In hormone receptor-positive, HER2-negative advanced breast cancer, CDK4/6 inhibitors have reshaped first-line treatment. Ribociclib plus endocrine therapy has demonstrated progression-free and overall survival benefits across the MONALEESA program, yet early progression remains a real clinical problem. Up to 30% of patients may progress within the first 6 months of CDK4/6 inhibitor therapy, and reliable biomarkers that identify early resistance are still limited (Hortobagyi et al., 2022; McCartney et al., 2019).
The phase IIIb BioItaLEE trial adds important prospective evidence to this unmet need. Published in Clinical Cancer Research, the study evaluated whether baseline and early dynamic ctDNA changes could predict outcomes in postmenopausal patients with HR+/HER2− advanced breast cancer treated with first-line ribociclib plus letrozole (Bianchini et al., 2026).

Read About MONALEESA Trial on OncoDaily
Why BioItaLEE Matters
Liquid biopsy is already clinically useful in advanced breast cancer for detecting actionable mutations such as PIK3CA and ESR1, but using ctDNA as an early treatment-monitoring tool remains investigational. International recommendations have not yet endorsed ctDNA dynamics for routine response monitoring because more prospective validation is needed (Pascual et al., 2022; Henry et al., 2022).
BioItaLEE is important because it prospectively collected serial ctDNA samples during ribociclib-letrozole therapy and linked early molecular changes with progression-free survival. The study does not yet make ctDNA-guided switching a standard of care, but it strengthens the concept that molecular response may appear far earlier than radiographic response.
How The Study Was Designed
BioItaLEE was a multicenter, open-label, single-arm phase IIIb trial conducted in Italy. The study enrolled 287 postmenopausal women with HR+/HER2− advanced breast cancer receiving first-line ribociclib plus letrozole. Patients received ribociclib 600 mg daily on days 1–21 of each 28-day cycle and letrozole 2.5 mg daily (Bianchini et al., 2026).
The biomarker population included 263 patients with valid baseline ctDNA samples. ctDNA was analyzed at baseline, cycle 1 day 15, cycle 2 day 1, and first imaging. The primary objective was to identify baseline ctDNA alterations, track their evolution during therapy, and evaluate associations with progression-free survival.
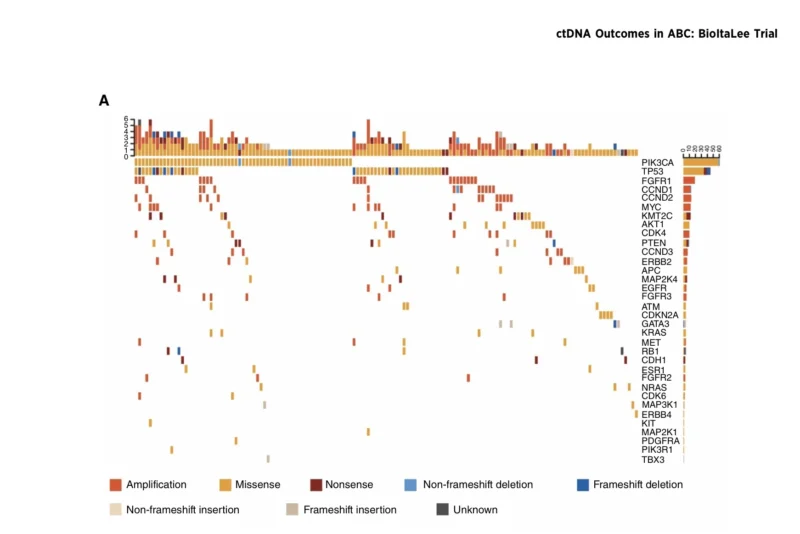
Baseline ctDNA Identified A Higher-Risk Population
At baseline, at least one copy-number alteration or sequence variant was detected in 51% of patients, and more than one alteration was present in 28%. The most frequent baseline alterations were PIK3CA in 22.1% and TP53 in 15.5%. Other recurrent alterations included FGFR1, CCND1, CCND2, KMT2C, MYC, CDK4, AKT1, and PTEN (Bianchini et al., 2026).
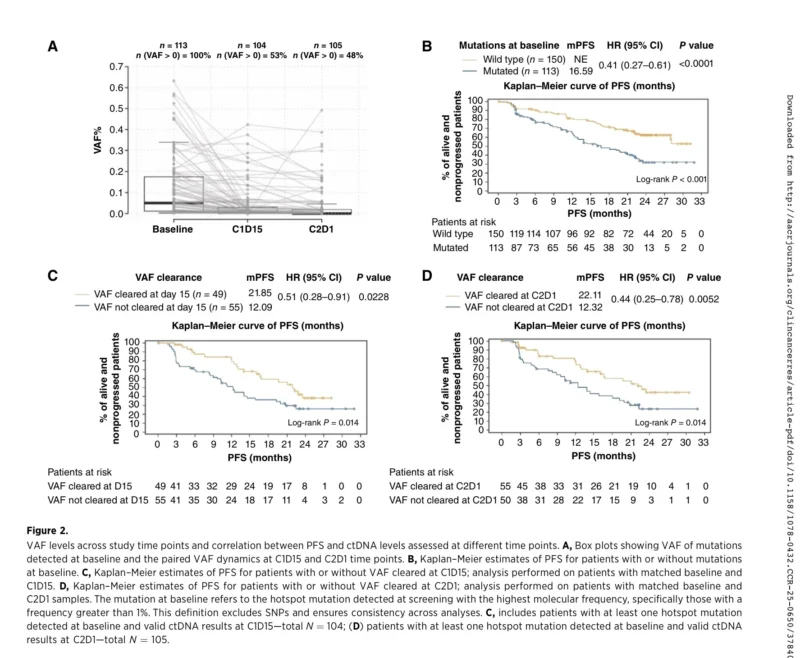
The presence or absence of detectable mutation at baseline was strongly associated with outcome. Patients without detectable baseline mutations had a better prognosis, with a hazard ratio for progression of 0.41 compared with those with detectable mutations. Median PFS was not estimable in patients without detectable baseline mutations versus 16.59 months in those with baseline mutations (Bianchini et al., 2026).
This suggests that baseline ctDNA detectability may reflect tumor burden, biological aggressiveness, or both. It also supports the potential use of baseline ctDNA as a stratification factor in future advanced breast cancer trials.
Early ctDNA Clearance Was Linked To Better PFS
The most clinically interesting finding came from early ctDNA dynamics. Among patients with detectable mutations at baseline, approximately half achieved early mutation clearance after starting ribociclib and letrozole.
At cycle 1 day 15, variant allele frequency clearance occurred in 47.1% of patients with baseline mutations. These patients had improved PFS compared with those without clearance, with a hazard ratio of 0.51. Median PFS was 21.85 months with clearance versus 12.09 months without clearance (Bianchini et al., 2026).
At cycle 2 day 1, clearance was observed in 52.4% of patients and was again associated with improved PFS, with a hazard ratio of 0.44. Median PFS was 22.11 months with clearance versus 12.32 months without clearance (Bianchini et al., 2026).
These results suggest that molecular response can be detected within the first two to four weeks of therapy, long before standard imaging evaluation.
ctDNA At Day 15 May Offer Early Risk Stratification
The investigators also examined whether ctDNA at cycle 1 day 15 alone could provide clinically useful prognostic information. Patients were grouped into those with no detectable mutation, those with low variant allele frequency, and those with high variant allele frequency at day 15.
The three groups had significantly different outcomes. Patients without detectable mutations at day 15 had the most favorable prognosis, while those with high VAF had the worst outcome. Median PFS was not estimable in the non-mutated group, 16.53 months in the low-VAF group, and 11.07 months in the high-VAF group (Bianchini et al., 2026).
This finding supports a more nuanced view of ctDNA response. It may not be enough to classify patients as simply “cleared” or “not cleared.” The amount of residual ctDNA may also carry prognostic meaning.
New Mutations After Baseline Signaled Higher Risk
BioItaLEE also evaluated patients without detectable baseline mutations. Among these patients, 22.7% developed new detectable mutations at early follow-up time points or first imaging. Patients who did not develop new mutations had a lower risk of progression, with a hazard ratio of 0.45 (Bianchini et al., 2026).
This finding is important because it suggests ctDNA may help identify emerging resistance before clinical progression becomes obvious. However, the study was not designed to test whether changing therapy based on ctDNA emergence improves outcomes. That question will require prospective interventional trials.
Resistance Signals: TP53, MYC, HER And CDK4/6 Pathways
Several baseline genomic alterations were linked to more aggressive disease features or early progression. Alterations in TP53, MYC, HER-family pathway genes, and CDK4/6-pathway genes were associated with progression at first imaging. FGFR pathway copy-number gains were more frequent in patients with visceral metastases and those with three or more metastatic sites (Bianchini et al., 2026).
These findings are biologically plausible. TP53 and MYC alterations are associated with endocrine resistance and aggressive luminal breast cancer biology, while alterations affecting HER-family signaling and CDK4/6 pathways may reduce dependence on the endocrine-CDK4/6 axis. Still, these analyses were exploratory and not adjusted for multiple testing, so they should be interpreted as hypothesis-generating.
Clinical Outcomes Were Consistent With Ribociclib Experience
In the intent-to-treat population, median PFS was 23.4 months. At 24 weeks, the objective response rate was 35.9%, and the clinical benefit rate was 72.3%. Among patients with measurable disease, 80.65% experienced tumor shrinkage (Bianchini et al., 2026).
Safety was broadly consistent with previous ribociclib-letrozole experience. Ribociclib discontinuation due to adverse events occurred in 15.7% of patients, and severe adverse events were reported in 6.3%. Three fatal serious adverse events were considered possibly related to ribociclib, including respiratory failure, pneumonia, and undetermined death (Bianchini et al., 2026).
What The Findings Mean For Practice
BioItaLEE supports the idea that ctDNA can provide three clinically relevant layers of information in HR+/HER2− advanced breast cancer: baseline molecular risk, early molecular response, and emergence of new alterations during therapy.
The most immediate clinical implication is not that treatment should be changed based on ctDNA dynamics today. The trial was single-arm, had no endocrine-only or placebo control group, and cannot fully separate prognostic from predictive effects. Instead, the key message is that early ctDNA dynamics are promising biomarkers that deserve prospective validation.
If validated, ctDNA clearance or persistence could eventually help identify patients who are doing well on ribociclib-letrozole and those who may need earlier treatment adaptation. This concept aligns with broader efforts in breast oncology to use liquid biopsy not only to identify actionable mutations at progression, but also to monitor response and resistance during treatment.
Key Takeaway
BioItaLEE shows that baseline ctDNA and early ctDNA dynamics are strongly associated with outcomes in patients with HR+/HER2− advanced breast cancer treated with first-line ribociclib plus letrozole. Patients without detectable baseline mutations had better prognosis, while early mutation clearance at day 15 or cycle 2 day 1 was associated with longer progression-free survival.
The findings position ctDNA dynamics as a promising early biomarker of treatment benefit, but clinical implementation will require prospective trials showing that ctDNA-guided treatment changes improve patient outcomes.
Read full article here