Bi-Specific T Cell Engagers (Bi-TCEs) are an emerging class of cancer immunotherapy designed to bring a patient’s own immune cells directly into contact with tumor cells, triggering precise and rapid cancer cell destruction. These engineered proteins act as molecular bridges, simultaneously binding to a tumor-associated antigen on cancer cells and the CD3 receptor on T cells, thereby activating the immune system without the need for complex antigen presentation.
Unlike personalized treatments such as CAR-T therapy, Bi-TCEs are produced as ready-to-use, “off-the-shelf” drugs, making them more accessible and easier to administer. This article explores how Bi-TCEs work, their current clinical applications—particularly in hematologic malignancies—their limitations in solid tumors, and the next generation of innovations aimed at improving their safety, durability, and overall effectiveness in cancer therapy.
Background
Bi-specific T cell engagers (Bi-TCEs) represent a rapidly evolving class of immunotherapy designed to harness the patient’s immune system to eliminate cancer cells with precision. Unlike conventional therapies, these engineered proteins simultaneously bind tumor-associated antigens (TAAs) on cancer cells and CD3 receptors on T cells, physically bringing them into close proximity and triggering targeted cytotoxicity. This mechanism bypasses the need for antigen presentation via the major histocompatibility complex, allowing for broader immune activation.
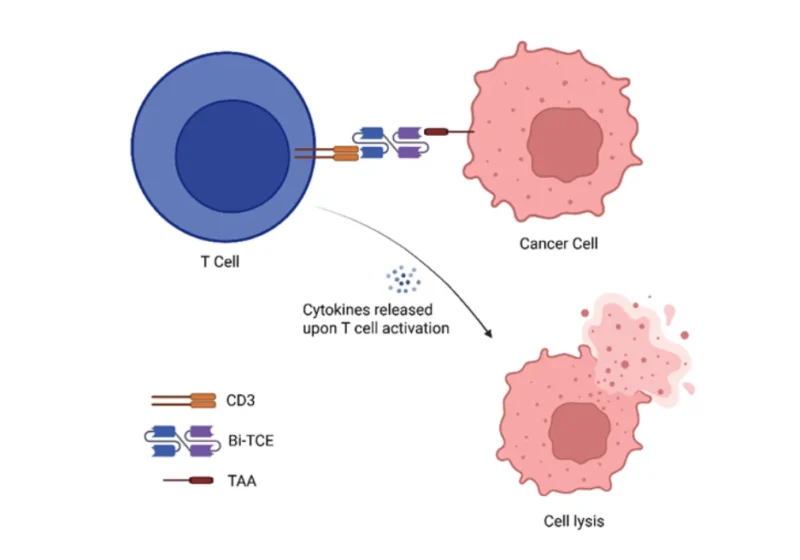
Over the past decade, immunotherapy has transformed oncology, and Bi-TCEs have emerged as a promising “off-the-shelf” alternative to personalized approaches such as CAR-T therapy, with increasing clinical development across both hematologic malignancies and solid tumors.
Approach and data sources
This analysis is based on a comprehensive review of scientific literature, patent data, and clinical trial databases, including the CAS Content Collection and ClinicalTrials.gov. Publication trends, therapeutic targets, and clinical development stages were evaluated to assess the evolution and future direction of Bi-TCE therapies. Mechanistic insights, safety profiles, and emerging engineering strategies were also examined to understand both the potential and limitations of this therapeutic class.
Overall landscape analysis approach
The study represents a large-scale landscape analysis rather than a single clinical trial. It integrates multiple data sources, including over a decade of research publications and patent filings, alongside ongoing and completed clinical trials. The analysis categorizes Bi-TCE development by therapeutic areas, identifies key molecular targets, and evaluates innovation strategies such as half-life extension, dual-targeting, and novel delivery systems. Clinical trial distribution across phases (phase 1 to phase 3) and disease indications was also assessed to understand the maturity of the field.
Results
The analysis demonstrates a significant increase in research activity over the last 10 years, with patent filings accounting for approximately 44% of total documents, reflecting strong commercial interest. Therapeutic development is predominantly focused on solid tumors, including lung, breast, pancreatic, prostate, and ovarian cancers, while hematologic malignancies remain the most clinically advanced indications.
Bi-TCEs have already achieved regulatory success in several hematologic cancers. The first approved agent, blinatumomab, received FDA approval in 2014 for relapsed or refractory B-cell acute lymphoblastic leukemia. Since then, multiple agents targeting CD19, CD20, BCMA, and other antigens have been approved, including therapies for multiple myeloma and diffuse large B-cell lymphoma. More recently, Bi-TCEs have expanded into solid tumors, with agents such as tarlatamab targeting DLL3 in small cell lung cancer.
Despite these advances, limitations remain significant. Classical Bi-TCEs have a short half-life of only a few hours, requiring continuous intravenous infusion. Additionally, treatment is associated with immune-related toxicities, including cytokine release syndrome (CRS) and immune effector cell-associated neurotoxicity syndrome (ICANS), which can be severe and require intensive management.
Clinical trial data reveal that most ongoing studies are in early to mid-stage development, with 122 trials in phase 2, 30 Phase 1/2 trials, 40 in phase 1, and only 28 in phase 3. The majority of trials (196) focus on hematologic malignancies, compared with 33 trials in solid tumors, highlighting the challenges of translating efficacy into solid tumor settings.
Key Findings
Bi-TCEs function as molecular bridges that redirect endogenous T cells to kill cancer cells, offering a unique and highly targeted immunotherapeutic mechanism. Their “off-the-shelf” nature provides a logistical advantage over CAR-T therapies, eliminating the need for patient-specific cell engineering. However, their short half-life, need for repeated administration, and risk of systemic toxicity remain major challenges.
The field is rapidly evolving with several engineering innovations. Half-life extended Bi-TCEs have demonstrated significantly prolonged circulation. in some CD19‑ and BCMA‑targeting HLE Bi‑TCEs reaching up to 210 hours, allowing for less frequent dosing. CD3 tuning and 2:1 molecular formats improve safety by reducing excessive T-cell activation while enhancing tumor selectivity. Dual- and tri-specific engagers are being developed to overcome antigen heterogeneity and prevent tumor escape mechanisms.
Additionally, novel delivery approaches, including gene therapy using adeno-associated viruses and extracellular vesicle systems, are being explored to enable sustained in vivo production and improved tumor targeting. These advancements aim to address key limitations and expand the therapeutic potential of Bi-TCEs, particularly in solid tumors.
Conclusion
Bi-specific T cell engagers are redefining the landscape of cancer immunotherapy by offering a highly targeted, scalable, and innovative approach to tumor eradication. While their clinical success has been most evident in hematologic cancers, ongoing research is rapidly expanding their application into solid tumors and beyond oncology. Despite challenges related to safety, pharmacokinetics, and tumor biology, continuous advancements in engineering and delivery are overcoming these limitations. As the clinical pipeline matures and next-generation constructs enter advanced trials, Bi-TCEs are positioned to become a cornerstone of future cancer treatment strategies, offering new hope for improved patient outcomes worldwide.


