Sentinel lymph node biopsy (SNB) has long been part of melanoma management because it improves nodal staging and helps identify patients at higher risk of recurrence. What has remained controversial, however, is whether SNB does more than refine prognosis. The key unresolved question has been whether performing SNB actually improves melanoma-specific survival. In this systematic review and meta-analysis published in The Lancet Oncology on March 5, 2026, Varey and colleagues addressed that question by pooling the largest body of comparative evidence to date. Their findings suggest that SNB is associated not only with better staging and regional disease control, but also with a meaningful reduction in the risk of melanoma-specific death and recurrence.
Background
Cutaneous melanoma often spreads first to the regional lymphatic basin, especially the directly draining sentinel lymph node. Sentinel node biopsy was developed as a minimally invasive surgical procedure to identify and remove these first draining nodes for pathological assessment. Its value as a staging tool is well established, and previous studies have shown that sentinel node status adds important prognostic information beyond standard clinicopathological features such as Breslow thickness and ulceration. SNB has also been associated with better regional nodal control because it can detect occult nodal metastases before they become clinically apparent.
Despite these advantages, the survival value of SNB has remained debated for years. The landmark MSLT-I randomized trial suggested a possible reduction in melanoma-specific mortality with SNB, but the result did not reach statistical significance, largely because the trial was underpowered for that endpoint. A previous smaller meta-analysis published in 2016 also hinted at a survival advantage, but that conclusion depended heavily on one influential study. As a result, uncertainty persisted. This new systematic review and meta-analysis was designed to provide a more precise and robust estimate of whether SNB truly improves survival outcomes in patients with cutaneous melanoma.
Methods
The investigators conducted a systematic review and meta-analysis according to PRISMA guidelines and registered the protocol in PROSPERO (CRD4203494674). They searched Medline, Embase, Cochrane CENTRAL, and ClinicalTrials.gov up to Jan 8, 2025, and also reviewed two conference proceedings from 2024 that were not indexed in Embase.
Studies were eligible if they included primarily adult patients with cutaneous melanoma and compared outcomes between those who underwent SNB and those who did not. To be included, studies had to report survival-related outcomes such as melanoma-specific death, overall survival, timepoint survival rates, or hazard ratios with confidence intervals. Studies involving only children, those without a comparator group, and those in which all patients underwent full regional lymph node dissection were excluded.
Title review and data extraction were performed independently by multiple authors. Extracted variables included study design, institution, data source, recruitment period, patient characteristics, melanoma features, follow-up, recurrence outcomes, and survival data. When multiple studies used overlapping datasets, the authors included only the largest or most appropriate study to avoid duplication.
The primary outcome was death from melanoma. Secondary outcomes included risk of recurrence. For studies that reported survival curves rather than standard effect estimates, graph digitization software was used to estimate data points. The authors pooled adjusted estimates when available, including results from randomized studies, multivariable analyses, and propensity score matched analyses. They used random-effects models for most analyses and evaluated heterogeneity with the I² statistic. Sensitivity analyses and subgroup analyses were also performed. Risk of bias was assessed with the Newcastle–Ottawa scale for non-randomized studies and the Cochrane risk of bias tool for randomized trials. Evidence certainty was evaluated using GRADE.
Study Design
A total of 1560 records were screened, and 60 studies were ultimately eligible for inclusion in the systematic review. These studies represented a broad range of designs, including one randomized controlled trial, multiple concurrent comparative cohort studies, historical-control studies, and mixed-control studies.
For the main melanoma-specific survival analysis, the authors focused on studies that attempted to control for prognostic imbalance between sentinel lymph node biopst and no-SNB groups. Of the 22 studies with unique patient datasets reporting melanoma death, 14 studies had used approaches such as randomization, multivariable adjustment, or propensity score matching. One of these was excluded from the principal meta-analysis because it included only patients with unresectable stage IIIC or IV disease, introducing major selection bias. The main pooled analysis therefore included 13 studies with 40,287 participants.
Among these 13 studies, 10 used multivariable analysis, 2 used propensity score matching, and 1 was a randomized trial with adjusted analysis. Median follow-up across these studies was 43.5 months, ranging from 32.4 to 50.4 months. For recurrence, 9 adjusted studies involving 11,625 participants were included in the pooled analysis.
Results
The main finding was that patients with melanoma who underwent SNB had a significantly reduced risk of death from melanoma compared with those who did not undergo the procedure. In the primary adjusted analysis of 13 studies, the pooled hazard ratio (HR) was 0.86 with a 95% confidence interval (CI) of 0.81–0.92, and the result was highly statistically significant (p<0.0001). Heterogeneity was low, with I²=16%, supporting the consistency of the finding across studies.
The analysis of 5-year melanoma-specific death produced a similar result. Across 5 studies involving 27,540 participants, the pooled HR was 0.84 (95% CI 0.78–0.90; p<0.0001), again with low heterogeneity (I²=15%). This suggests that the survival benefit of SNB is already visible within 5 years of follow-up.
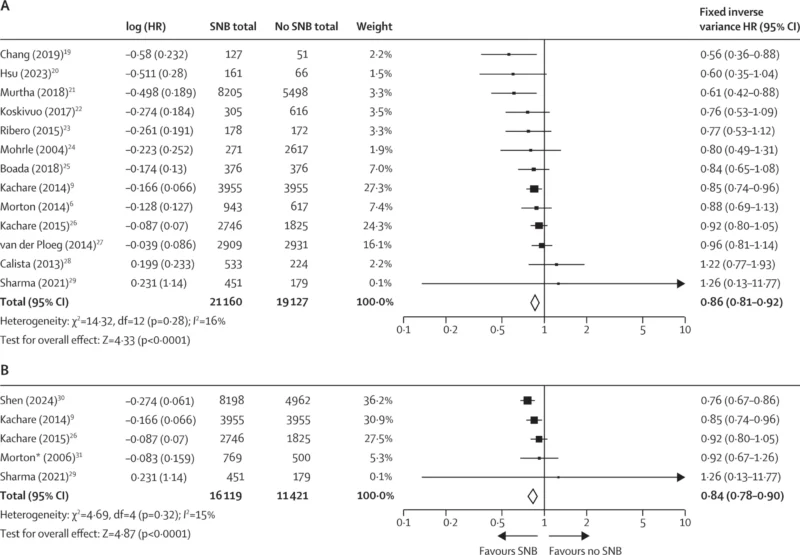
For 10-year melanoma-specific death, the result was less definitive. Only 2 studies with 3 estimates contributed to this analysis, and the pooled HR was 0.87 (95% CI 0.71–1.06; p=0.17), with somewhat greater heterogeneity (I²=41%). This longer-term analysis was not statistically significant, likely reflecting the smaller evidence base rather than clear absence of benefit.
SNB was also associated with a significant reduction in recurrence risk. In 9 studies including 11,625 participants, the pooled HR for recurrence was 0.71 (95% CI 0.66–0.76; p<0.0001), with low heterogeneity (I²=23%). This means SNB was associated with a 29% relative reduction in recurrence risk, which is clinically important and consistent with the known role of the procedure in regional disease control.
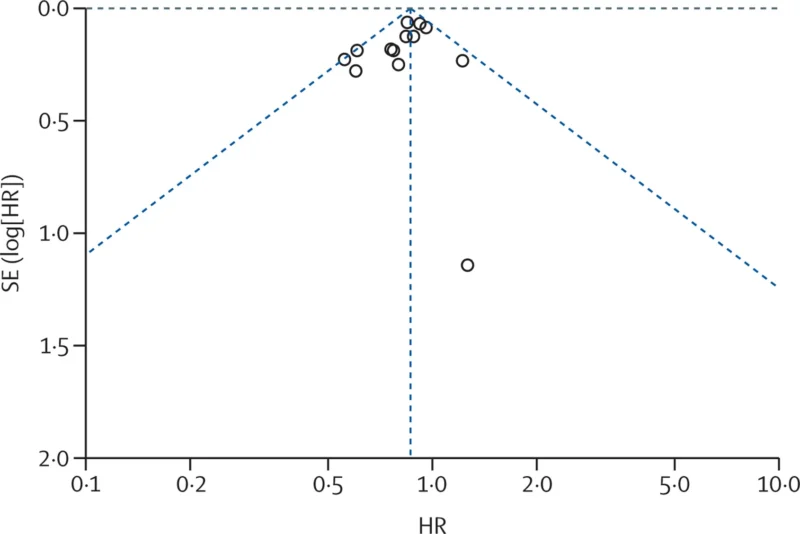
The authors also performed multiple sensitivity analyses. Excluding heavily weighted studies did not materially alter the effect estimates, confidence intervals, or statistical significance. Subgroup analyses based on median age, Breslow thickness, adjustment method, and follow-up duration showed no significant differences that would challenge the main conclusion. Analyses related to immune checkpoint inhibitors and targeted therapies also did not show evidence that modern systemic therapy explained the observed survival advantage. Publication bias appeared low by both funnel plot assessment and Egger’s test (p=0.251).
Key Findings
This meta-analysis provides the strongest evidence to date that SNB in cutaneous melanoma is associated with improved melanoma-specific survival. The 14% relative reduction in melanoma death risk seen in the main analysis is modest but meaningful, especially given the large population studied and the consistency across analyses. The 16% reduction in 5-year melanoma-specific mortality and the 29% reduction in recurrence risk strengthen the argument that SNB is not only a staging procedure, but also one with therapeutic value.
Importantly, the magnitude of benefit was similar to what had been observed in the only randomized trial, MSLT-I, even though that study lacked sufficient power to prove significance. This consistency supports biological plausibility. The authors also highlight that SNB remains less morbid and less costly than many later-stage interventions, including therapeutic lymphadenectomy and adjuvant immunotherapy. With an estimated additional cost of about USD 6,170 per patient for SNB compared with USD 100,000 to 200,000 per patient for immunotherapy, preventing recurrence and reducing the need for later systemic therapy may have important health-economic implications.
Conclusion
This large systematic review and meta-analysis shows that sentinel node biopsy for cutaneous melanoma is associated with a significant reduction in melanoma-specific death and recurrence. In the most robust analysis, SNB reduced the risk of melanoma death by 14% and recurrence by 29%, with low heterogeneity and stable results across sensitivity analyses. These findings reinforce the role of SNB as more than a diagnostic staging tool. For eligible patients with melanoma, SNB appears to offer meaningful clinical benefit by improving outcomes while potentially reducing the need for more morbid and costly therapies later in the disease course.


