The European Lung Cancer Congress (ELCC 2026), taking place in Copenhagen, Denmark, and online from 25–28 March 2026, is a major multidisciplinary meeting dedicated to thoracic oncology. Bringing together specialists from across the field, the congress serves as a platform for the presentation of new scientific data, educational exchange, and discussion aimed at improving the care of patients with lung cancer worldwide.
Here are 25 insightful posts you shouldn’t miss from leading experts highlighting key research findings, clinical advances, and important discussions shared during ELCC 2026.
25 Posts Not To Miss From ELCC 2026
ESMO – European Society for Medical Oncology:
“As discussed by Umberto Malapelle, multimodal strategies combining clinical parameters, molecular hallmarks and radiomics signatures are increasingly converging.
In an Opinion piece in the hashtag ESMO Daily Reporter, he explains how presentations at ELCC26 have sought to incorporate multimodal insights into early detection, progression prediction and rehabilitation.
Read the full article here https://ow.ly/osGu50Yz7EO”

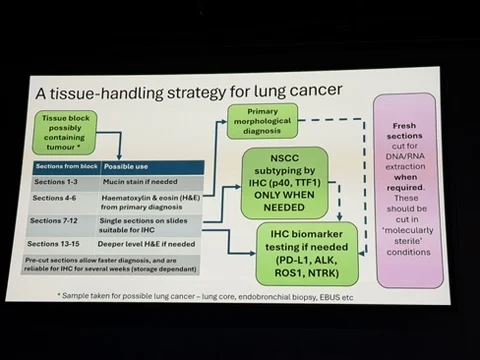
“Prof Keith Kerr of University of Aberdeen provided an extremely informative overview on how an integrated biomarker strategy is required for efficent and effective selection of treatment in the era of novel therapeutics in lung cancer.”
ESMO – European Society for Medical Oncology:
“Congratulations to the Merit Award recipients at ELCC26
Through the ELCCMeritAwards ESMO and International Association for the Study of Lung Cancer support young researchers by recognising the quality of their work.
These awards strengthen the next generation of oncology professionals.

“A great section at the ELCC 2026 in Copenhagen on predictive IHC biomarkers for ADCs in lung cancer co-chaired by Umberto Malapelle and Fernando Lopez-Rios. Very inspiring talks on emerging biomarkers and digital pathology.”

“Fantastic to see the Accord team making an impact at ELCC 2026! It’s inspiring to witness the exchange of knowledge and innovation in thoracic oncology. Proud to be a strategic partner in Turkey and helping bring innovative oncology treatments to patients who need them most. Looking forward to the updates from Copenhagen!”
ESMO – European Society for Medical Oncology:
“ELCC26: Recipient of the 2026 Heine H. Hansen Award, Filippo de Marinis, sadly passed away in February. In an interview in the ESMO Daily Reporter, Antonio Passaro describes his mentor’s long-lasting legacy, discussing de Marinis’ considerable contributions in shaping not only Lung Cancer treatment, but also the Thoracic Oncology community.
Read the full interview here https://ow.ly/uiYH50YyAPm”

“Excited to present our ELCC poster on developing a machine‑learning risk prediction model for NSCLC based on large‑scale real‑world EMR data.
This work illustrates the potential of AI to enhance risk stratification and early detection, complementing existing lung cancer screening paradigms.”
ESMO – European Society for Medical Oncology:
“ELCC26: Results from the ETOP ADEPPT Clinical Trial demonstrate the benefits of adagrasib in older patients but indicate that caution is needed in patients with poor performance status. Despite making up a large proportion of the real-world population, these patient subgroups are often underrepresented in trials.
Read more about the latest insights in KRAS G12C-mutated NSCLC in the ESMO Daily Reporter https://ow.ly/SYQp50Yzf3R “
“𝗬𝗲𝘀𝘁𝗲𝗿𝗱𝗮𝘆, 𝗜 𝗿𝗲𝗰𝗼𝗿𝗱𝗲𝗱 𝗮 𝘃𝗶𝗱𝗲𝗼 𝘀𝗵𝗼𝘄𝗶𝗻𝗴 𝗵𝗼𝘄 𝘁𝗼 𝗴𝗲𝗻𝗲𝗿𝗮𝘁𝗲 𝗮 𝗳𝘂𝗹𝗹 𝗦𝗟𝗥 𝗶𝗻 𝘂𝗻𝗱𝗲𝗿 𝗮 𝗺𝗶𝗻𝘂𝘁𝗲 𝗶𝗻 𝗡𝗦𝗖𝗟𝗖.
Today… it’s already outdated.
𝗧𝗵𝗶𝘀 𝘄𝗲𝗲𝗸’𝘀 𝗘𝗟𝗖𝗖 𝟮𝟬𝟮𝟲 𝗿𝗲𝗹𝗲𝗮𝘀𝗲𝗱 𝟰𝟵𝟯 𝗮𝗯𝘀𝘁𝗿𝗮𝗰𝘁𝘀 – we already reviewed!
• 64 records for 𝟱𝟰 𝘀𝘁𝘂𝗱𝗶𝗲𝘀 𝗶𝗻𝗰𝗹𝘂𝗱𝗲𝗱 𝗶𝗻 𝗦𝗟𝗥
• 𝟯𝟱 𝗻𝗲𝘄 𝘀𝘁𝘂𝗱𝗶𝗲𝘀 (including multiple Phase 2/3 trials)
• And 𝟱 𝗹𝗮𝘁𝗲-𝗯𝗿𝗲𝗮𝗸𝗲𝗿𝘀 still coming
That’s how fast oncology evidence moves.
And that’s exactly why static SLRs don’t work anymore.
If you’re an Oncoscope subscriber, nothing breaks.
Because today, you’ll get an alert showing 𝗲𝘅𝗮𝗰𝘁𝗹𝘆 𝘄𝗵𝗶𝗰𝗵 𝗻𝗲𝘄 𝘀𝘁𝘂𝗱𝗶𝗲𝘀 𝗶𝗺𝗽𝗮𝗰𝘁 𝘆𝗼𝘂𝗿 𝘀𝗮𝘃𝗲𝗱 𝗣𝗜𝗖𝗢.
You log in → check your saved PICO→ 𝗿𝗲𝗴𝗲𝗻𝗲𝗿𝗮𝘁𝗲 𝘆𝗼𝘂𝗿 𝗿𝗲𝗽𝗼𝗿𝘁
𝗜𝗻 𝘂𝗻𝗱𝗲𝗿 𝗮 𝗺𝗶𝗻𝘂𝘁𝗲.
No re-running searches.
No starting from scratch.
No waiting weeks.
That’s what 𝗹𝗶𝘃𝗶𝗻𝗴 𝗲𝘃𝗶𝗱𝗲𝗻𝗰𝗲 𝗮𝗰𝘁𝘂𝗮𝗹𝗹𝘆 𝗹𝗼𝗼𝗸𝘀 𝗹𝗶𝗸𝗲 𝗶𝗻 𝗽𝗿𝗮𝗰𝘁𝗶𝗰𝗲.
In oncology, “up-to-date” doesn’t mean last month.
It means today.”

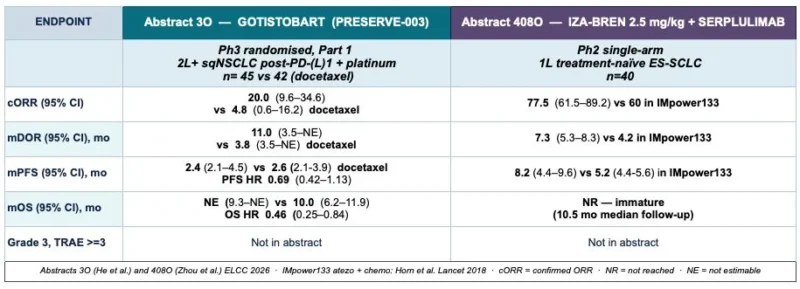
“ELCC 2026 abstracts (new anti-CTLA4) and ADCs (EGFRxHER3 bi & HER3 monospecific)
Gotistobart a Treg depleting anti-CTLA4. Finally pure IO in 2L sqNSCLC?
PRESERVE-003 Stage 1 results (Abstract 3O) are honestly quite striking for a population where we have nothing much to offer after IO failure. Yes the median PFS is similar to docetaxel, 2.4 vs 2.6 months but this is not the right way to look at this data. At 12 months 25% of patients are still progression free vs literally 0% on chemo. Confirmed ORR 20% vs 4.8%, duration of response 11 vs 3.8 months, and OS HR 0.46. So 1 in 5 patients respond but when they respond it lasts. If Stage 2 confirm this, we may finally have a chemotherapy-free ICI option as standard of care in ≥2L squamous NSCLC. Long overdue.
Iza-bren in SCLC. The momentum continues.
Abstract 408O presents first line iza-bren + serplulimab (anti-PD1) in ES-SCLC. At the selected 2.5mg/kg dose (n=40): confirmed ORR 77.5%, mPFS 8.2 months. Compare this to atezolizumab + chemo which is a current standard (ORR 60%, mPFS 5.2 month). Yes this is single arm, Phase 2 from China only, same pitfalls so we need to be careful. But still.
Interesting strategic question here. Pumitamig (BNT327, PD-L1xVEGF bispecific from BioNTech/BMS) also showed Phase 2 SCLC follow-up data at this meeting with mOS 16.9 months vs 12.3 for atezo+chemo (Abstract 426P). Phase 3 enrolling (ROSETTA Lung-01). Iza-bren + pumitamig in SCLC would be a very rational combination but no trial announced yet. Worth watching because BMS has hands in both programs and is starting combo therapies in both NSCLC and TNBC (NCT06618287).
On the ADC side more broadly. BioNTech’s own HER3 ADC YL202/BNT326 will also be presented here (abstract 11MO) with preliminary ORR numerically better than HER3-DXd in EGFRm NSCLC (which had its BLA withdrawn after Phase 3 OS failure). And notably Hengrui SHR-A2009 (another HER3ADC) in combination with bevacizumab showed 59% confirmed ORR with zero ILD in post-TKI/platinum EGFRm NSCLC, suggesting anti-angiogenic combinations may help with both efficacy and the ILD problem of HER3 ADCs (abstract 23B). Pumitamig targets VEGF-A so the question of whether this class could play a similar role is interesting. BioNtech is moving ahead with an heavy BNT326 and pumitamig combo early program in lung cancer (NCT07111520).
Lot of moving pieces. Exciting time for lung cancer.”
“Hot off the press. Just published NEJM. In conjunction with presentation ESMO ELCC26. Results of phase 1 trial of: Setidegrasib in Patients with KRAS/G12D mutant non-small cell Lung Cancer & Pancreas Cancer. Adverse Events (AEs) ≥ 3: 42% Response Rate (RR): 36% in NSCLC 24% in Pancreatic Cancer https://nejm.org/doi/full/10.1056/NEJMoa2600752?query=featured_home “

“Addition of local RT to Osimertinib improves PFS in metastatic NSCLC ( egfr mutated ) Northstar Trial update. ”

“Impressive data in SCLC26 on IZA-BREN ( EGFRxHER3 ADC) with ICI surplulimab in 1L ES-SCLC: ORR 85-92%! across IZABREN doses mDOR =7-8 m Discontinuation due to TRAEs only 7.3% Dawn of a carbo-Etop- free 1L treatment in SCLC?”

“Today presentation ELCC26 phase 1 of Obrixtamig + ezabenlimab in SCLC, LCNEC or other NECs Overall: ORR 30%, DCR 58%, mPFS 4.4m Highest dose: ORR 58%, DCR 83%, mPFS 10m SCLC: ORR 33%, PFS 5.7 m.”

“HER2 loss… may not be the whole story. Data presented at ELCC 2026 suggest that resistance to T-DXd in HER2-mutant NSCLC is unlikely to be explained by a single mechanism. The underlying biology appears more complex, likely involving multiple co-occurring alterations.”

“Phase II ETOP ADEPPT trial: Adagrasib in patients with KRASG12C-mutant NSCLC who elderly or poor performance status, trial met its primary endpoint in elderly, didn’t met thr primary endpoint in PS 2 Dr JNaidoo, GECP, ELCC 26.”

“Beyond traditional ADCs at ELCC26! Le Xiuning Masked ADCs → tumor-activated targeting Nanobodies & peptides: better penetration, new epitopes Bicycle toxin conjugates: ultra-small, fast delivery Innovation across 3 axes: target, linker, payload From ADCs to “anything-drug conjugates”

“Catching up with the ELCC26 excitement for new targeted therapies for KRASG12D (5% of lung cancer) Setidegrasib – first-in-class, PROTAC (Proteolysis Targeting Chimera) protein degrader that targets the KRAS G12D mutation in Advanced NSCLC and Pancreatic Cancer Phase 1 publication N=203 Partial Response (PR) 36% in NSCLC 24% in Pancreatic Cancer Adverse Events (AEs) ≥ 3: 42% (IRRs, nausea)”
“Honored to be delivering two educational lectures at ELCC26 ESMO in Copenhagen. March 26. Beyond traditional ADCs: Advances in targeting, conjugation, payload design, and emerging modalities March 28 Mechanisms of resistance: Lessons from EGFR-mutant and ALK-rearranged NSCLC Grateful to the conference chairs for your excellent work on organizing ELCC26. Looking forward to great discussions and connecting with global colleagues. ”

“Setidegrasib in Adv NSCLC and Pancreatic Cancer NEJM – Ph I KRAS G12D degrader, n=76 RP2D 600mg – NSCLC (n=45): ORR 36%, mPFS 8.3m, 12m OS 59% – PDAC (n=21): ORR 24%, mPFS 3m, mOS 10.3m A new class of targeted therapy enters the fray.”
“FLAURA2 regimen raises the bar! New data from a China study shows impressive PFS/OS gains for Osi + Chemo in EGFRm + TP53 co-mutated pts. Confirms 1L SOC status across the board: TP53 mut, WT, and Asian subgroups…Finding a better future strategy won’t be easy. ”

“Amazing session on stage 3b NSCLC by both Drs Stiles and Chan at ELCC26 wonderfully moderated by Jarushka Naidoo. One thing made clear by all three in this rapidly changing space: “Dogma is just evidence that stopped evolving”- we cannot stay stagnant as a field. Yes- all of our specialties together will need to keep working on the evidence to be able to move the needle even further!”

“Original Article: Setidegrasib in Advanced Non–Small-Cell Lung Cancer and Pancreatic Cancer https://nejm.org/doi/full/10.1056/NEJMoa2600752″
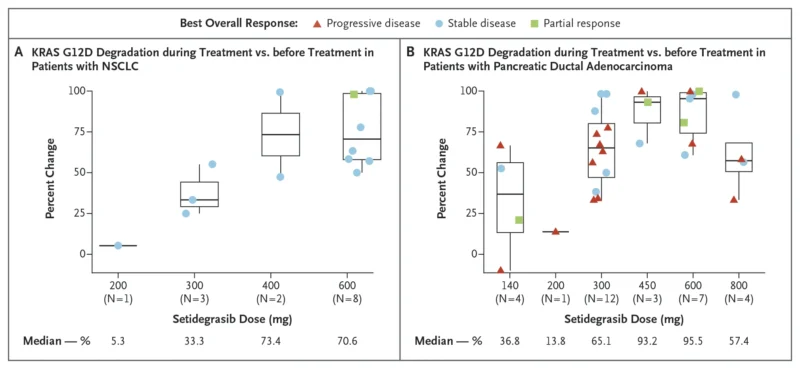
“KRAS G12D – long considered “undruggable.” Now we finally have real clinical signal. Concurrent NEJM publication in ELCC26 Setidegrasib (ASP3082), a KRAS G12D degrader, shows promising activity in heavily pretreated NSCLC. Population 2L+ KRAS G12D NSCLC Most had prior chemo + IO Results (600 mg weekly) ORR: 36% mPFS: 8.3 mo 2L/3L subgroup → ORR 38%, mPFS 11.2 mo Safety Infusion reactions common (80%) but manageable Grade ≥3 TRAEs: 9% Takeaway First meaningful signal in KRAS G12D NSCLC Not practice-changing yet But clearly practice-watching Full paper in comment.”

The start of ELCC26 with Sanjay Popat, Aɴᴛᴏɴɪᴏ Pᴀssᴀʀᴏ beautiful opening remarks around enhancing the care of lung cancer patients and bringing our lung cancer “family” together. Excited to start a great meeting and to connect with colleagues and clinicians!”

Find more exclusive oncology content on OncoDaily.
Written by Nare Hovhannisyan, MD


