The OrigAMI-1 trial evaluated amivantamab, a bispecific EGFR–MET antibody, as monotherapy in 94 patients with chemorefractory RAS/BRAF wild-type metastatic colorectal cancer who had received a median of two prior treatment lines.
The study showed clinically meaningful activity across tumor locations, with objective response rates of 29% in left-sided anti-EGFR–naïve disease, 19% in left-sided disease previously treated with anti-EGFR therapy, and 22% in right-sided disease. Responses were durable, with median duration of response ranging from 6.1 to 9.8 months, supporting further investigation of amivantamab in this difficult-to-treat mCRC population.
Title: Amivantamab Monotherapy in Chemorefractory RAS/BRAF Wild-Type Metastatic Colorectal Cancer: Results From OrigAMI-1, an Open-Label, Phase Ib/II Study
Authors: Paul E. Oberstein, J. Randolph Hecht, Kanwal Raghav, MD, MBBS, Filippo Pietrantonio, MD, MD, Victor Moreno, MD, PhD, Eric Van Cutsem, MD, PhD, Rozita Abdul Malik, MBBS, Yong Sang Hong, MD, PhD, Myung Ah Lee, MD, PhD, Harvey Yu-Li Su, MD, Jeeyun Lee, MD, Sreenivasa Chandana, MD, PhD, Marcia Cruz-Correa, MD, PhD, AGAF, FASGE, Ying Yuan, MD, PhD, Azura Ahmad, MD, Kuan-Ming Lai, MD, Hung-Chih Hsu, MD, PhD,
Eric Xueyu Chen, MD, PhD, Elena Elez, MD, PhD, Chia-Chi Lin, MD, PhD, Carlos Lopez, MD, PhD, Hans Prenen, MD, PhD, Susana Roselló-Keränen, MD, PhD, Hector Velez, MD, Yu-Min Yeh, MD, Volker Heinemann, MD , Cathy Eng, MD, FASCO, FACP, Seung-Hoon Beom, MD, Sabine Tejpar, MD, PhD, Sanjib Chowdhury, PhD, Xuesong Lyu, PhD, Medha Kamat, PhD, Joshua C. Curtin, PhD,
Bharvin Patel, PhD, John Xie, PhD, Rianka Bhattacharya, PhD, Robert W. Schnepp, MD, PhD, Emrullah Yilmaz, MD, PhD, Ryota Iwasawa, PhD, Mahesh Daksh, PhD, Patricia Lorenzini, MS, Meena Thayu, MD, Mahadi Baig, MD, Han Sang Kim, MD, PhD, and Sae-Won Han, MD, PhD
Background
The OrigAMI-1 trial evaluated amivantamab, a bispecific EGFR-MET antibody, in patients with chemorefractory metastatic colorectal cancer (mCRC), a setting where treatment options remain limited and outcomes are poor. Colorectal cancer accounts for approximately 10% of all cancer diagnoses globally and remains a leading cause of cancer-related mortality. Among patients with metastatic disease, nearly 50% harbor RAS/BRAF wild-type tumors but still experience disease progression after standard chemotherapy and biologic therapies.
Existing late-line treatments, including anti-EGFR antibodies, offer modest efficacy, with response rates typically ranging from 8% to 22%. Resistance mechanisms, particularly involving MET signaling, represent a critical therapeutic challenge. Amivantamab, through dual inhibition of EGFR and MET and immune-mediated cytotoxic activity, was developed to overcome these resistance pathways and improve outcomes in heavily pretreated patients.
Methods
OrigAMI-1 (NCT05379595) is a global, multicenter, open-label phase Ib/II study investigating amivantamab as monotherapy or in combination with chemotherapy in advanced colorectal cancer. This analysis focuses on patients receiving monotherapy. Eligible patients had unresectable or metastatic colorectal cancer with confirmed RAS, BRAF, and EGFR ectodomain wild-type status and no HER2 amplification.
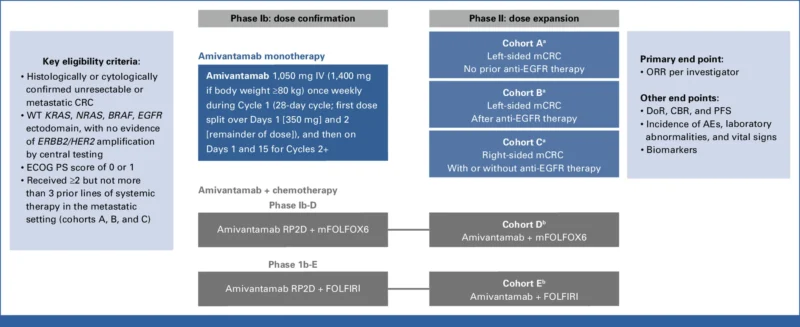
All participants had received at least two prior lines of systemic therapy, including fluoropyrimidine-, oxaliplatin-, irinotecan-based chemotherapy, and anti-VEGF therapy. Tumor tissue and circulating tumor DNA (ctDNA) analyses were required for molecular confirmation.
Study Design
Patients were stratified into three cohorts based on tumor sidedness and prior anti-EGFR exposure. Cohort A included patients with left-sided tumors without prior anti-EGFR therapy, cohort B included left-sided tumors with prior anti-EGFR exposure, and cohort C included right-sided tumors regardless of prior anti-EGFR therapy.
Amivantamab was administered intravenously at a dose of 1,050 mg (or 1,400 mg for patients ≥80 kg), initially weekly in cycle 1 and then every two weeks thereafter until disease progression or unacceptable toxicity. The primary endpoint was objective response rate (ORR) per RECIST v1.1, while secondary endpoints included duration of response (DoR), progression-free survival (PFS), clinical benefit rate (CBR), and safety.
Results
A total of 94 patients received amivantamab monotherapy, with a median follow-up of 11.9 months. The median age was 60 years, and patients had received a median of two prior treatment lines. Notably, 94% had prior exposure to bevacizumab.
Efficacy outcomes varied across cohorts but consistently demonstrated meaningful activity. In cohort A (left-sided, anti-EGFR naïve), the ORR was 29%, with a median DoR of 9.0 months and median PFS of 5.7 months. In cohort B (left-sided, prior anti-EGFR), the ORR was 19%, with a median DoR of 6.1 months and PFS of 4.6 months. In cohort C (right-sided disease), the ORR reached 22%, with a median DoR of 9.8 months and PFS of 3.7 months. These findings are notable given the historically limited responsiveness of right-sided tumors to anti-EGFR therapy.
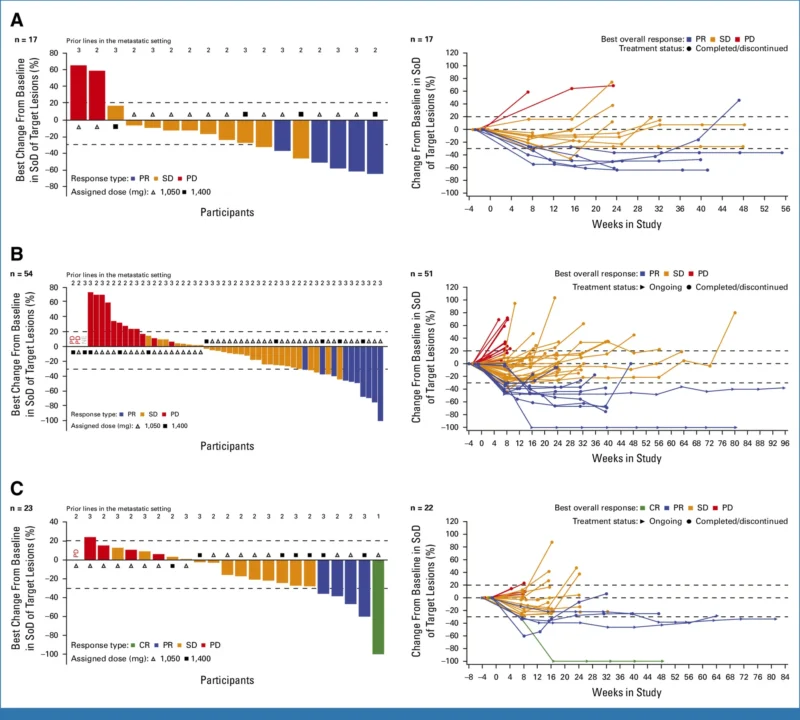
Further analysis in cohort B revealed that patients with a longer interval (>8.8 months) since prior anti-EGFR therapy achieved higher response rates (32%) compared to those with shorter intervals (7%), suggesting a potential role for treatment sequencing and rechallenge strategies.
The clinical benefit rate was substantial across cohorts, reaching 71% in cohort A, 52% in cohort B, and 48% in cohort C. Importantly, responses were durable, with many patients maintaining responses beyond six months.
Key Findings
Amivantamab demonstrated clinically meaningful antitumor activity in heavily pretreated mCRC, with response rates exceeding those historically observed with anti-EGFR monotherapy in similar populations. Activity was observed across both left- and right-sided tumors, addressing an important unmet need in right-sided disease where therapeutic options are particularly limited. The durability of responses, with median DoR approaching 10 months in some cohorts, highlights the potential of dual EGFR-MET inhibition.
Biomarker analyses provided mechanistic insights, showing that higher baseline expression of EGFR ligands AREG and EREG correlated with improved responses. Additionally, treatment induced immune activation, including increased dendritic cell and T-cell signatures, supporting the immune-modulating properties of amivantamab.
Conclusion
The OrigAMI-1 trial establishes amivantamab as a biologically active and clinically meaningful option in chemorefractory metastatic colorectal cancer. The combination of durable responses, activity across tumor subtypes, and a manageable safety profile positions amivantamab as a potential new standard in the late-line setting.
Ongoing phase III trials are expected to further define its role, particularly in earlier lines of therapy and in combination with chemotherapy. These results represent an important step forward in addressing the unmet needs of patients with advanced colorectal cancer who have exhausted standard treatment options.


