Breast cancer is often discussed through the lens of tumor biology, driver alterations, and treatment response. But cancer does not arise in a vacuum. It begins inside normal tissue that has already been shaped by age, hormones, immune surveillance, and structural remodeling. That is why the new study, Single-Cell Spatial Atlas of the Aging Human Breast, is so important. Rather than focusing on cancer itself, the investigators asked a more foundational question: what happens to normal breast tissue as women age, and how might those changes influence where and how breast cancer begins? (Gupta et al., 2026).
The answer is far more complex than simple involution. Using imaging mass cytometry to analyze more than 3.3 million cells from 527 women, the investigators show that the aging breast becomes less cellular, less proliferative, more structurally reorganized, and more inflammatory. Lobules decline, fat increases, immune composition changes, and the normal spatial relationships between epithelial, stromal, and immune cells begin to loosen. Menopause appears to be the dominant turning point in this remodeling process, creating a major biologic shift in the late 40s rather than a slow, perfectly linear process across life (Gupta et al., 2026).
This matters for oncology because the local environment in which breast cancer starts is not static. It changes with age. And if tissue context changes, then the biology of the tumors that emerge from that tissue is likely to change as well.
Why Aging Breast Tissue Matters in Breast Cancer
Breast cancer clearly does not behave the same way across age groups. Tumors in younger women often differ from those in older women in phenotype, endocrine sensitivity, and clinical behavior. For decades, researchers have suspected that part of this difference reflects changes in the tissue of origin itself. The classic concept of “breast tissue age” proposed that hormonal exposure helps explain why breast cancer does not follow the simple age-incidence pattern seen in many other tumors (Pike et al., 1983). More recently, sequencing studies have shown that normal breast epithelium steadily accumulates somatic mutations with chronological aging, reinforcing the idea that age is not just a background variable but a biologically active force in the breast (Nishimura et al., 2023).
What has been missing is a detailed spatial understanding of that process. Most prior work has described age-related shifts in epithelial proliferation, hormonal signaling, or mutational burden, but not the full tissue ecosystem. This study addresses that gap by combining single-cell resolution with spatial context, showing not only which cells are present, but also how they are organized and how they interact across the aging breast (Gupta et al., 2026).
© The Author(s) 2026
How the Study Was Built
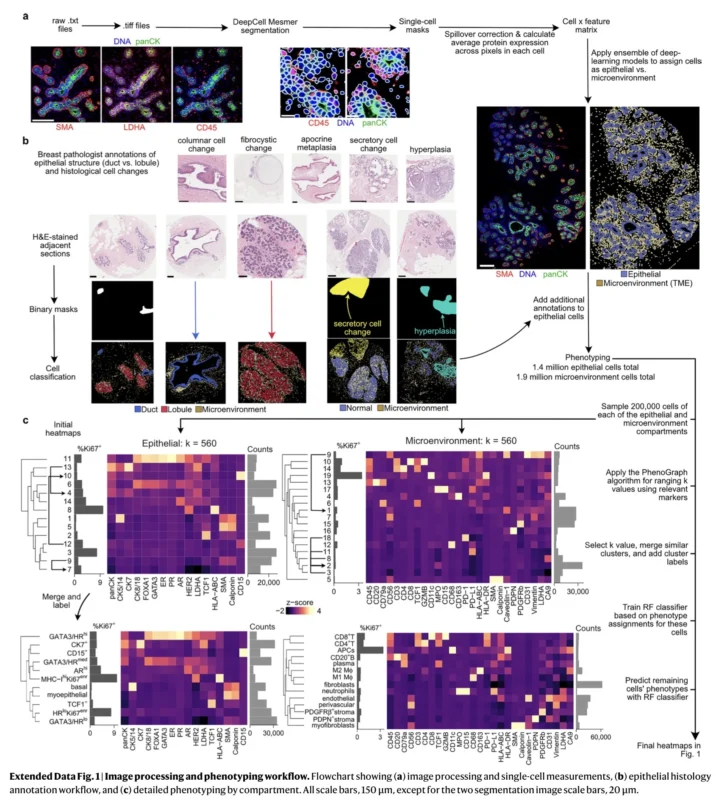
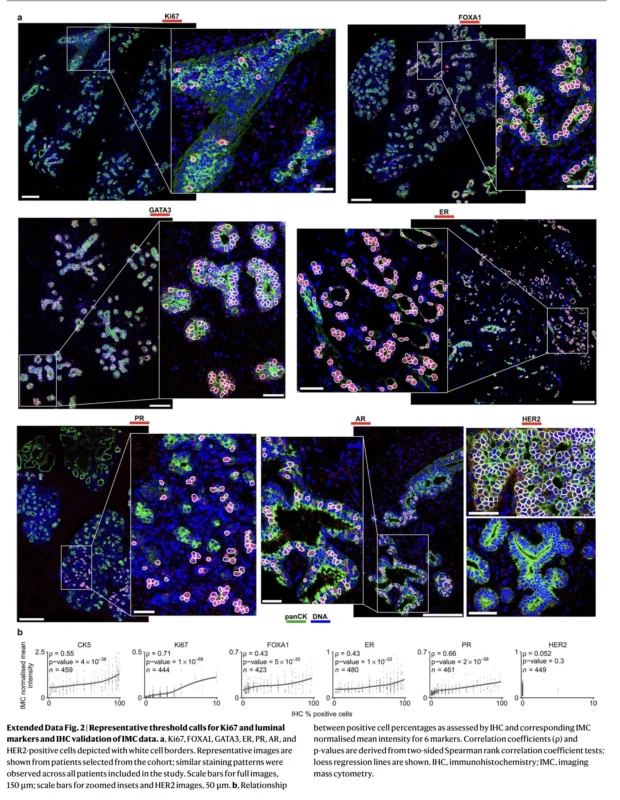
The investigators used tissue from 527 women undergoing reduction mammoplasty or other non-cancer breast procedures, spanning an age range from 15 to 86 years, with a median age of 38. They analyzed 1,710 tissue microarray cores using imaging mass cytometry, a technology that preserves spatial localization while quantifying dozens of proteins at subcellular resolution. Their antibody panel targeted 40 proteins covering epithelial lineage markers, hormone receptors, stromal markers, immune cell markers, immune checkpoints, hypoxia-related proteins, and proliferation markers such as Ki67 (Gupta et al., 2026).
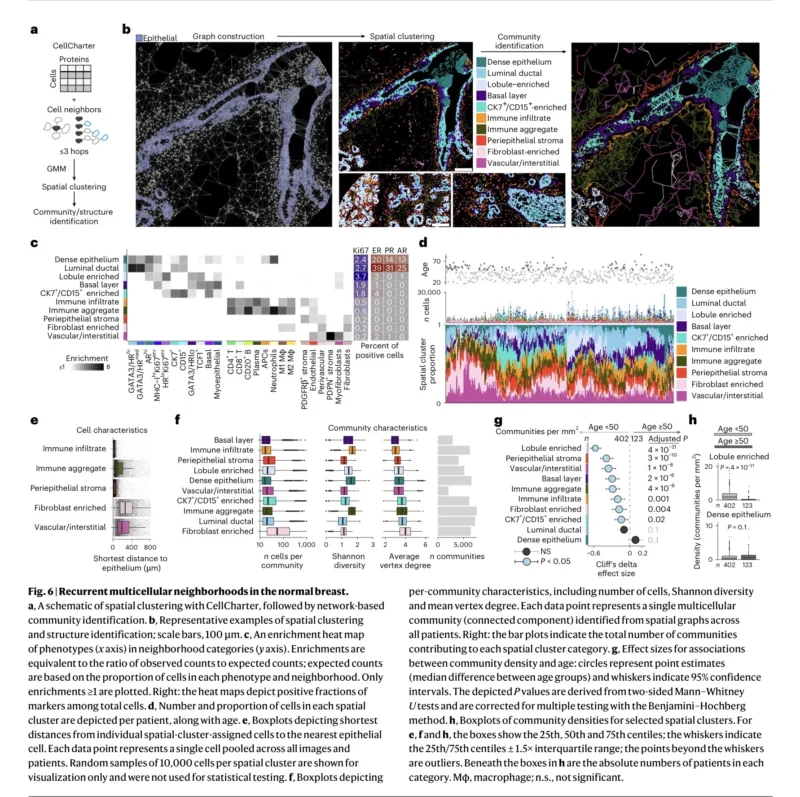
From these data, the team classified cells into 11 epithelial phenotypes and 14 microenvironmental phenotypes, then layered on multiscale analyses of cell density, proliferation, cell shape, cell–cell interactions, multicellular neighborhoods, ducts, lobules, adipose tissue, and vasculature. This is what makes the paper unusually powerful. It is not simply a cell atlas. It is an atlas of aging breast architecture.
The Aging Breast Becomes Less Cellular
One of the clearest messages in the paper is also one of the most striking: aging is associated with a broad decline in breast cellularity. The density of epithelial, stromal, and immune cells all dropped with age, and these declines occurred at comparable rates across the major tissue compartments (Gupta et al., 2026).
That finding suggests aging is not merely a selective loss of one compartment, such as epithelium alone. Instead, it reflects a more general tissue involution. The breast becomes less populated overall. This helps explain why older breast tissue often appears more adipose-rich and less structurally complex, but the study shows that this change is not just histologic. It is measurable at the single-cell level across multiple lineages.
That matters biologically because fewer cells also means fewer opportunities for normal tissue homeostasis to be maintained through cell turnover, signaling, and structural balance.
© The Author(s) 2026
Proliferation Falls Across Nearly Every Cell Type
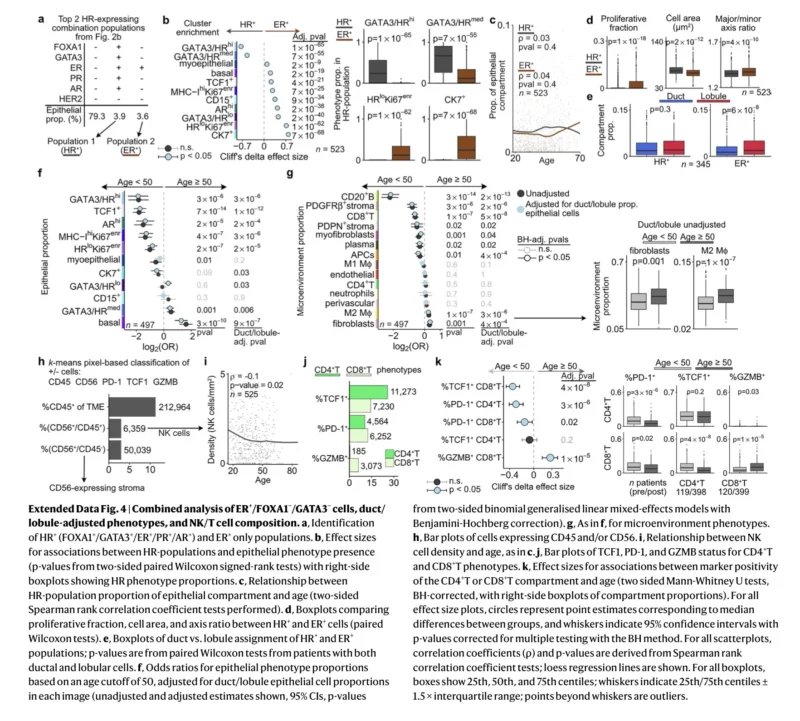
The decline in cell number was accompanied by a parallel decline in proliferation. Ki67-positive fractions fell not just in epithelial cells, where such a change might be expected, but also in stromal and immune compartments. The strongest age-related decrease in proliferation was seen in epithelial cells, but the trend was remarkably broad, affecting nearly every cell phenotype the authors defined, with the exception of neutrophils (Gupta et al., 2026).
This is one of the most important biologic signals in the paper. The aging breast is not simply changing composition. It is becoming more quiescent. The reduction in proliferation likely contributes directly to the decline in cellular density and also reflects a less regenerative tissue state.
At the same time, the study uncovered something more nuanced in hormone receptor biology. Most proliferating epithelial cells were ER-negative in absolute terms, but when the authors analyzed individual cells more closely, they found that the likelihood of proliferation could be higher in ER-positive cells within lobules. That suggests the relationship between estrogen receptor expression and proliferation in normal tissue may be more context-dependent than previously appreciated, especially when examined at very high resolution (Gupta et al., 2026).
The Immune Microenvironment Changes Direction with Age
The paper’s immune findings are especially interesting because they do not show a simple decline. Instead, they show immune remodeling.
Younger breast tissue was strongly enriched for B cells, with additional enrichment of CD8-positive T cells and antigen-presenting cells. Older breast tissue, by contrast, had a more inflammatory and potentially more immunosuppressive composition, with relative enrichment of M2 macrophages and granzyme B–positive T cells (Gupta et al., 2026).
This shift is important because the immune microenvironment helps determine whether altered epithelial cells are ignored, contained, or eliminated. A tissue with fewer resident immune cells near the epithelium, fewer epithelial–immune interactions, and more M2-type macrophages may be less effective at suppressing early neoplastic growth. The study does not claim to prove that this directly causes cancer, but it strongly supports the idea that the aged breast may become more permissive to carcinogenesis through weakening local immune surveillance and altering inflammatory tone (Fane & Weeraratna, 2019; Gupta et al., 2026).
© The Author(s) 2026
Cell–Cell Interactions Become Less Structured
A major strength of spatial proteomics is the ability to study not just cell identity, but cell relationships. Here, the authors show that heterotypic interactions decline with age, especially those between epithelial cells and surrounding stromal or immune cells. In contrast, homotypic epithelial–epithelial interactions were relatively stable (Gupta et al., 2026).
That means the epithelial compartment becomes more spatially insulated from the surrounding tissue as aging progresses. This is a subtle but important finding. Normal epithelial behavior depends on being embedded within a regulated niche. As those epithelial–microenvironment interactions weaken, one can imagine how early abnormal clones may become less constrained.
The study also showed that several microenvironmental cell types, including endothelial cells, fibroblasts, M2 macrophages, and B cells, were located farther from the epithelium in older tissues. No major microenvironmental phenotype moved closer to the epithelium with age. The spatial message is clear: the older breast is a tissue in which epithelial cells are less tightly watched and less tightly supported (Gupta et al., 2026).
Menopause Is the Major Biological Turning Point
One of the most compelling aspects of the study is its analysis of nonlinear aging. Rather than assuming tissue change progresses evenly across life, the authors used sliding-window analysis to identify the ages at which tissue features were changing most strongly. The result was clear: the breast showed a dominant peak of remodeling in the late 40s, corresponding closely to menopause (Gupta et al., 2026).
This is not a trivial observation. It suggests that breast tissue aging is driven less by recurring age-related “waves” and more by one dominant physiologic transition. Hormonal withdrawal around menopause appears to trigger a marked restructuring of the breast, affecting epithelial organization, immune composition, fat content, vessel density, and multicellular neighborhoods.
This also helps explain why breast tissue aging may differ from circulating plasma aging patterns or microbiome-based aging models. The breast is a hormone-sensitive organ, and its aging trajectory seems to be dominated by endocrine transition.
© The Author(s) 2026
Lobules Decline, Fat Expands, and Tissue Architecture Changes
At the tissue level, the structural findings are especially relevant for breast cancer biology. The study showed a marked reduction in lobule density with age and a corresponding increase in duct density and adipose content. Lobules fell sharply around menopausal age, while adipocytes became larger and occupied more of the total tissue area (Gupta et al., 2026).
This is highly relevant because many breast cancers arise in terminal ductal-lobular units. If lobules become depleted and the surrounding microenvironment becomes more adipose-rich and less vascularized, then the physical context of tumor initiation changes substantially. The study also found that blood and lymphatic vessel area declined with age, adding another layer to the structural simplification of the tissue.
Taken together, these observations suggest that aging is not just about cell loss. It is about architectural conversion. The breast becomes a different organ in later life than it was in early adulthood.
Hormone-Related Cells Increase with Age
Another notable finding was the gradual increase in epithelial cells positive for ER, AR, FOXA1, and GATA3 with age. That does not mean the tissue becomes more proliferative. In fact, the opposite is true. These hormone-related cells appear to accumulate in a more differentiated, less proliferative environment (Gupta et al., 2026).
This may help explain, at least in part, why hormone receptor–positive breast cancers are more common in older women. The tissue of origin itself is shifting toward a more hormone-linked epithelial landscape. The study does not argue that this alone determines tumor subtype, but it provides a biologically plausible framework for why age and tumor phenotype are linked.
Why This Study Matters for Breast Cancer Research
This paper is valuable not just because it is large, but because it reframes aging as a multiscale tissue process. Aging in the breast is not only mutation accumulation. It is also a coordinated remodeling of epithelial differentiation, immune composition, stromal morphology, multicellular organization, and tissue structure.
That matters for breast cancer risk. A mutated epithelial cell in a young, highly interactive, B-cell-rich, lobule-dense tissue environment is likely experiencing very different constraints from a similarly altered epithelial cell in an older, less cellular, more inflammatory, more adipose-rich breast.
That is a major conceptual point. Cancer risk is not only about what happens inside the cell. It is also about what happens around it.
© The Author(s) 2026
The Bottom Line
The single-cell spatial atlas of the aging human breast shows that normal breast tissue undergoes a profound age-related reorganization. With age, the breast becomes less cellular, less proliferative, less spatially integrated, more inflammatory, and more structurally simplified. Lobules decline, adipose tissue expands, immune composition shifts, and menopause emerges as the central turning point in this remodeling process (Gupta et al., 2026).
For breast oncology, this study matters because it gives substance to an idea long suspected but rarely mapped in such detail: the aging breast is not just older tissue. It is biologically different tissue. And that changing tissue context may be one of the keys to understanding why breast cancer risk, subtype, and behavior vary so strongly with age.
You can read full article here