Rezatapopt (PC14586) is a first-in-class, oral, selective p53 reactivator designed to restore the function of the TP53 Y220C mutant protein. Although TP53 is the most frequently mutated gene in cancer, therapeutic strategies directly targeting mutant p53 have historically been unsuccessful. The Y220C mutation is structurally distinct because it creates a specific binding pocket within the p53 protein, enabling rational drug development aimed at stabilizing the mutant protein in its wild-type conformation.
The PYNNACLE phase 1 study evaluated rezatapopt in patients with advanced solid tumors harboring a TP53 Y220C mutation, with the goal of defining its safety profile, optimal dose, and early signals of clinical activity. This trial provides early clinical evidence supporting the concept of mutation-selective p53 reactivation as a targeted treatment strategy in solid tumors.
Background
TP53 is the most frequently mutated gene in human cancer, with alterations present in more than 50% of solid tumors. The p53 protein plays a central role in tumor suppression by regulating cell-cycle arrest, apoptosis, DNA repair, and genomic stability. When TP53 is mutated, this protective mechanism fails, contributing to tumor progression and treatment resistance. Among TP53 variants, the Y220C mutation accounts for approximately 1% of all solid tumors and represents the ninth most common TP53 mutation. Unlike many TP53 mutations that disrupt DNA binding or zinc coordination, the Y220C mutation creates a distinct structural pocket in the p53 protein, leading to thermodynamic instability and loss of tumor-suppressor function.
Rezatapopt (PC14586) is a first-in-class, oral, selective small-molecule p53 reactivator specifically designed to bind this Y220C-created pocket. By stabilizing the mutant protein in a wild-type conformation through noncovalent hydrogen bonding and enhanced hydrophobic interactions, rezatapopt restores p53 transcriptional activity. Preclinical studies demonstrated selective activation of Y220C-mutated p53 without affecting wild-type or other mutant p53 forms, resulting in dose-dependent tumor inhibition. The PYNNACLE phase 1 trial evaluated rezatapopt in patients with advanced solid tumors harboring TP53 Y220C mutations.
Methods
This was a phase 1–2, multicenter, single-group study. Phase 1 consisted of dose-escalation and dose-optimization components. The primary objectives were to determine the maximum tolerated dose (MTD), establish the recommended phase 2 dose (RP2D), and evaluate safety. Primary endpoints included dose-limiting toxic effects (DLTs), adverse events (AEs), and serious adverse events. Secondary endpoints included preliminary efficacy (overall response rate, duration of response, progression-free survival, overall survival) and pharmacokinetics.
Adverse events were graded according to CTCAE v5.0. Tumor response was assessed using RECIST v1.1 (or PCWG3-modified RECIST for prostate cancer) by both investigators and blinded independent central review. Pharmacokinetic analyses included maximum plasma concentration (Cmax), area under the curve (AUC), and half-life.
Approximately 80 patients were planned for enrollment in phase 1. Time-to-event endpoints were analyzed descriptively using Kaplan–Meier methods.
Study Design
Eligible patients were adults (≥18 years) with locally advanced or metastatic solid tumors harboring TP53 Y220C mutations confirmed by validated sequencing assays. All patients had progressive disease after at least one prior systemic therapy and ECOG performance status 0–1. Patients were heavily pretreated, with a median of three prior lines of therapy (range, 1–9).
Rezatapopt was administered orally in continuous 21-day cycles. Eight dose levels were explored: 150 mg, 300 mg, 600 mg, 1150 mg, 1500 mg, 2000 mg (with or without food), 2500 mg once daily, and 1500 mg twice daily.
Dose escalation followed an accelerated design until the first grade ≥2 treatment-related adverse event occurred, after which a modified toxicity probability interval method was used. Following identification of the MTD, dose optimization was performed under FDA Project Optimus principles, emphasizing safety–efficacy balance. A cohort receiving 2000 mg once daily with food was added after pharmacokinetic modeling demonstrated improved exposure and tolerability.
Results
A total of 77 patients received rezatapopt. The most common tumor types were ovarian cancer (30%), pancreatic cancer (18%), breast cancer (13%), and colorectal cancer (12%). Twenty-nine percent had co-occurring KRAS mutations.
No DLTs occurred with once-daily dosing up to 2500 mg. At 1500 mg twice daily, DLTs occurred in 2 of 10 patients (20%), including grade 3 transaminase elevation and grade 3 acute kidney injury. The MTD was 1500 mg twice daily.Based on integrated safety, efficacy, and pharmacokinetic data, 2000 mg once daily with food was selected as the RP2D.
Ninety-nine percent of patients experienced at least one adverse event. The most common AEs were:
- Nausea: 58%
- Vomiting: 44%
- Increased creatinine: 39%
- Fatigue: 39%
- Anemia: 36%
Anemia was the most common grade ≥3 event (16%). Treatment-related AEs occurred in 87% of patients; grade 3–4 treatment-related AEs occurred in 19 patients. No grade 5 treatment-related AEs were reported. Two patients (3%) discontinued treatment due to treatment-related toxicity.
Administration with food reduced gastrointestinal toxicity and increased exposure: at steady state, Cmax was ~30% higher and AUC ~40% higher with food. Median half-life was 20.5 hours.
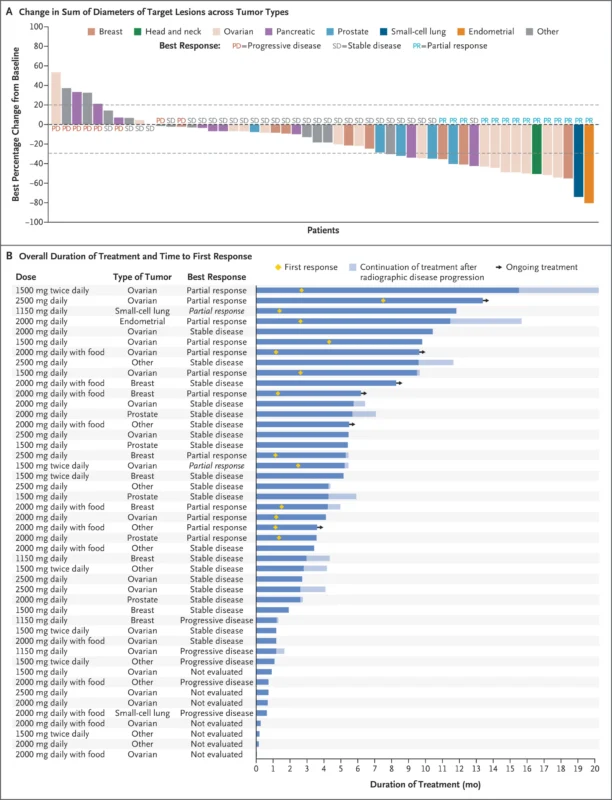
Among 71 efficacy-evaluable patients, the investigator-assessed overall response rate (ORR) was 20%. Fourteen confirmed responses were observed across tumor types, including ovarian (7 patients), breast (3), endometrial (1), head and neck (1), prostate (1), and small-cell lung cancer (1). Two complete responses (3%) were reported.
Median time to response was 1.4 months. Median duration of response was 7.0 months (95% CI, 2.8–10.6). Median progression-free survival was 4.3 months (95% CI, 2.8–5.5). Median overall survival was 11.4 months (95% CI, 6.4–14.9).
Importantly, all responding tumors harbored TP53 Y220C and wild-type KRAS. Among patients with KRAS wild-type tumors treated at efficacious doses (≥1150 mg daily), ORR reached 30% (14/46 patients). None of the 22 patients with co-occurring KRAS mutations achieved confirmed response.
Circulating tumor DNA analysis showed on-target activity: 95% of patients had a reduction in TP53 Y220C variant allele frequency by cycles 2–3, with ≥50% reduction in 78% and ≥95% reduction in 39%.
Key Findings
Rezatapopt demonstrated proof of concept for direct pharmacologic reactivation of a mutant p53 protein in solid tumors. The drug showed single-agent activity across multiple tumor types in heavily pretreated patients, with a 20% overall response rate and durable responses lasting a median of 7 months.
Clinical activity was strongly associated with KRAS wild-type status. Co-occurring KRAS mutations appeared to limit response, highlighting potential biological resistance mechanisms.
The safety profile was manageable, dominated by gastrointestinal toxicity and anemia. Food administration improved pharmacokinetics and tolerability, leading to selection of 2000 mg once daily with food as the recommended phase 2 dose.
Conclusion
The PYNNACLE phase 1 study demonstrates that rezatapopt, a first-in-class selective p53 reactivator targeting TP53 Y220C, has manageable toxicity and meaningful antitumor activity in heavily pretreated patients with advanced solid tumors. With a recommended phase 2 dose of 2000 mg once daily with food, ongoing expansion cohorts are evaluating efficacy in TP53 Y220C and KRAS wild-type tumors.
For the first time, direct pharmacologic reactivation of a structurally defined p53 mutant has translated into clinical responses, representing a major milestone in targeting TP53-driven cancers.


