Can advanced radiation technologies translate into meaningful improvements for patients? In the phase 3 TORPEdO trial, intensity-modulated proton therapy (IMPT) was compared with standard IMRT in patients with oropharyngeal cancer, focusing not only on disease control but also on functional outcomes and quality of life. The results provide important real-world evidence on whether dosimetric advantages translate into clinical benefit.
Title:Proton beam therapy for oropharyngeal cancer (TORPEdO): a phase 3, randomised controlled trial
Authors:David J Thomson , James Price, Matthew Tyler, Matthew Beasley, Jim Lester, Christopher Nutting, Nachi Palaniappan, Robin Prestwich, Shanmugasundaram Ramkumar, Anna Thompson, Guy Betts, Helen Bulbeck, Frances Charlwood, Matthew Clarke, Matthew Lowe, Justin Roe, Justine Tyler, Lorna Wilson, Jane L Wolstenholme, Kevin Chiu, Judith Christian, Clare Cruickshank, Deborah Gardiner, Holly Tovey, Prof Catharine M West, Emma Hall
Published in The Lancet, March 21, 2026
Background
Intensity-modulated radiation therapy (IMRT) with concurrent chemotherapy remains the standard of care for locally advanced oropharyngeal squamous cell carcinoma, achieving high cure rates but often at the cost of significant long-term toxicity. Persistent impairments in swallowing, taste, and salivation continue to affect quality of life (QoL), with up to one-third of patients experiencing poor swallowing outcomes two years after treatment.
Intensity-modulated proton therapy (IMPT) has been proposed as a strategy to reduce radiation exposure to surrounding organs at risk, including salivary glands and swallowing structures, potentially translating into improved functional outcomes. However, robust randomized evidence comparing IMPT with IMRT, particularly in terms of patient-reported outcomes and long-term function, has been limited.
Methods
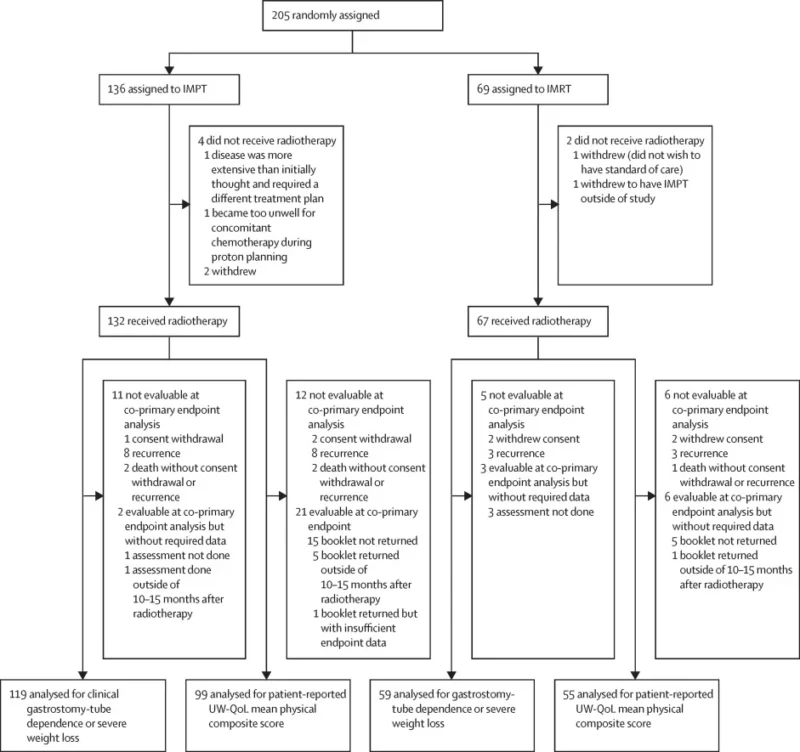
The TORPEdO trial was a phase 3, multicentre, open-label, randomized controlled study conducted across 20 UK National Health Service hospitals. Patients with newly diagnosed locally advanced oropharyngeal squamous cell carcinoma were randomly assigned in a 2:1 ratio to receive either IMPT or IMRT.
All patients received 70 Gy in 33 fractions over 6.5 weeks, alongside concurrent chemotherapy with high-dose cisplatin (100 mg/m² every 3 weeks). The co-primary endpoints at 12 months were:
- Gastrostomy-tube dependence or severe weight loss (≥20% from baseline)
- University of Washington Quality of Life (UW-QoL) physical composite score
Secondary endpoints included adverse events, functional outcomes, loco-regional control, and overall survival.
Study Design and Patient Population
Between February 2020 and June 2023, 205 patients were enrolled, with 136 (66%) assigned to IMPT and 69 (34%) to IMRT. Nearly half of patients (48%) had T3–T4 disease, and 22% had bilateral lymph node involvement.
The median age was approximately 57 years, and the majority of patients were male (80%). Most tumors were p16-positive (96%), reflecting a predominance of HPV-associated disease. Treatment compliance was high, with 97% of patients receiving the allocated therapy.
Results
Clinical Endpoints: Weight Loss and Feeding Tube Dependence
At 12 months, the composite endpoint of gastrostomy-tube dependence or grade 3 weight loss occurred in:
- 21 of 119 patients (18%) in the IMPT group
- 4 of 59 patients (7%) in the IMRT group
This difference did not reach statistical significance (adjusted OR 2.80; 97.5% CI 0.75–10.41; p=0.079), but was primarily driven by higher rates of severe weight loss in the IMPT group.
Gastrostomy-tube dependence remained low and identical between groups (2% vs 2%), suggesting that the observed difference was not related to feeding-tube use but rather to post-treatment weight trajectories.
Grade 3 weight loss peaked at 18% in the IMPT group at 12 months, compared with consistently low rates (5–8%) in the IMRT group. By 24 months, grade 3 weight loss persisted in 13% of IMPT patients, with no cases reported in the IMRT group.
Quality of Life Outcomes
At 12 months, mean UW-QoL physical composite scores were:
- 78.3 in the IMPT group
- 77.1 in the IMRT group
The adjusted difference was 1.3 (97.5% CI –3.7 to 6.2; p=0.56), indicating no meaningful difference between treatment modalities.
Both groups demonstrated a similar trajectory: QoL declined during treatment, improved by 12 months, and remained stable thereafter. Although most patients recovered to mild or minimal symptoms, a subset continued to report persistent deficits in taste and salivation.
Adverse Events
Acute grade 3 radiotherapy-related adverse events were less frequent with IMPT:
- 50% in IMPT vs 72% in IMRT (p=0.0035)
Notably:
- Grade 3 mucositis: 37% vs 54%
- Grade 3 dysphagia: 12% vs 24%
However, late grade 3 adverse events were numerically higher in the IMPT group:
- 25% vs 14% (p=0.081)
When excluding hearing loss and weight loss, late grade 3 toxicity rates were low and similar between groups (6% vs 3%).
No grade 4 radiotherapy-related events or treatment-related deaths were reported. A total of 14 serious adverse events occurred in 12 patients, with only five considered treatment-related.
Disease Control and Survival
At a median follow-up of 28.3 months:
24-month loco-regional control:
- 94% (IMPT) vs 97% (IMRT)
- HR 2.6; p=0.24
24-month overall survival:
- 95% in both groups
- HR 1.6; p=0.47
These findings demonstrate comparable oncologic outcomes between IMPT and IMRT.
Key Findings
IMPT did not reduce gastrostomy-tube dependence or improve long-term quality of life compared with IMRT, despite achieving lower radiation doses to organs at risk. Severe weight loss was more frequent with IMPT, although it rarely translated into clinically significant underweight status.
Patient-reported outcomes, swallowing function, and survival outcomes were similar between groups. IMPT was associated with fewer acute toxicities but did not confer sustained long-term functional benefits.
Conclusion
In this phase 3 randomized trial, intensity-modulated proton therapy did not demonstrate superiority over IMRT in improving functional outcomes or quality of life in patients with oropharyngeal squamous cell carcinoma. Disease control and survival were comparable between modalities.
Although IMPT reduced acute toxicity, these advantages did not translate into meaningful long-term clinical benefit. In settings where IMPT is not routinely available, IMRT remains the standard of care.
These findings highlight the importance of randomized evidence in evaluating advanced, resource-intensive technologies and underscore the need to balance dosimetric advantages with clinically meaningful patient outcomes.
Written by Aren Karapetyan, MD


