The first week of April reflects the accelerating pace of progress in GI Oncology, bringing together new clinical data, translational insights, and evolving treatment strategies across colorectal, pancreatic, hepatobiliary, and esophageal cancers.
This week’s highlights capture the growing complexity of modern cancer care—from surgical innovation and organ preservation approaches to the expanding role of immunotherapy, targeted treatments, and biomarker-driven decision-making. At the same time, emerging data continue to challenge established assumptions, particularly regarding surrogate endpoints, treatment sequencing, and the interpretation of response across different disease settings.
Notably, advances in global molecular profiling, early-phase clinical development, and large-scale randomized trials are reshaping how clinicians approach patient selection and therapeutic strategies. Alongside these developments, new approvals and guideline updates further emphasize the importance of timely access to effective, personalized treatments.
Together, these perspectives underscore a field in transition, where multidisciplinary collaboration, precision oncology, and critical clinical judgment remain central to improving outcomes for patients with gastrointestinal cancers.
Riziero Esposito Abate – Healthy Researcher at IRCCS Istituto Nazionale Tumori Fondazione G. Pascale, Napoli, Italy
“I am pleased to share our latest publication on the results of the NIVACOR phase II trial recently published in Nature Communications.
Great team effort and cooperation!”
Laura Moolenaar, MD, PhD – Candidate in Colorectal Surgery
“Very pleased to share that the short-term outcomes of the COLOR III trial have been published in The Lancet Gastroenterology & Hepatology!
The COLOR III trial is the first international multicenter randomised controlled trial with stringent quality control to compare transabdominal total mesorectal excision (TaTME) with laparoscopic TME (lapTME) in patients with (y)cT1–3N0–2M0 rectal adenocarcinoma located within 10 cm from the anal verge. In total, 1103 patients across 28 hospitals globally were randomised.
𝗞𝗲𝘆 𝗳𝗶𝗻𝗱𝗶𝗻𝗴𝘀
TaTME significantly reduced conversion rates compared with lapTME (1% vs 17%; adjusted OR, 0.03 [95% CI, 0.01 to 0.08])
TaTME achieved similar rates of 90-day morbidity (20% vs 18%) and complete resection rates (84% vs 86%) compared with LapTME
Independent predictors for conversion were male sex, BMI >30 kg/m2, and tumours located within 5 cm of the anal verge
𝗜𝗺𝗽𝗼𝗿𝘁𝗮𝗻𝗰𝗲
Collectively, these data demonstrate that TaTME is a safe surgical technique and a valuable addition to the surgical management of this patient population when performed by experienced surgeons with proven quality standards. Long-term follow-up with mature OS and DFS data is necessary to adequately assess functional outcomes and the oncological safety of TaTME.”
Arndt Vogel – Head of the Center for Personalized Medicine, MHH at Medical University of Hanover
“off the press: EMERALD-3 Phase III trial positive.
Significant PFS benefit for STRIDE/Lenva + TACE, trend of OS benefit
Trend for PFS benefit for STRIDE + TACE
Looking forward to see more details”
Tiago Cordeiro Felismino – Chief of Upper Gastrointestinal Oncology at A.C.Camargo Cancer Center, Brazil
“Sharing our recent publication on borderline resectable pancreatic cancer:
Neoadjuvant radiotherapy added to modern chemotherapy did not improve resection, margins, or survival.
Systemic therapy remains the cornerstone, with surgery as the key determinant of outcomes.”
Sepideh Gholami – Surgical Director of the Liver Multidisciplinary Clinic and Director of Hepatic Artery Infusion Pump and Translational Research in Surgical Oncology at Northwell Health / Cold Spring Harbor Laboratory
“Excited to share our recent work on CXCR4 in metastatic colorectal cancer.
Across >15,000 samples, higher CXCR4 expression was linked to distinct immune features (CXCR4 mRNA expression was positively associated with high TMB, dMMR and positive PD-L1 status), worse outcomes in primary tumors, but improved survival in metastatic disease—especially with immunotherapy.A reminder that biomarkers are context-dependent, and the liver/metastatic setting changes the story.”
Michael Sapienza – Chief Executive Officer at the Colorectal Cancer Alliance
“The FDA’s full approval of a BRAF V600E targeted combination therapy for metastatic colorectal cancer is an important step forward for patients.
For a long time, individuals with this mutation have faced some of the most aggressive disease courses and the fewest effective first line options. This approval represents clinical progress, but it’s also a reminder that we need to invest more and move faster to continue to make a real impact on survival and quality of life for those battling this disease.Scientific breakthroughs matter most when they reach patients in time. Approvals like this show what is possible when the system moves with both rigor and urgency.
At the Colorectal Cancer Alliance, we see every day how urgent progress is needed. Patients need smarter, more targeted approaches from the very beginning of their care and they need it now. The Alliance’s Project Cure CRC initiative is working to accelerate treatments and deliver personalized therapies to patients at a faster pace so we can end this disease.
No one should face this disease alone or without better options.”
Lawrence Kwong – Associate Professor at MD Anderson Cancer Center
“Our paper summarizing global patterns of biliary tract cancer mutational patterns is now online at the Journal of Hepatology. We hope that this effort from our 3 co-first authors Yan, Jason (an undergraduate!), and Zhang will help guide both preclinical modeling and geographically-aware clinical trial designs.”
Federico Nichetti – MD, Medical Oncologist, Italy
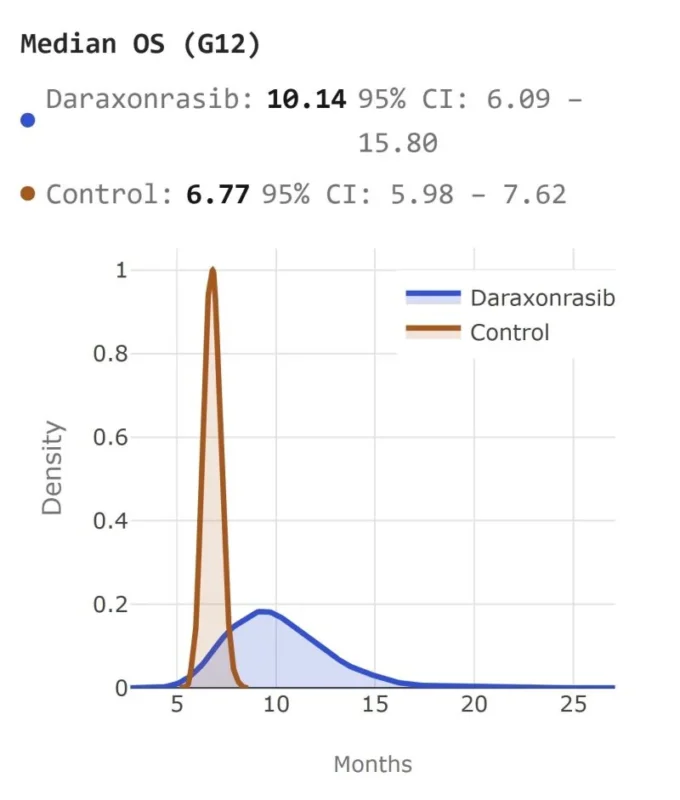
“Warpspeed.sh ran an AI-based simulation of RASolute-302, predicting that daraxonrasib could demonstrate an advantage both in PFS and OS (>10 months!!) vs chemotherapy in pretreated patients with metastatic pancreatic cancer.
Will this be the real result?”
Georgios Efthymiou – Postdoctoral Researcher at INSERM, France
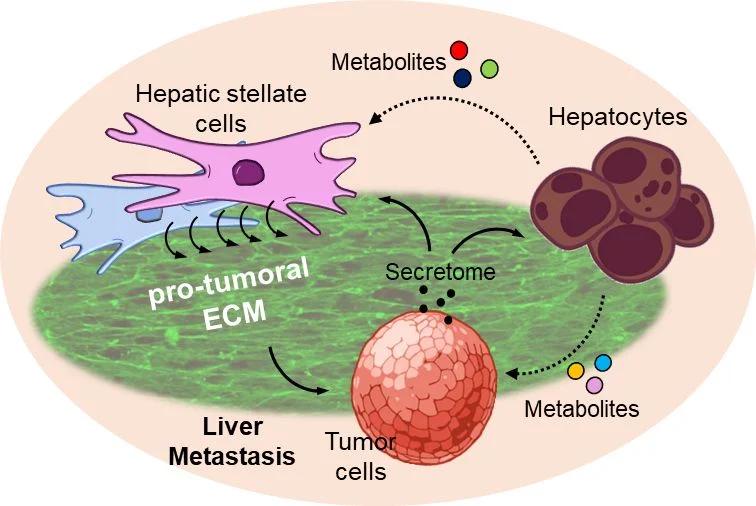
“I am so excited to have been awarded the Young Investigator Grant from the French Research Network against Pancreatic Adenocarcinoma (FRAP) to kickstart my research on pancreatic cancer metastasis in the liver, a determinant of patient outcomes.
With the invaluable support from FRAP, I am setting out to explore communication circuits between metastatic tumor and resident liver cells. By targeting metabolic reprogramming and extracellular matrix remodeling, key drivers of metastatic expansion in the liver, my work aims to limit metastatic growth and spread. This will increase the number of eligible patients for surgical resection, improving patient survival.
Many thanks to the FRAP Network – Pancreatic Cancer for this honor, my host team DISARM-PDAC and my ever-supporting mentors Sophie Vasseur and richard tomasini, my host institute CRCM – Centre de Recherche en Cancérologie de Marseille and everyone involved directly or indirectly in this project!”
Thaís Megid – Medical Oncologist at Dr. Leon Richard Cancer Centre, New Brunswick, Canada, and Assistant Professor at Université de Sherbrooke, Quebec
“Organ preservation strategy in esophageal cancer aims to avoid esophagectomy and better quality of life. The SANO trial was able to demonstrate non inferiority results with this approach in patients who achieve clinical complete response.
On the other hand, we know that esophageal squamous cell carcinoma histology presents with a different biology than adenocarcinoma. Can we really trust that complete response is a surrogate endpoint for survival in adenocarcinoma patients?
We did a meta analyses including more than 7000 patients with Gastroesophageal adenocarcinoma who received neoadjuvant therapies.
Pathological complete response was a WEAK surrogate endpoint for survival in this population.
We need to be careful when offering “watch and wait” strategy for this population.Hopefully, in the era of immunotherapy and precision medicine, pCR will be stronger surrogate EP for DFS and OS in clinical trials”
Find out 10 Must-Read Posts in GI Oncology from the last week of March on OncoDaily.