PREcoopERA trial, presented during the ESMO Breast Cancer Annual Congress 2026, evaluated whether the oral SERD giredestrant could effectively reduce tumor proliferation in premenopausal patients with untreated ER-positive, HER2-negative early breast cancer, either with or without ovarian function suppression.
In this 28-day window-of-opportunity study, both giredestrant-based approaches showed strong Ki67 reduction, but giredestrant plus LHRH analogue was not superior to anastrozole plus LHRH analogue, and giredestrant alone did not meet non-inferiority compared with giredestrant plus LHRH analogue, suggesting that ovarian function suppression remains important for the strongest anti-proliferative effect.
Background
Endocrine therapy in premenopausal patients is influenced by higher circulating estradiol levels, which may impact treatment efficacy. Oral SERDs, such as giredestrant, act through estrogen receptor degradation and competition for estradiol binding, potentially maintaining activity in this setting.
Giredestrant has previously demonstrated improved anti-proliferative activity compared with anastrozole in postmenopausal patients in the coopERA trial, and improved invasive disease-free survival in the Phase III lidERA trial, where LHRH analogue was required for premenopausal patients.
PREcoopERA was designed to investigate the anti-proliferative activity of giredestrant with or without ovarian function suppression in premenopausal patients.
Methods
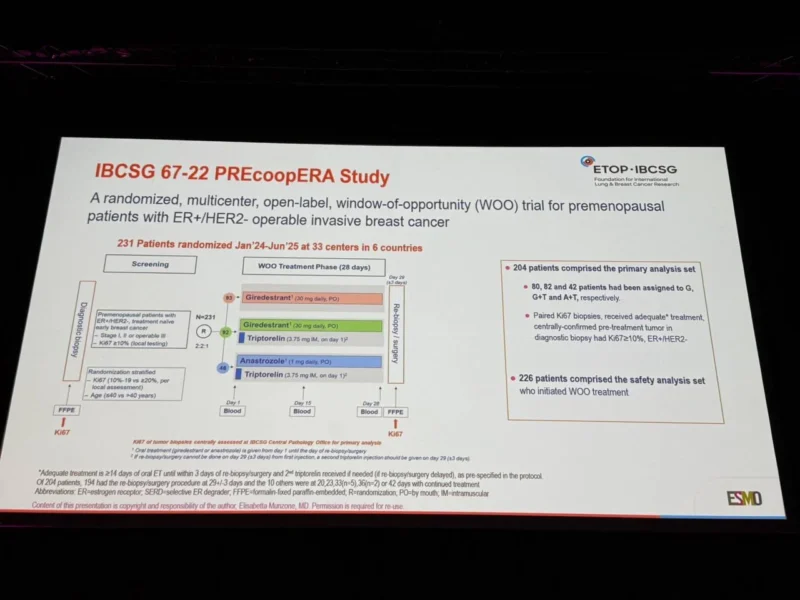
PREcoopERA is a window-of-opportunity trial enrolling premenopausal patients with untreated ER-positive, HER2-negative stage I–III early breast cancer with baseline Ki67 greater than 10%.
Patients were randomized to 28 days of treatment with:
- Giredestrant 30 mg daily (G)
- Giredestrant plus triptorelin (GT)
- Anastrozole 1 mg daily plus triptorelin (AT)
Tumor re-biopsy was performed on Day 29. The primary endpoint was change in Ki67, centrally assessed on paired tumor biopsies, blinded to treatment allocation.The trial was powered to test superiority of GT over AT and non-inferiority of G compared with GT.

Study Design
A total of 231 patients were randomized across 33 centers between January 2024 and June 2025. The primary analysis population included 204 patients, with 82 assigned to GT, 80 to G, and 42 to AT.
Most patients were older than 40 years (79%), and 53% had baselinebaseline Ki67 ≥10%.
Results
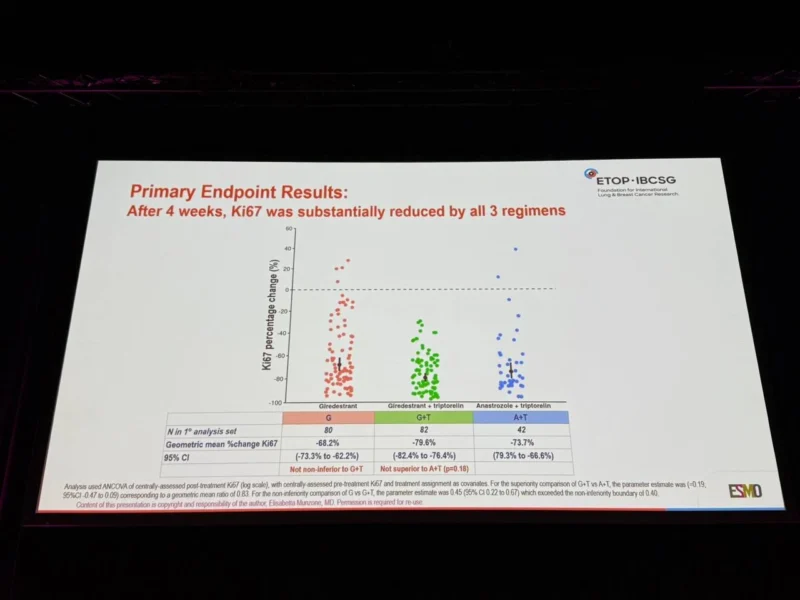
All treatment arms demonstrated substantial reductions in Ki67. The geometric mean reduction in Ki67 was:
- −79.6% (95% CI −82.4 to −76.4) with GT
- −73.7% (95% CI −79.3 to −66.6) with AT
- −68.2% (95% CI −73.3 to −62.2) with G
The comparison between GT and AT did not demonstrate statistical superiority, with a log difference of −0.19 (95% CI −0.47 to 0.09; p=0.18).Giredestrant alone did not meet the predefined non-inferiority margin compared with GT, with a log difference of 0.45 (95% CI 0.22 to 0.67), exceeding the non-inferiority bound of 0.40.
What PREcoopERA Shows About Ki67 Suppression and Ovarian Function Suppression
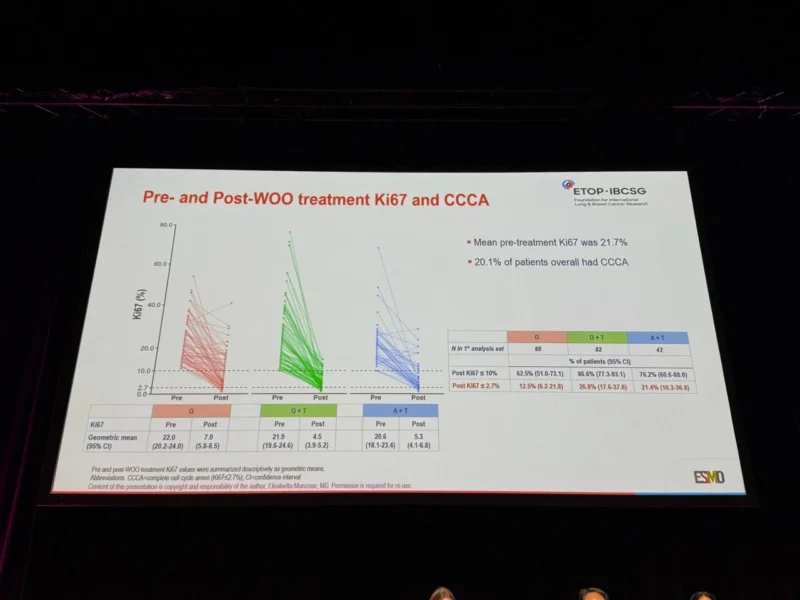
Beyond the primary endpoint, PREcoopERA also showed differences in the depth of Ki67 suppression across treatment arms. The mean pre-treatment Ki67 level was 21.7%, and 20.1% of patients overall achieved complete cell cycle arrest, defined as Ki67 ≤2.7%, after the 28-day treatment period.
Post-treatment Ki67 ≤10% was observed in 86.6% of patients receiving giredestrant plus triptorelin, compared with 76.2% receiving anastrozole plus triptorelin and 62.5% receiving giredestrant alone. A deeper suppression to Ki67 ≤2.7% was achieved in 26.8%, 21.4%, and 12.5% of patients, respectively.
These findings support the conclusion that giredestrant has biological activity in premenopausal patients, including when given without ovarian function suppression. However, the deeper Ki67 suppression observed with giredestrant plus triptorelin suggests that ovarian function suppression remains important for achieving the strongest anti-proliferative effect in this population.
The study also reported a correlation between estrogen receptor degradation and Ki67 reduction, supporting the on-target activity of giredestrant, while age appeared to contribute to variability in response.
Safety
Treatment was generally well tolerated across all groups. Grade 3 adverse events were reported in:
- 2.2% of patients receiving GT
- 4.4% receiving G
- 4.4% receiving AT
Two patients treated with giredestrant alone developed ovarian cysts. No grade 4 or 5 adverse events were reported.
Key Findings of PREcoopERA Trial
Both giredestrant-based regimens demonstrated robust anti-proliferative activity, as reflected by substantial reductions in Ki67.
However, superiority of giredestrant plus LHRH analogue over anastrozole plus LHRH analogue was not demonstrated.
Giredestrant alone did not meet non-inferiority criteria compared with combination therapy, suggesting that ovarian function suppression remains important for optimal anti-proliferative effect in premenopausal patients.
Conclusion
PREcoopERA showed that all three regimens substantially reduced Ki67 after 4 weeks. Giredestrant plus triptorelin achieved the greatest numerical anti-proliferative effect, but it was not statistically superior to anastrozole plus triptorelin.
Giredestrant alone was biologically active but did not meet non-inferiority compared with giredestrant plus triptorelin, supporting the continued importance of ovarian function suppression in achieving deeper Ki67 suppression in this premenopausal population.


