Breast cancer in young women continues to represent one of the most complex challenges in oncology. Compared with older populations, these patients often present with more aggressive disease, distinct tumor biology, and a higher likelihood of hereditary predisposition. Among these, germline mutations in BRCA1 and BRCA2 remain the most clinically significant drivers, shaping both cancer risk and therapeutic decision-making (Foulkes, Smith, & Reis-Filho, 2010).
However, a critical question has persisted: do all BRCA mutations carry the same biological and clinical consequences? While BRCA mutation status has long been treated as a binary variable, emerging evidence suggests that this approach may oversimplify a far more nuanced reality. Differences in mutation type, functional impact, and genomic location may influence tumor behavior, treatment response, and survival (Rebbeck et al., 2015).
A large international analysis from the BRCA BCY Collaboration, recently published in Annals of Oncology, provides important insight into this question (Lambertini et al., 2026). By analyzing outcomes in over 3,000 young women with BRCA-mutated breast cancer, the study moves beyond mutation presence and begins to define which mutations matter most, and why.
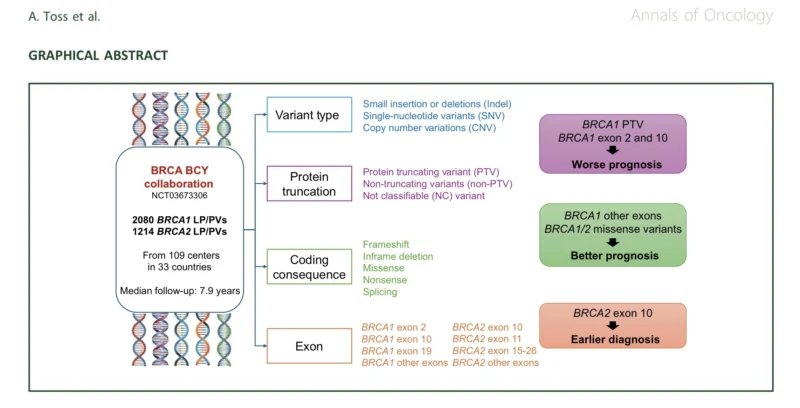
A Global Cohort With Long-Term Follow-Up
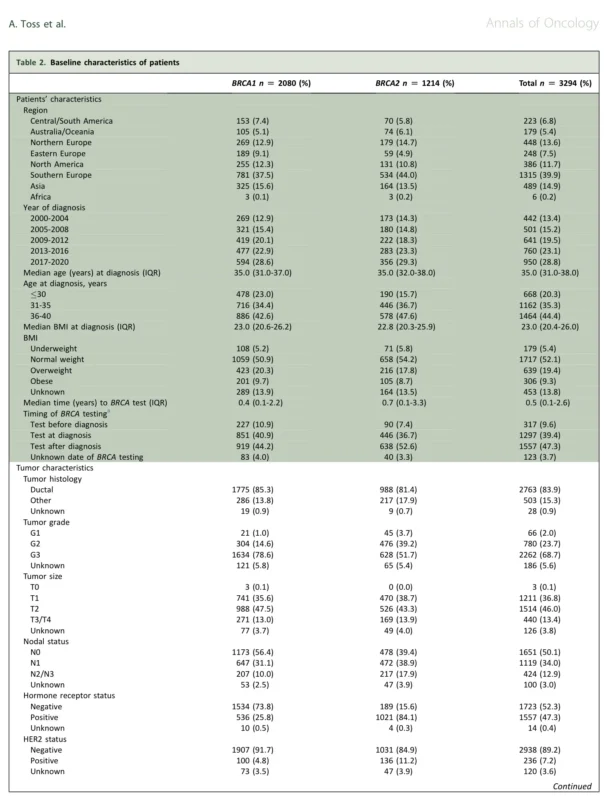
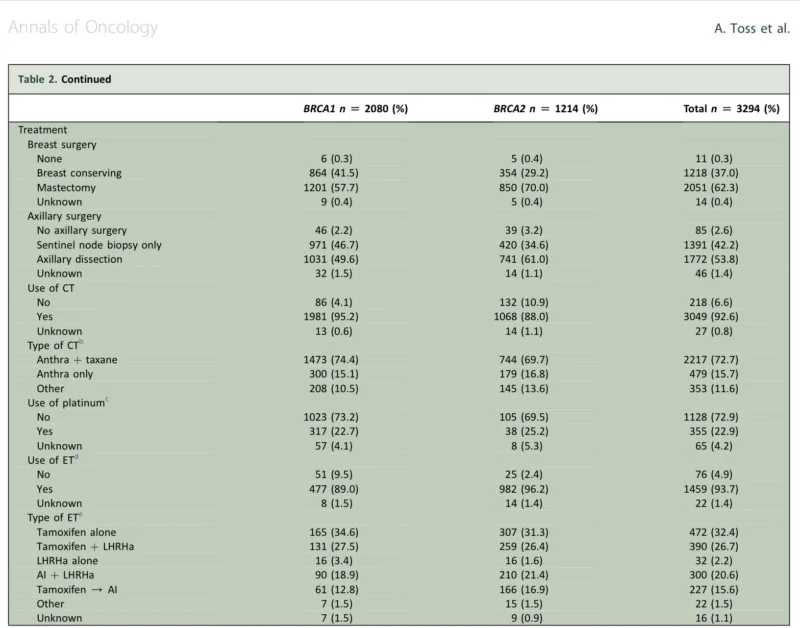
This study represents one of the largest and most comprehensive efforts to characterize outcomes in young BRCA mutation carriers. A total of 3,294 women aged 40 years or younger were included, drawn from 109 centers across 33 countries. The global nature of the cohort enhances the generalizability of the findings and reflects real-world diversity in clinical practice (Lambertini et al., 2026).
With a median follow-up of 7.9 years, the analysis captures long-term survival patterns that are often missing from smaller or shorter studies. The cohort was predominantly composed of BRCA1 mutation carriers, consistent with the well-established association between BRCA1 and early-onset disease (Foulkes et al., 2010).
Crucially, the study did not stop at identifying mutation carriers. Instead, it stratified variants based on their molecular characteristics, including structural type, predicted protein impact, and precise genomic location, allowing for a deeper understanding of genotype–phenotype relationships (Rebbeck et al., 2015).
When Mutation Type Does Not Matter
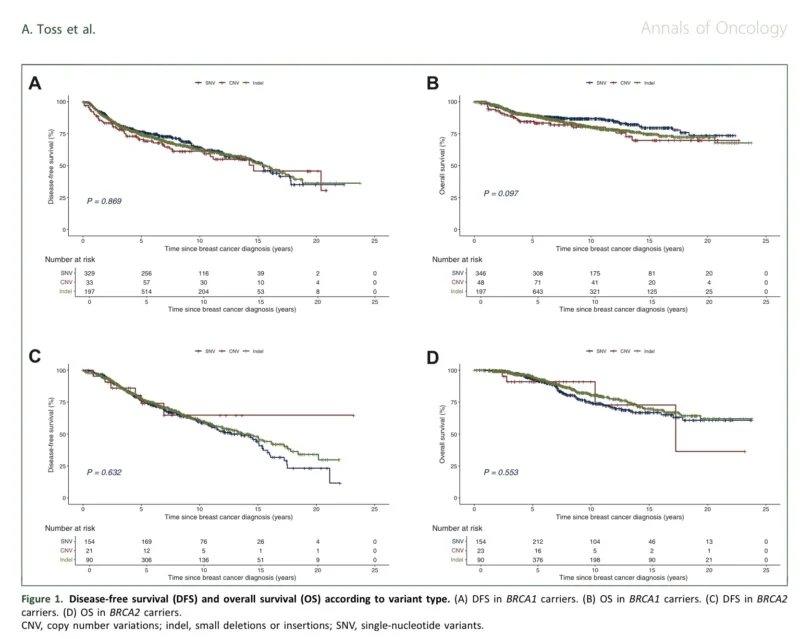
One of the most important findings of the study is that variant type alone does not predict survival. Whether a mutation was an insertion, deletion, single nucleotide variant, or copy number alteration had no meaningful impact on overall survival or disease-free survival (Lambertini et al., 2026).
This finding challenges a common assumption in clinical genetics, where mutation categories are often used as proxies for biological impact. Instead, the data suggest that structural classification alone is insufficient and that functional consequences must be considered to understand prognosis (Rebbeck et al., 2015).
In practical terms, this means that two mutations with different molecular structures may result in similar clinical outcomes, while mutations of the same type may behave very differently depending on their biological effect.
The Impact of Protein Function: Truncating vs Missense
A clearer and clinically meaningful pattern emerged when mutations were analyzed according to their effect on protein function. In BRCA1 mutation carriers, protein-truncating variants were associated with significantly worse overall survival, with approximately a two-fold increase in mortality risk (Lambertini et al., 2026).
This observation is biologically plausible. Truncating mutations lead to complete loss of BRCA1 function, impairing homologous recombination repair and increasing genomic instability, a hallmark of aggressive tumor behavior (Lord & Ashworth, 2016).
In contrast, missense variants were associated with more favorable survival outcomes in both BRCA1 and BRCA2 carriers. These mutations often preserve partial protein activity, which may reduce the degree of genomic instability and limit tumor aggressiveness (Rebbeck et al., 2015).
The effect was particularly pronounced in BRCA2 mutation carriers, highlighting important biological differences between BRCA1- and BRCA2-driven cancers (Foulkes et al., 2010).
Genomic Location: A Critical but Underrecognized Factor
Beyond mutation type and function, the study demonstrated that variant location within BRCA genes significantly influences outcomes.
In BRCA1, mutations located in exons 2 and 10 were associated with worse overall survival, suggesting that these regions may encode critical functional domains of the protein (Lambertini et al., 2026). Conversely, mutations in other regions appeared to be associated with more favorable outcomes.
In BRCA2, mutations in exon 10 were linked to earlier disease onset, while variants in exons 15 through 26 were associated with worse disease-free survival. These findings align with previous research indicating that specific regions of BRCA genes may differentially affect DNA repair function and genomic stability (Rebbeck et al., 2015).
Together, these observations highlight that the genomic context of a mutation is a key determinant of its clinical impact, adding an important layer of complexity to hereditary breast cancer.
Clinical Interpretation: Moving Toward Precision Stratification
The implications of these findings extend beyond academic interest and have direct clinical relevance. Traditionally, BRCA mutations have been treated as a binary marker, guiding decisions related to surgery, systemic therapy, and genetic counseling. However, this study suggests that such an approach may overlook important biological heterogeneity.
Incorporating variant-level information into clinical practice could improve risk stratification and allow for more personalized treatment approaches. For example, patients with truncating BRCA1 mutations may require closer monitoring or more aggressive treatment strategies, while those with missense variants may have a more favorable prognosis (Lambertini et al., 2026).
This shift reflects a broader trend in oncology, where precision medicine is evolving from mutation detection to functional interpretation.
Implications for Targeted Therapy
The findings also raise important questions regarding treatment response. BRCA mutations are central to the use of PARP inhibitors, which exploit defects in DNA repair pathways. It is plausible that the degree of functional impairment associated with different mutations may influence sensitivity to these therapies (Lord & Ashworth, 2016).
Although this study did not directly evaluate treatment outcomes, it provides a strong rationale for future research exploring whether variant-specific differences impact response to targeted therapies. Such insights could further refine treatment selection and improve outcomes.
Conclusion
The BRCA BCY Collaboration study provides compelling evidence that not all BRCA mutations are clinically equivalent. While mutation type alone does not appear to influence survival, protein function and genomic location emerge as critical determinants of outcome (Lambertini et al., 2026).
These findings challenge the traditional binary view of BRCA mutations and emphasize the importance of understanding their biological consequences. As oncology moves toward increasingly personalized approaches, integrating variant-level detail into clinical decision-making will be essential.
Ultimately, this study reinforces a central message in modern cancer research: precision oncology requires not only identifying mutations, but understanding what they do.


