PHERGain-2 TrialAt the ESMO Breast Cancer Annual Congress 2026, Antonio Llombart Cussac from Valencia, Spain, presented results from the Phase II PHERGain-2 study, evaluating a chemotherapy-free, pCR-guided treatment strategy using trastuzumab–pertuzumab (HP) and T-DM1 in patients with HER2-positive early breast cancer. The study explores whether selected patients can safely omit chemotherapy while maintaining efficacy and preserving quality of life.
The study reported a 59.6% pCR rate, clinically meaningful preservation of health-related quality of life, expected HP/T-DM1 toxicity, and ongoing three-year recurrence-free interval follow-up.

Background
Neoadjuvant treatment strategies in HER2-positive early breast cancer have traditionally relied on chemotherapy combined with HER2-targeted agents. However, treatment-related toxicity remains a significant concern, particularly in patients with lower tumor burden or favorable biology.
PHERGain-2 was designed to evaluate a response-adapted, chemotherapy-free approach guided by pathologic complete response. The study investigates whether dual HER2 blockade with trastuzumab and pertuzumab, followed by selective escalation to T-DM1 based on residual disease, can achieve meaningful clinical outcomes while reducing treatment burden.
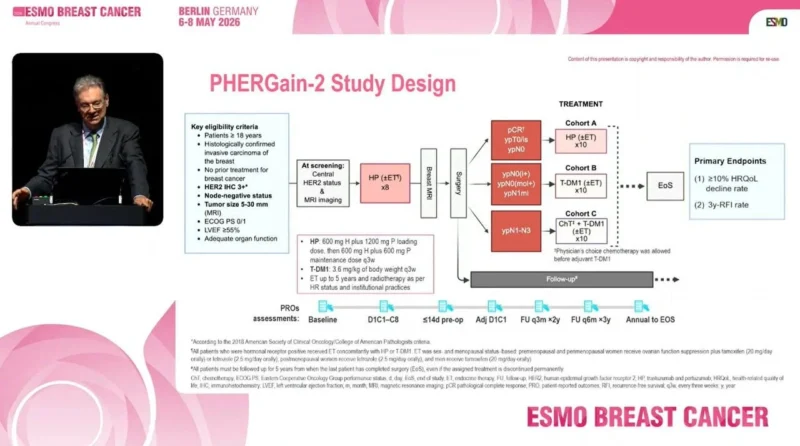
Methods
PHERGain-2 (NCT04733118) is a multicenter, single-arm, open-label Phase II study. It enrolled patients with untreated, centrally confirmed HER2-positive (IHC 3+), node-negative early breast cancer, with tumor size between 5 and 30 mm assessed by central MRI.
Patients received neoadjuvant subcutaneous trastuzumab–pertuzumab every three weeks for eight cycles. Following surgery, adjuvant treatment was adapted according to pathologic response.
Patients achieving pCR (ypT0/is, ypN0) continued trastuzumab–pertuzumab to complete a total of 18 cycles (cohort A). Patients with residual invasive disease or minimal nodal involvement received T-DM1 for 10 cycles (cohort B). Those with higher nodal burden (ypN1–3) could receive optional adjuvant chemotherapy before T-DM1 (cohort C). Patients with hormone receptor-positive tumors received endocrine therapy during both neoadjuvant and adjuvant phases.
Co-primary endpoints were one-year health-related quality of life decline, defined as a ≥10% decrease in EORTC QLQ-C30 global score, and three-year recurrence-free interval. Secondary endpoints included pCR and safety.
Results
Between August 2021 and March 2024, 396 patients were enrolled. The median tumor size was 18 mm, and 72.7% of tumors were hormone receptor-positive. T1 disease was present in 255 of 396 patients, approximately 64.4%.
A total of 391 patients (98.7%) underwent surgery. Pathologic complete response was achieved in 236 patients, corresponding to 59.6%, who were assigned to cohort A. Among patients with residual disease, 148 (37.8%) entered cohort B and seven (1.8%) cohort C.
Baseline mean global health-related quality of life score was 78.6 (95% CI 76.8–80.5). At one year, a ≥10% decline in quality of life was observed in 154 of 360 evaluable patients, corresponding to 42.8% (95% CI 36.9–48.8).
Quality of life decline differed according to response. Among patients achieving pCR, 37.3% experienced decline, compared with 51.9% in those without pCR. By hormone receptor status, decline occurred in 36.1% of HR-negative patients and 45.2% of HR-positive patients.
Treatment-related adverse events of any grade were reported in 86.6% of patients, with grade 3 events in 5.6%. Serious adverse events occurred in 6.1% of patients. One patient (0.3%) died due to T-DM1-related pneumonitis.

Key Findings
The PHERGain-2 study demonstrated a pCR rate of 59.6% in a chemotherapy-free neoadjuvant setting. Most patients were able to avoid chemotherapy, with escalation to T-DM1 reserved for those with residual disease.
Health-related quality of life was preserved in a substantial proportion of patients, although a notable proportion experienced decline at one year, particularly among those without pCR.
The safety profile was consistent with known toxicities of HER2-targeted therapies, with a low incidence of grade 3 events and rare treatment-related mortality.
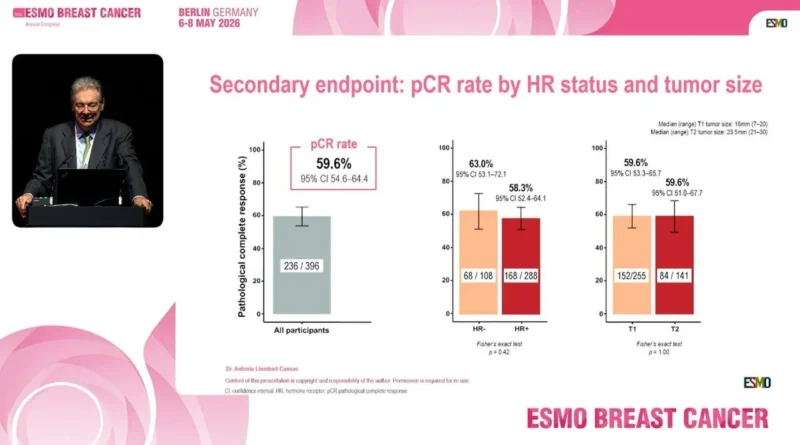
pCR Was Similar Across HR Status And Tumor Size
In PHERGain-2, the overall pCR rate was 59.6%. By hormone receptor status, pCR was achieved in 63.0% of patients with HR-negative disease and 58.3% of patients with HR-positive disease, with no significant difference between groups. By tumor size, pCR was 59.6% in both T1 and T2 tumors.
Conclusion
PHERGain-2 suggests that a response-adapted, chemotherapy-free strategy using trastuzumab–pertuzumab followed by selective T-DM1 may be clinically feasible in selected low-risk patients with HER2-positive early breast cancer.
The study reported a pCR rate of 59.6%, with similar pCR rates across HR status and tumor size. The safety profile was consistent with known effects of trastuzumab/pertuzumab and T-DM1. Mature three-year recurrence-free interval results are still pending.


