SATEEN trial was presented during the ESMO Breast Cancer Annual Congress 2026 by Paolo Tarantino and evaluated sacituzumab govitecan plus trastuzumab in patients with HER2-positive metastatic breast cancer previously treated with trastuzumab deruxtecan. The study addressed an important clinical question: whether another TOPO1 inhibitor–based antibody-drug conjugate strategy could provide meaningful activity after prior T-DXd exposure.
Background
Treatment sequencing after trastuzumab deruxtecan remains an unresolved challenge in HER2-positive metastatic breast cancer. Although T-DXd has changed the treatment landscape, outcomes after progression on or after this agent are less clearly defined, and prospective data on subsequent ADC-based approaches remain limited.
Sacituzumab govitecan is an anti-Trop2 antibody-drug conjugate that delivers a topoisomerase I inhibitor payload. In SATEEN, investigators combined sacituzumab govitecan with trastuzumab to explore whether continued HER2 blockade plus a different ADC target could generate activity in this heavily pretreated population.
Study Design
SATEEN was a phase II, single-arm, investigator-initiated, multicenter trial. Eligible patients had advanced HER2-positive cancer and had previously received taxane, trastuzumab, and T-DXd in any setting.
Patients received sacituzumab govitecan at 10 mg/kg intravenously on days 1 and 8 of each 21-day cycle, together with trastuzumab given subcutaneously or intravenously. Treatment continued until disease progression or unacceptable toxicity.
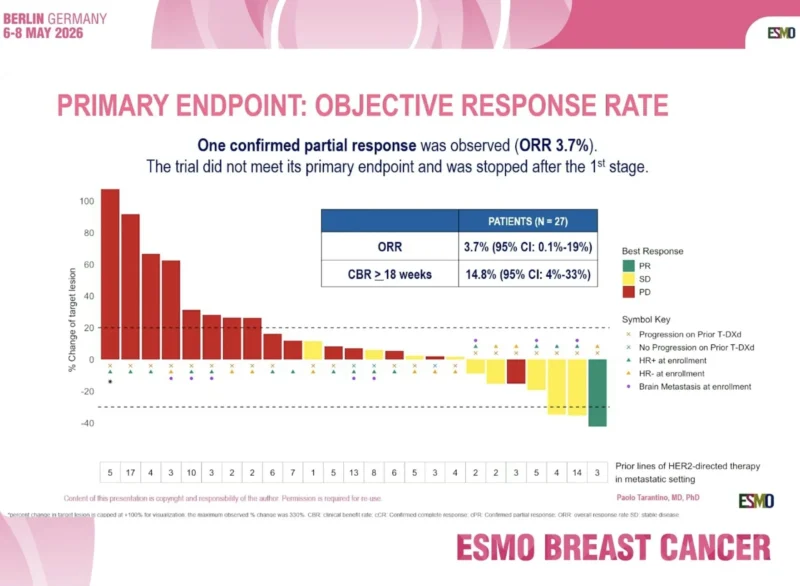
The primary endpoint was objective response rate. The study used a Simon’s two-stage design, with 27 patients planned in stage 1 and up to 40 patients overall. At least 3 responses among the first 27 patients were required to proceed to stage 2.
Results from SATEEN Trial
A total of 27 patients were enrolled in stage 1 and started treatment. The population was heavily pretreated, with a median of 5 prior lines of HER2-directed therapy for metastatic disease. Overall, 59% had estrogen receptor–positive disease, and 33% had brain metastases at enrollment, including 18% with active and 15% with stable brain metastases.
The trial did not meet its primary endpoint. Only one confirmed partial response was observed, corresponding to an objective response rate of 3.7% with a 95% confidence interval of 0.1% to 19.0%. Because the required response threshold was not reached, the trial was stopped after the first stage.
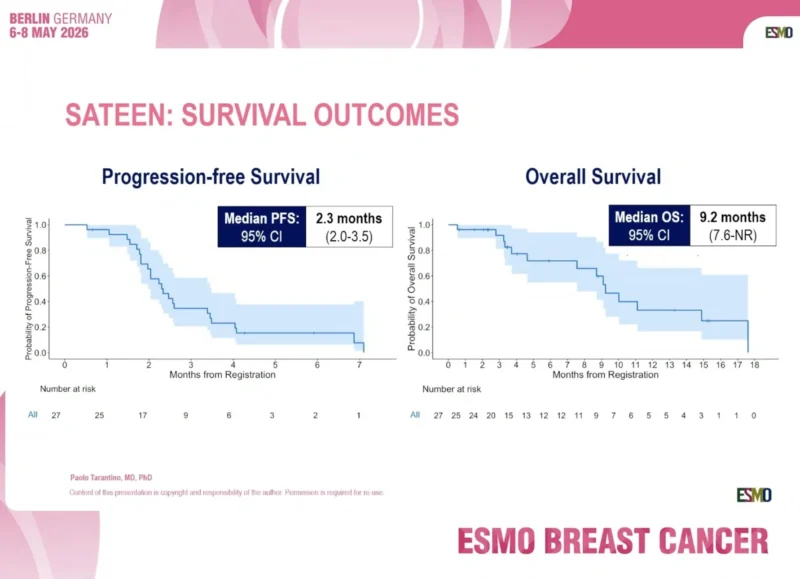
The 18-week clinical benefit rate was 14.8%. Median progression-free survival was 2.3 months, with a 95% confidence interval of 2.0 to 3.5 months. Median overall survival was 9.2 months, with a 95% confidence interval of 7.6 months to not reached.
The most common grade 3 or higher treatment-related adverse events were neutropenia, anemia, and diarrhea. Grade ≥3 neutropenia occurred in 59% of patients, anemia in 26%, and diarrhea in 19%.
One grade 5 treatment-related adverse event occurred, reported as neutropenic sepsis with multi-organ failure. After this event, mandatory growth factor support was implemented in the study.
Conclusion
In patients with HER2-positive metastatic breast cancer previously treated with T-DXd, sacituzumab govitecan plus trastuzumab did not demonstrate clinically meaningful activity. The SATEEN results help clarify treatment expectations after T-DXd and reinforce the importance of developing new approaches for patients progressing after modern HER2-directed ADC therapy.


