At the ESMO Breast Cancer Congress 2026, Lajos Pusztai presented residual cancer burden analysis from the DESTINY-Breast11 study, evaluating neoadjuvant T-DXd-THP versus ddAC-THP in patients with previously untreated, high-risk HER2-positive early breast cancer.
In the primary endpoint analysis, T-DXd-THP showed a pCR rate of 67.3%, compared with 56.3% for ddAC-THP, representing an absolute difference of 11.2%. The presentation also reported a favorable safety profile and a statistically significant, clinically meaningful improvement in pCR rate with T-DXd-THP versus ddAC-THP.
Background
In the DESTINY-Breast11 Phase 3 study, neoadjuvant T-DXd-THP previously demonstrated a statistically significant and clinically meaningful improvement in pathologic complete response compared with ddAC-THP, along with an improved safety profile.
Residual cancer burden is a continuous prognostic measure that quantifies residual invasive disease in the breast and axillary lymph nodes after neoadjuvant therapy. It is categorized into four classes, with RCB-0 (no residual disease) and RCB-I (minimal residual disease) associated with more favorable long-term outcomes compared with RCB-II or RCB-III.
Earlier analyses showed higher rates of RCB-0+I with T-DXd-THP compared with ddAC-THP, prompting further detailed RCB evaluation.

Methods
RCB was assessed by blinded central review based on the amount of residual viable tumor identified by hematoxylin and eosin staining after mapping the surgical specimen.
Both RCB class (categorical) and continuous RCB index were analyzed post hoc in patients treated with T-DXd-THP and ddAC-THP.
Study Design
This analysis is based on the Phase 3 DESTINY-Breast11 trial (NCT05113251), comparing two neoadjuvant strategies in high-risk HER2-positive early breast cancer:
- T-DXd followed by paclitaxel, trastuzumab, and pertuzumab (T-DXd-THP)
- Dose-dense doxorubicin/cyclophosphamide followed by THP (ddAC-THP)
The current analysis focuses on residual cancer burden outcomes following neoadjuvant therapy.
Results
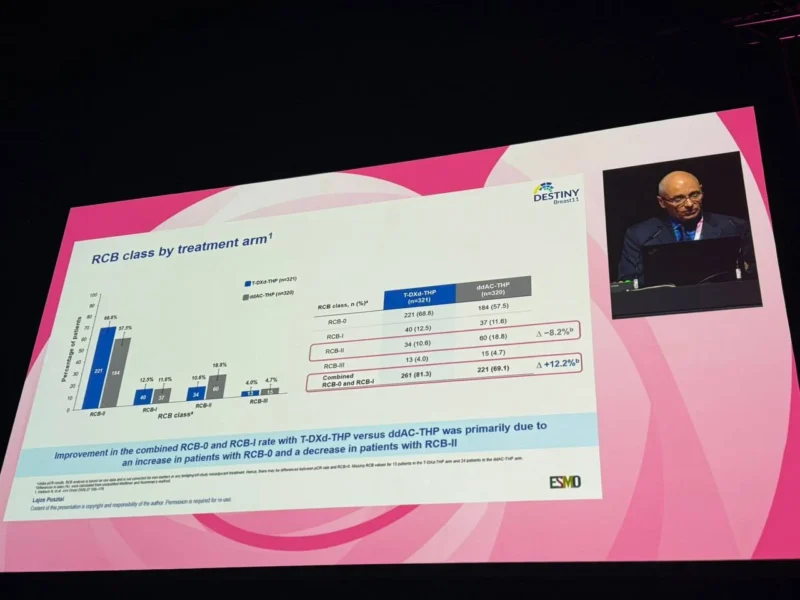
RCB-0+I rates were higher with T-DXd-THP than with ddAC-THP across all evaluated populations.
Overall, 261 of 321 patients (81.3%) in the T-DXd-THP arm achieved RCB-0+I, compared with 221 of 320 patients (69.1%) in the ddAC-THP arm, corresponding to an absolute difference of 12.2%.
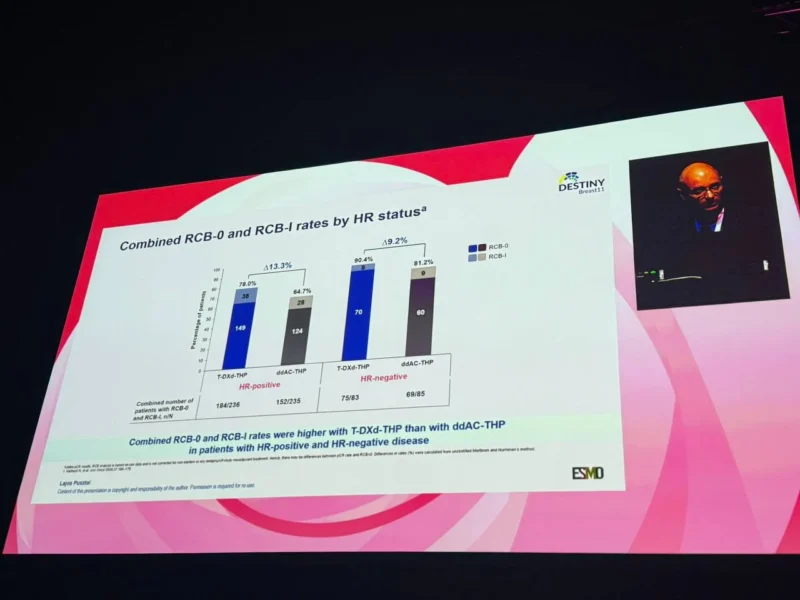
In patients with hormone receptor-positive disease, RCB-0+I was observed in 78.0% with T-DXd-THP versus 64.7% with ddAC-THP.
Across disease stage, rates remained consistently higher with T-DXd-THP. In Stage II disease, RCB-0+I was 83.6% vs 71.7%, and in Stage III disease, 78.9% vs 66.0%.
Subgroup analyses also showed consistent trends:
- Among patients with HER2 IHC 3+ tumors, RCB-0+I rates were 82.9% vs 74.2%, while in the “other” HER2 subgroup, rates were 72.5% vs 30.6%.
- In nodal-positive disease, rates were 81.5% vs 69.8%, and in nodal-negative disease, 80.8% vs 68.6%
- According to tumor stage, RCB-0+I was 83.5% vs 75.0% for cT0–2 tumors and 78.6% vs 60.6% for cT3–4 tumors.
- Continuous RCB index distributions also favored T-DXd-THP, showing lower residual cancer burden compared with ddAC-THP, with a nominal P value of 0.0059.
Key Findings
T-DXd-THP resulted in higher rates of RCB-0+I compared with ddAC-THP overall and across all evaluated subgroups.The benefit was consistent regardless of hormone receptor status, disease stage, tumor size, or nodal involvement.Continuous RCB index analysis further supported reduced residual disease burden with T-DXd-THP, including in patients who did not achieve pathologic complete response.
Conclusion
The DESTINY-Breast11 RCB analysis further supports the activity of neoadjuvant T-DXd-THP in patients with high-risk HER2-positive early breast cancer. Compared with ddAC-THP, T-DXd-THP achieved a higher pCR rate and a higher combined RCB-0/I rate, with fewer patients remaining in the RCB-II category.
The benefit was observed across both HR-positive and HR-negative subgroups, with combined RCB-0/I rates favoring T-DXd-THP in each group. Overall, these findings reinforce the primary endpoint results of DESTINY-Breast11 and highlight T-DXd-THP as a promising neoadjuvant approach in this high-risk population.


