Thymic health and immunotherapy outcomes in patients with cancer highlights a new direction in biomarker research by focusing on the patient’s immune system rather than the tumor alone. While current markers like PD-L1 and tumor mutational burden are limited, this study shows that thymic health—assessed from routine CT scans—is strongly associated with immunotherapy outcomes across multiple cancers, suggesting a novel, host-driven approach to precision oncology.
Title: Thymic health and immunotherapy outcomes in patients with cancer
Authors: Simon Bernatz, Vasco Prudente, Suraj Pai, Asbjørn K. Attermann, Alessandro Di Federico, Andrew Rowan, Selvaraju Veeriah, Lars Dyrskjøt, Leonard Nürnberg, Joao V. Alessi, Patrick A. Ott, Elad Sharon, Allan Hackshaw, Nicholas McGranahan, Christopher Abbosh, Raymond H. Mak, Danielle Bitterman, Mark Awad, Biagio Ricciuti, Charles Swanton, Mariam Jamal-Hanjani, Nicolai J. Birkbak, Hugo J. W. L. Aerts
Background
Immune checkpoint inhibitors have changed the treatment landscape for many cancers, including non-small cell lung cancer, melanoma, and renal cell carcinoma. However, only a subset of patients derive durable benefit, and currently used biomarkers such as PD-L1 expression and tumor mutational burden do not fully explain response variability. This Nature study explored whether a host-related factor, thymic health, could help fill that gap.
The thymus is central to T-cell maturation and adaptive immune competence, yet it is often overlooked in adults because it involutes with age. Using routine chest CT imaging and a deep-learning model, the investigators quantified “thymic health” as a radiographic proxy for thymic function and tested whether it was associated with outcomes in patients treated with immune checkpoint inhibitors. Across a large real-world pan-cancer population of 3,476 patients, and with biological validation in the prospective TRACERx lung cancer cohort, the study showed that better thymic health was consistently linked with improved immunotherapy outcomes.
Methods
The investigators analyzed two main retrospective real-world cohorts from the Dana-Farber Harvard Cancer Center. The Harvard-NSCLC cohort included 1,218 patients with non-small cell lung cancer treated with checkpoint inhibitors. The Harvard-PAN cohort included 2,258 additional patients with other cancers treated with immune checkpoint inhibitors, including melanoma, renal, breast, bladder, esophageal, and several smaller pooled tumor groups. Eligible patients were adults with baseline chest CT scans obtained within 3 months before the start of immunotherapy.
To quantify thymic health, the authors developed an automated deep-learning system trained on 5,674 independent thoracic CT scans. The model generated a continuous thymic health score from 0 to 1, reflecting increasing preservation of thymic structure and presumed function. For outcome analyses, patients were grouped as low thymic health (bottom 25%), average thymic health (middle 50%), or high thymic health (top 25%).
For biological validation, the study used the prospective TRACERx cohort of 464 treatment-naive, operable stage I–IIIB NSCLC patients. In this cohort, thymic health was correlated with biological markers of thymic activity and adaptive immunity, including signal joint T-cell receptor excision circles (sjTRECs), T-cell receptor diversity, blood T-cell fraction, tumor T-cell infiltration, and plasma proteomic signatures.
Clinical endpoints included progression-free survival in the immunotherapy-treated NSCLC cohort, overall survival in NSCLC and pan-cancer cohorts, and disease-free survival in TRACERx.
Study Design
This was a multi-cohort imaging biomarker study combining retrospective real-world clinical outcome analysis with prospective biological validation. The design had three major components.
First, the authors evaluated whether thymic health measured on baseline CT scans was associated with survival outcomes in 1,218 patients with NSCLC treated with immunotherapy. Among these patients, 793 received immunotherapy monotherapy and 425 received immunotherapy plus chemotherapy. Of those treated with monotherapy, 245 patients, or 31%, received first-line treatment. Of those treated with chemo-immunotherapy, 407 patients, or 96%, received first-line treatment.
Second, they compared thymic health with established tumor-centric biomarkers, particularly PD-L1 and tumor mutational burden, to determine whether thymic health provided overlapping or independent prognostic information.
Third, they extended the analysis to a pan-cancer cohort of 2,258 patients with melanoma, renal, breast, bladder, esophageal, and other cancers, and then linked imaging-derived thymic health to immune biology in the TRACERx cohort.
This structure allowed the authors not only to test clinical associations, but also to provide mechanistic support for the idea that thymic health reflects real immune competence rather than being just an imaging correlate.
Results
The strongest findings were seen in NSCLC. In the full Harvard-NSCLC cohort, patients with higher thymic health had significantly better progression-free survival after immune checkpoint inhibitor therapy. Compared with the low thymic health group, the high thymic health group had a hazard ratio for progression or death of 0.65, with a 95% confidence interval of 0.54 to 0.77. The average thymic health group also performed better, with a hazard ratio of 0.67 and a 95% confidence interval of 0.58 to 0.78. These findings were statistically significant with a type III P value below 0.001.
The benefit remained visible in both treatment settings. In patients treated with chemo-immunotherapy, high versus low thymic health was associated with a hazard ratio of 0.58 for progression or death. In patients treated with immunotherapy monotherapy, the hazard ratio was 0.66. Associations were stronger in the first-line setting and remained significant after adjustment for sex, age, ECOG performance status, histology, PD-L1, and tumor mutational burden.
Overall survival findings were similarly compelling. In the full NSCLC cohort, high versus low thymic health was associated with a hazard ratio for death of 0.56, with a 95% confidence interval of 0.46 to 0.68. Average versus low thymic health had a hazard ratio of 0.64, with a 95% confidence interval of 0.55 to 0.76. Again, both were highly significant with type III P below 0.001. These associations also held across treatment subgroups and after multivariable adjustment.
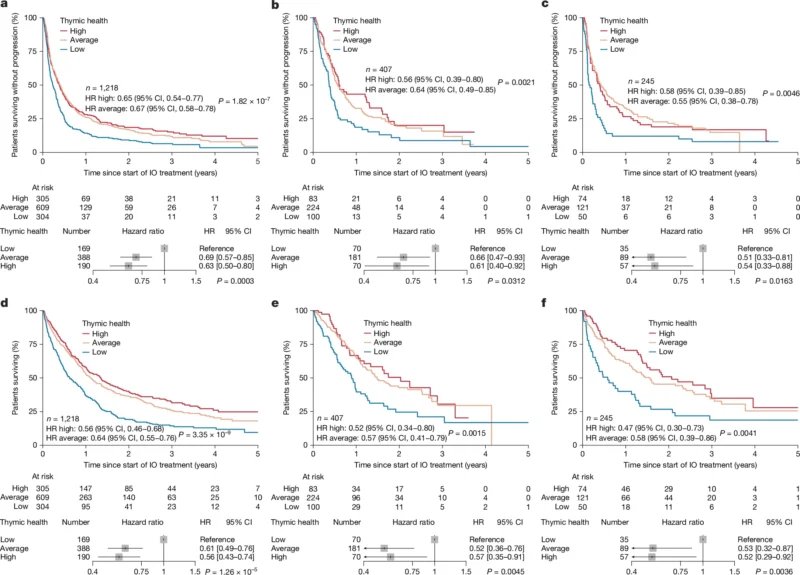
An especially important part of the paper was the comparison with PD-L1 and tumor mutational burden. Thymic health retained prognostic value across clinically relevant PD-L1 and TMB subgroups. Among patients with PD-L1 less than 1%, those with high thymic health had a 44% lower risk of progression compared with those with low thymic health. In first-line NSCLC, PD-L1 expression of at least 50% was associated with a progression hazard ratio of 0.77 and a death hazard ratio of 0.64 compared with PD-L1 below 50%. By contrast, high thymic health was associated with a progression hazard ratio of 0.57 and a death hazard ratio of 0.50 compared with low thymic health.
The study also found no significant interaction between thymic health and PD-L1, and no significant correlation between thymic health and either PD-L1 or TMB, suggesting that thymic health captures a different biological dimension.
The biological validation in TRACERx strengthened the clinical findings. Patients with high thymic health had significantly higher sjTREC levels, consistent with greater thymic output of naive T cells. Higher thymic health was also associated with greater peripheral blood T-cell receptor alpha and beta chain diversity, higher circulating T-cell abundance, and higher intratumoral beta-chain T-cell receptor diversity. Proteomic analysis in 108 patients showed that higher thymic health correlated with adaptive immune signaling pathways. These results support the interpretation that imaging-defined thymic health reflects real thymic activity and broader adaptive immune competence.
In the pan-cancer cohort of 2,258 patients, higher thymic health was associated with lower risk of death across all examined cancer types. Statistically significant associations were reported in melanoma, breast cancer, renal cancer, and the pooled “other” cancers. Although effect size varied by tumor type, the overall direction was consistent and supported a tumor-agnostic prognostic role.
Key Findings
This study introduces a new perspective in cancer immunotherapy by emphasizing the role of the patient’s immune system, rather than focusing only on tumor characteristics.
- Shifts the focus of biomarker research from tumor-centric factors to the patient’s immune system and overall immune competence
- Highlights the limitations of current biomarkers such as PD-L1 expression and tumor mutational burden, which do not fully predict immunotherapy response
- Introduces thymic health as a novel, host-driven biomarker reflecting the functional state of the adaptive immune system
- Demonstrates that thymic health can be assessed non-invasively using routine chest CT scans through a deep-learning approach
- Shows a strong and consistent association between higher thymic health and improved immunotherapy outcomes across multiple cancer types
- Suggests that thymic health provides independent and complementary prognostic value beyond established biomarkers
- Supports a shift toward more holistic, precision oncology strategies that integrate both tumor biology and host immune status
Conclusion
This Nature study presents strong evidence that thymic health is associated with immunotherapy outcomes across cancers, especially in NSCLC. Using a deep-learning model applied to routine CT scans, the investigators showed that high thymic health correlates with reduced risks of progression and death, independent of standard biomarkers like PD-L1 and tumor mutational burden.
The accompanying biological analyses support thymic health as a surrogate for thymic function, T-cell diversity, and host immune competence. Although the authors acknowledge the need for broader validation across scanners, institutions, and more diverse populations, the findings introduce a compelling new host-based biomarker for precision immuno-oncology. This work suggests that future treatment stratification may benefit from considering not only the tumor, but also the functional state of the patient’s immune system.


