VALOR Trial randomized clinical trial designed to compare lung cancer surgery with stereotactic body radiation therapy (SBRT) in patients with operable early-stage non-small cell lung cancer (NSCLC). Because both approaches are potentially curative, ensuring that treatment is delivered exactly according to protocol is essential for patient safety and for the scientific validity of the study. In this report, investigators described the development and early performance of a centralized quality assurance program for the SBRT arm of VALOR, focusing on the first 100 treated participants across the Veterans Health Administration.
The analysis shows how structured credentialing, standardized planning guidance, and centralized plan review helped maintain high compliance with protocol-defined SBRT delivery in a complex multicenter trial.
Title: Centralized Quality Assurance of Stereotactic Body Radiation Therapy for the Veterans Affairs Cooperative Studies Program Study Number 2005: A Phase 3 Randomized Trial of Lung Cancer Surgery or Stereotactic Radiotherapy for Operable Early-Stage Non-Small Cell Lung Cancer (VALOR)
Authors: Timothy A. Ritter, PhD, DABR, Robert D. Timmerman, MD, Hena I. Hanfi, MBA, Hairong Shi, PhD, Matthew K. Leiner, MS, Hua Feng, PhD, MS, BMed, Vicki L. Skinner, RN, MSN, Lisa M. Robin, MA, Cheryl Odle, MBA, Gabriella Amador, MPH, Tom Sindowski, BS, Amanda J. Snodgrass, PharmD, Grant D. Huang, MPH, PhD, Domenic J. Reda, PhD, Christopher Slatore, MD, MS, Catherine R. Sears, MD, Lorraine D. Cornwell, MD, Tomer Z. Karas, MD, FACS, David H. Harpole, MD, Jatinder Palta, PhD, Drew Moghanaki, MD, MPH.
Background
The VALOR trial is a phase 3 randomized study designed to compare lung cancer surgery with stereotactic body radiation therapy (SBRT) for patients with operable early-stage non-small cell lung cancer (NSCLC). In this setting, both treatment arms must be delivered with high technical precision to preserve patient safety and maintain the scientific integrity of the trial. Radiation therapy quality assurance is especially important in multicenter studies because differences in contouring, prescription selection, motion management, treatment planning, and dose delivery can influence both toxicity and clinical outcomes.
This report from Practical Radiation Oncology focuses on the first 100 participants treated with SBRT in VALOR and describes how a centralized quality assurance (QA) system was built and implemented across the Veterans Health Administration. The main goal was to determine whether study sites could consistently deliver protocol-compliant lung SBRT and whether centralized monitoring could detect and correct potentially important deviations before they became recurrent problems.
Methods
The investigators developed a centralized SBRT QA program specifically for the VALOR study. This program was approved by the Veterans Affairs Cooperative Studies Program and overseen by the VA Central Institutional Review Board. Its purpose was to credential participating treatment centers, standardize technical practice, and continuously monitor compliance with protocol-defined SBRT requirements.
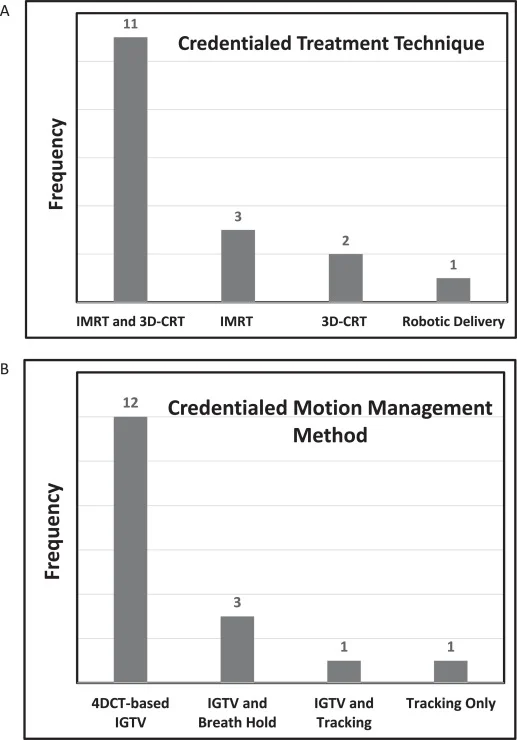
The framework was adapted from the standards used by the National Cancer Institute’s Image and Radiation Oncology Core (IROC), which supports radiotherapy oversight in National Clinical Trials Network studies. Participating VA radiation oncology departments had to demonstrate that they met both general institutional standards and study-specific requirements before enrolling patients to the SBRT arm.
General VA requirements included board-certified radiation oncologists, experienced medical physicists, departmental accreditation, successful completion of IROC end-to-end phantom testing every 3 years, IROC site visit audits, and participation in the VA Radiation Oncology Quality Surveillance Program. For VALOR specifically, each site also had to complete a facility questionnaire, undergo IROC anthropomorphic lung phantom validation, show image-guided radiotherapy credentialing, and submit its first SBRT plan for prospective central review.
The protocol included a detailed SBRT Planning Guideline that covered simulation, immobilization, contouring, structure nomenclature, dose prescription, and dosimetric evaluation. The internal gross target volume (IGTV) had to be expanded by 5 to 7 mm to create the planning target volume (PTV). Acceptable prescriptions for peripheral tumors were 18 Gy × 3 fractions, 14 Gy × 4 fractions, or 11.5 Gy × 5 fractions, while central tumors had to be treated with 10 Gy × 5 fractions. The protocol also defined thresholds for PTV coverage, dose heterogeneity, conformality, intermediate-dose spillage, and organ-at-risk (OAR) protection.
After credentialing, all subsequent SBRT plans were reviewed retrospectively within 30 days of treatment delivery. Sites submitted dosimetry worksheets and deidentified DICOM radiotherapy datasets. These were analyzed centrally using a vendor-neutral treatment evaluation system, and a report card was sent back to each site documenting whether the plan met protocol requirements or contained deviations.
Study Design
This was not a treatment efficacy analysis, but rather a quality assurance report embedded within an ongoing phase 3 randomized trial. The report evaluated the first 100 SBRT plans submitted from VALOR participants treated between April 2017 and October 2022.
The broader VALOR trial enrolls patients with biopsy-confirmed, early-stage localized NSCLC measuring up to 5 cm, without regional or distant metastases, and with tumors located at least 1 cm away from key central thoracic structures at study entry. The main trial plans to compare 5-year overall survival between surgery and SBRT in 670 participants, randomized 1:1.
For this QA analysis, the investigators examined how well participating centers adhered to protocol-defined requirements for contouring, PTV margin expansion, prescription selection, target coverage, dose conformality, intermediate-dose spillage, and OAR constraints. They also documented the site credentialing process, the types of treatment delivery methods used, and the corrective actions taken when deviations were identified.
Results
The findings show that the centralized QA system was feasible and highly effective. The first 100 SBRT plans were submitted from 9 VA facilities, and 98% were received within the required 30-day timeframe after treatment completion.
During the site credentialing phase, the initial SBRT plan submission met protocol requirements in 79% of cases. When resubmissions were needed, the main issues involved inaccurate contouring of the trachea, bronchial tree, or chest wall, use of multiple IGTV contours, PTV expansions smaller than the required minimum, or incorrect participant identification labeling.
The IROC lung phantom validation results were strong. For stationary targets, the mean difference between measured and calculated dose was −0.2% at one point and −0.5% at another, with an average gamma pass rate of 97.9%. For moving targets, the differences were −1.0% and −2.4%, with an average gamma pass rate of 92.8%. All values met IROC passing standards, supporting the technical readiness of participating sites.
Target contouring performance was excellent overall. Tumor contours were considered appropriate in all submissions. PTV expansion was protocol compliant in 98% of cases, with only 2% showing margins smaller than the required 5 mm. Contouring accuracy for critical OARs varied somewhat by structure: it was 75% for the proximal bronchial tree, 92% for the trachea, 100% for the esophagus, 100% for the spinal cord, and 95% for the brachial plexus.
Prescription compliance was also high. Peripheral tumors accounted for 72% of participants. Among them, 43% were treated in 3 fractions, 13% in 4 fractions, and 44% in 5 fractions. Among central tumors, 26 patients received the correct prescription of 10 Gy × 5 fractions. However, 2 central tumors were incorrectly treated using peripheral prescriptions, one with 11.5 Gy × 5 fractions and one with 18 Gy × 3 fractions. Importantly, these errors did not result in violation of critical OAR dose limits, but they were considered protocol violations and triggered formal corrective action.
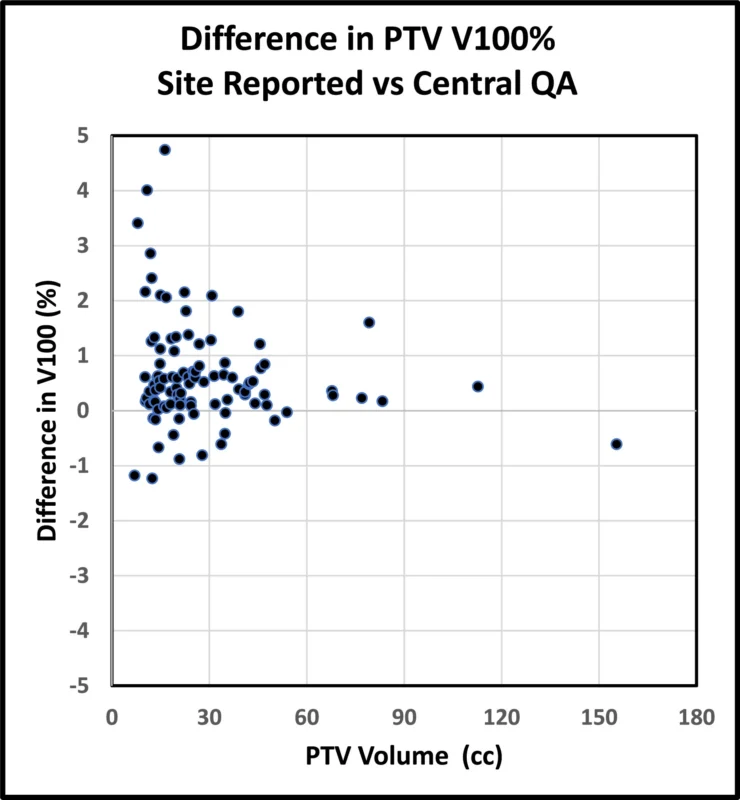
The PTV volume ranged from 7.0 to 155 cm³, with a median of 21.7 cm³. The PTV V100% met protocol in nearly all cases, with only 1 plan showing coverage below the minimum threshold of 94%, achieving 93.2% instead. The median Dmax within the PTV was 125.4% of the prescription dose, with a range of 105.8% to 149.0%. High-dose conformality was acceptable or variation acceptable in 100% of cases, while intermediate-dose compactness metrics were acceptable or variation acceptable in 94%.
Treatment delivery techniques reflected contemporary practice. The most common method was volumetric modulated arc therapy (VMAT), used in 73% of cases. Robotic delivery accounted for 17%, and fixed-field IMRT was used in only 2%.
From a safety standpoint, the results were reassuring. No submission exceeded dose limits to protocol-defined critical OARs. Noncritical OAR limits were also generally respected, except for chest wall and rib constraints, which were exceeded in 26% and 20% of cases, respectively. These were classified as minor violations.
Key Findings
This report demonstrates that a centralized QA system can be successfully implemented across a large health care network, even one without previous experience sponsoring a multisite SBRT trial. The most important finding is that the first 100 patients randomized to SBRT in VALOR were treated appropriately and without major safety concerns.
Protocol compliance was high across most technical domains. Tumor contouring was appropriate in all cases, target coverage was excellent, and critical OAR protection was maintained throughout. The few deviations that did occur were identifiable, understandable, and correctable.
A particularly important observation was the difficulty some centers had in determining whether a tumor should be classified as central or peripheral. This led to the two incorrect prescriptions. In response, the study team created a new target assessment worksheet and amended the protocol to require a 2 cm boundary around the IGTV, reviewed in three dimensions against nearby central structures. After this measure was introduced, no additional incorrect central tumor prescriptions were observed.
The report also highlights that minor discrepancies can occur when dose-volume metrics are compared across different planning systems, especially for small targets. This is clinically relevant because it shows why centralized review remains valuable even when local planning appears acceptable.
Conclusion
The centralized QA report from the VALOR trial provides strong evidence that protocol-defined SBRT for operable early-stage NSCLC can be delivered safely and consistently across multiple VA sites. Among the first 100 participants, compliance with contouring, prescription, target coverage, dose conformality, and critical OAR protection was high. No critical OAR dose violations were found, and the few important deviations that emerged were addressed through structured corrective action.
This work is valuable not only for the ongoing VALOR trial, but also for the wider field of thoracic radiation oncology. It shows that a carefully designed QA program can preserve patient safety, support scientific rigor, and serve as a model for other institutions seeking to implement centralized radiotherapy oversight in clinical trials.