SERENA-6 trial was presented during the ESMO Breast Cancer Annual Congress 2026 by François Clément Bidard and showed that repeated circulating tumor DNA monitoring can identify emerging ESR1 mutations during first-line endocrine-based therapy for HR-positive/HER2-negative advanced breast cancer. The findings support continued ctDNA testing during treatment with aromatase inhibitors and CDK4/6 inhibitors, even in patients with initially negative results, allowing earlier identification of endocrine resistance before disease progression.
Background
ESR1 mutations are among the most common mechanisms of acquired resistance in patients receiving aromatase inhibitor–based therapy for HR-positive/HER2-negative advanced breast cancer. These mutations can emerge during treatment and activate estrogen receptor signaling despite ongoing endocrine therapy.
Importantly, ESR1 mutations may be detected in circulating tumor DNA before radiologic disease progression becomes apparent. Previous analyses have shown that approximately 30–50% of tumors harbor ESR1 mutations at progression on aromatase inhibitor therapy.
Camizestrant is a next-generation oral selective estrogen receptor degrader (SERD) designed to target both wild-type and mutant estrogen receptors. Earlier findings from the Phase III SERENA-6 trial demonstrated that switching patients from aromatase inhibitor therapy to camizestrant while continuing CDK4/6 inhibition significantly improved progression-free survival compared with continuing aromatase inhibitor therapy.
At the second data cutoff, median progression-free survival reached 16.6 months with camizestrant plus CDK4/6 inhibitor versus 9.2 months with continued aromatase inhibitor plus CDK4/6 inhibitor therapy (HR 0.46). The regimen also improved chemotherapy/ADC-free survival and delayed deterioration in global health status and quality of life.
The current presentation focused specifically on the ESR1 mutation detection and surveillance phase of SERENA-6.
Study Design and Methods
Patients with HR-positive/HER2-negative advanced breast cancer receiving first-line aromatase inhibitor plus CDK4/6 inhibitor therapy could enter the SERENA-6 surveillance program at any point during treatment, provided they had received at least six months of therapy.
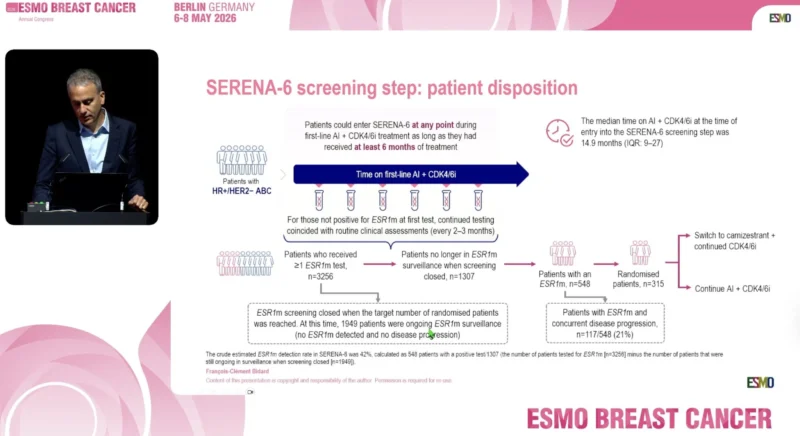
ESR1 mutation testing was performed using the Guardant360 CDx assay. For patients without detectable ESR1 mutations on the first test, repeat ctDNA assessments were conducted every two to three months alongside routine clinical evaluations.
The median duration of AI plus CDK4/6 inhibitor therapy at study entry was 14.9 months.Patients who developed ESR1 mutations became eligible for randomization to either switch to camizestrant while continuing CDK4/6 inhibition or continue aromatase inhibitor therapy plus CDK4/6 inhibitor treatment.
Results
Between June 2021 and June 2024, 3,256 patients across 264 sites in 23 countries underwent at least one valid ESR1 mutation test during first-line therapy.The median number of ESR1 mutation tests per patient was three, with an interquartile range of one to five tests.
At the first ctDNA assessment, approximately 10% of patients were identified as having an ESR1 mutation. The highest first-test detection rates were observed during 12–48 months of AI plus CDK4/6 inhibitor therapy.
Detection rates at the first test included:
- 6.4% during 6–12 months of therapy
- 10.1% during >12–24 months
- 12.8% during >24–36 months
- 11.5% during >36–48 months
- 8.8% during >48–60 months
- 6.3% after more than 60 months of therapy
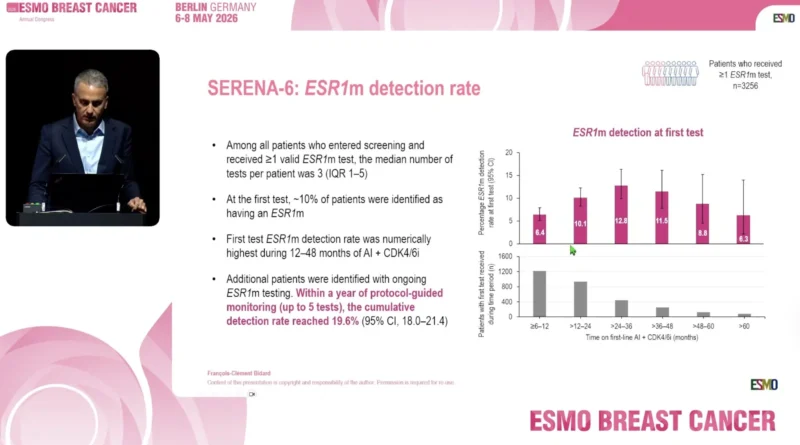
Importantly, repeated testing identified many additional patients whose first ctDNA assessment had been negative. Within one year of protocol-guided monitoring, including up to five sequential tests, the cumulative ESR1 mutation detection rate increased to 19.6% (95% CI 18.0–21.4) for patiens who were screened over 12 months. Overall, 548 patients developed detectable ESR1 mutations during surveillance. Among randomized patients, 94% had at least one of the four most common ESR1 mutations, all detectable using currently available assays.
The most frequently identified ESR1 mutations included:
- D538G (48%)
- Y537S (38%)
- Y537N (17%)
- E380Q (10%)
Multiple ESR1 variants were identified in 18% of patients, suggesting ongoing molecular evolution during endocrine therapy.
Key Findings
The SERENA-6 analysis demonstrated that ESR1 mutations can emerge across a broad treatment timeframe during first-line endocrine therapy. While mutation detection was numerically highest between 12 and 48 months of therapy, clinically relevant mutations were identified both earlier and later during treatment.
Repeated ctDNA monitoring substantially increased mutation detection compared with single-timepoint testing and allowed ongoing identification of patients who may benefit from therapeutic intervention before disease progression.
The study also demonstrated the practical feasibility of performing ctDNA monitoring alongside routine blood work during standard clinic visits.
Conclusion
SERENA-6 supports the integration of serial ctDNA monitoring into routine management of HR-positive/HER2-negative advanced breast cancer treated with aromatase inhibitors and CDK4/6 inhibitors. The findings suggest that ESR1 mutation testing should continue throughout treatment rather than being performed only once, as ongoing surveillance may identify endocrine resistance earlier and help guide treatment adaptation before clinical progression occurs.


