials are needed. But for now, this work by Carleton and colleagues provides one of the strongest arguments yet that ctDNA could become a practical tool for personalizing surgical de-escalation in older adults with breast cancer.
You can read full article here


