Myelofibrosis (MF) treatment was once defined largely by symptom control and limited therapeutic options. The arrival of JAK inhibitors transformed care by reducing splenomegaly and improving quality of life, however, many patients still experience disease progression or loss of response over time.
Clinicians increasingly face more nuanced questions: which JAK inhibitor or combination strategy best matches the patient’s dominant problem? When should transplantation be reconsidered? Which patients may benefit from emerging approaches? Let’s explore some of the answers through anemia-focused therapies, newer drugs and treatment sequencing options in 2026.
The Essentials of MF
Myelofibrosis (MF) is a Ph-negative myeloproliferative neoplasia that may arise de novo as primary myelofibrosis (PMF) or develop secondary to Essential Thrombocythemia (ET) or Polycythemia Vera (PV). Biologically, MF is characterized by clonal proliferation of malignant hematopoietic stem and progenitor cells, and its pathogenesis has been linked to aberrant cytokine production driven by upregulation of the JAK-STAT signaling pathway. The majority of patients harbor a somatic mutation in one of three genes involved in this pathway: JAK2 Mutation, CALR, or MPL.
Clinical presentation is highly heterogenous and includes:
- Bone marrow fibrosis (BMF)
- Extramedullary hematopoiesis
- Hepatosplenomegaly
- Anemia
- Constitutional symptoms
- Cachexia
- Risk of leukemic transformation
Inferior survival is associated with older age, anemia, leukocytosis, thrombocytopenia, the presence of constitutional symptoms, and transfusion dependence (TD). Prognosis is further influenced by cytogenetic abnormalities and high- risk mutations (ASXL1, SRSF2, U2AF1‐Q157, EZH2, IDH1/2).
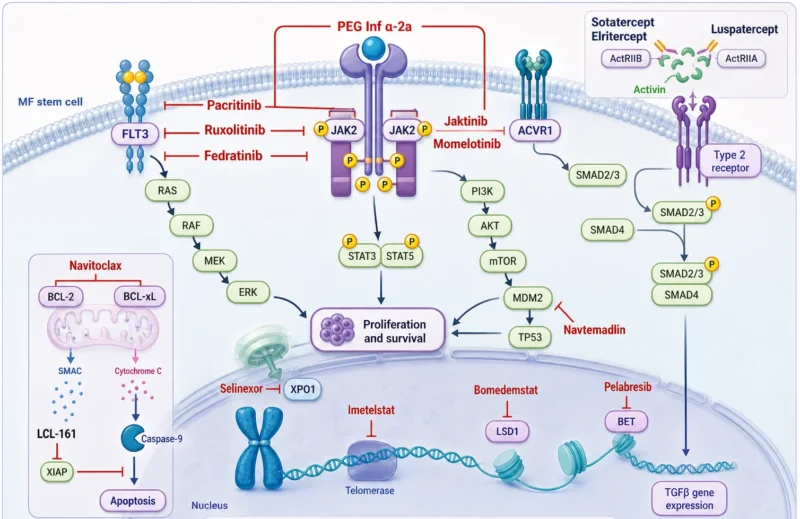
Management is typically risk‐adapted, being guided by disease risk scores category (IPSS, DIPSS+, MIPSS70 etc.), symptoms and medical fitness. JAK inhibitors transformed landscape of intermediate- to high-risk MF, beginning with Ruxolitinib in 2011, followed by Fedratinib (2019), Pacritinib (2022), and Momelotinib (2023). Yet JAK inhibitors have shown limited disease-modifying effects and are not known to prevent leukemic transformation.
If Prior JAK Therapy Failed
Ruxolitinib is commonly considered in first-line settings for MF, as it is the most established JAK inhibitor, supported by a large body of data. In clinical practice, however, many patients experience treatment-limiting cytopenias or suboptimal responses. At least 40% of patients relapse or become refractory to ruxolitinib within a few years of initiating therapy.
Yet “JAK inhibitor failure” is biologically heterogeneous, ranging from primary refractory disease to progressive symptomatic decline. Recognizing these distinctions is critical, as the optimal next step may differ substantially between switching to Fedratinib, selecting a cytopenia-adapted agent, enrollment in a clinical trial, or reassessing candidacy for allo-SCT.
The RR6 prognostic model comprises dose, spleen response, and transfusion requirements to help identify patients who may benefit from an early therapy switch. A recent U.S.-based consensus broadly defines treatment failure as a lack of symptom or spleen improvement, or progression/intolerance after at least 3 months of therapy at the highest tolerated dose (Coltoff, A., et al., Chan T.W. et al., 2025).
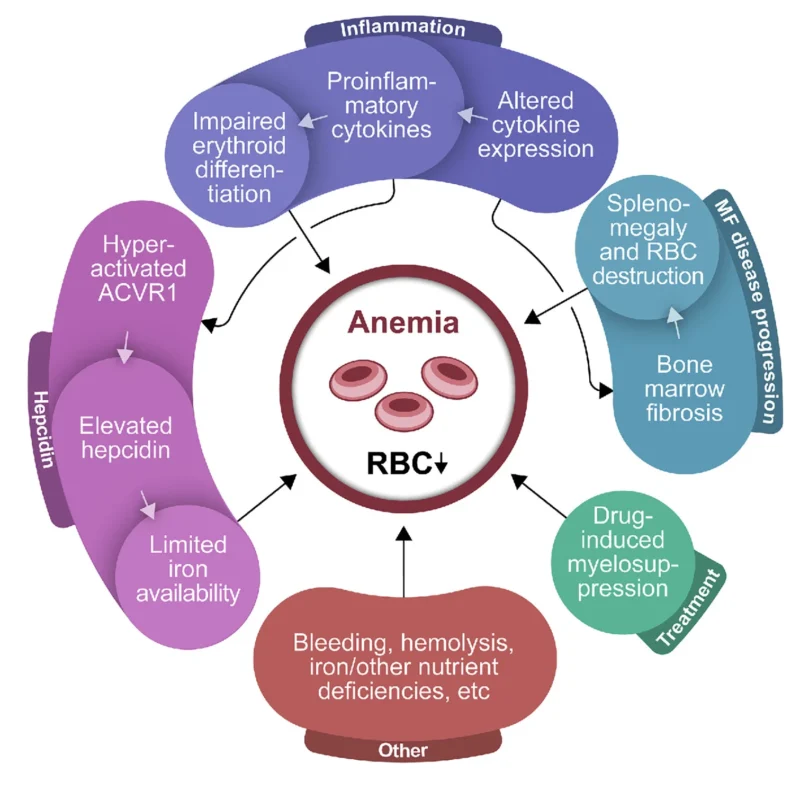
If Anemia Is the Main Problem
Anemia, which affects an estimated 38% of patients at MF diagnosis and 58% within 1 year, is driven by diverse mechanisms and can present a significant burden.
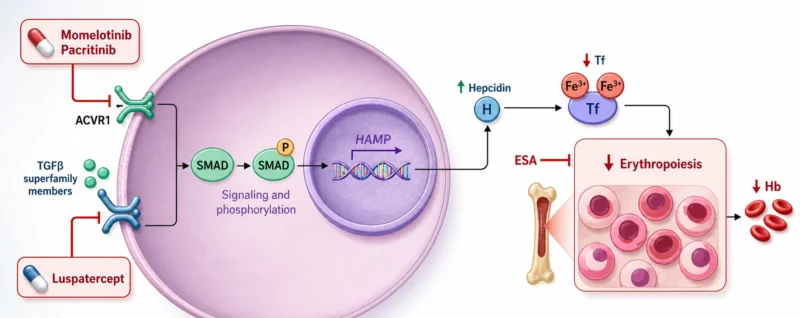
Additional biologic activity beyond JAK blockade, particularly ACVR1 inhibition with downstream reduction in hepcidin and improved erythropoiesis, has positioned Momelotinib as a valuable option.
haematologica.org
According to data from the SIMPLIFY-1 trial, among patients with moderate to severe anemia,72% of patients receiving momelotinib maintained transfusion independence through week 24. In patients with baseline platelet counts <200×10⁹/L, response rates also favored momelotinib (over ruxolitinib), with spleen responses of 39% , transfusion independence rates of 51%, and achievement of both endpoints in 33% of patients.
Off-label anemia-directed therapies in MF include erythropoiesis-stimulating agents (ESA), Danazol, and Luspatercept. In two small studies, ESA produced response rates of 40%-60%, particularly in patients with low serum erythropoietin levels (<125 IU/L) and higher baseline hemoglobin. Danazol has shown response rates of approximately 30%, with better outcomes observed in transfusion-independent patients.
Luspatercept is an activin receptor ligand trap that binds selected TGF-β superfamily ligands, thereby reducing aberrant SMAD2/3 signaling and promoting late-stage erythroid maturation. Phase II data showed more than 50% reduction in transfusion burden when added to stable JAK inhibitor therapy, and the ongoing phase III trial is evaluating its role in transfusion-dependent patients (Vachhani et al., 2026).
If Platelets Are Very Low
Pacritinib is an important option for severe thrombocytopenia. In patients with baseline platelets <50×10⁹/L, a retrospective pooled analysis of the PERSIST trials favored pacritinib over best available therapy, with ≥35% spleen volume reduction in 23.1% and ≥50% symptom-score reduction in 25% at week 24. Hemoglobin and platelet counts remained generally stable through week 24, without additional myelosuppression.
Immunomodulatory agents (IMiDs) remain an option in selected patients. Lenalidomide showed anemia responses in 22% and platelet improvement in 50% of patients in pooled phase 2 data. Thalidomide reduced transfusion burden in 29% of patients with moderate/severe anemia and improved thrombocytopenia in 38%. In phase 3 evaluation, pomalidomide did not significantly improve transfusion independence versus placebo, but showed greater activity in thrombocytopenia (S. B. Reynolds et al., 2025).
The Need for Standardized Endpoints
Cross-trial comparison of anemia outcomes remains challenging because studies use nonuniform endpoints. Definitions of transfusion independence and anemia response often differ by baseline transfusion status and hemoglobin thresholds, making the relative benefit of available therapies harder to interpret.
This is further complicated by the lack of direct head-to-head comparisons between JAK inhibitors with recognized anemia-related benefits. As a result, treatment selection often relies on indirect evidence, differences in trial design, and individual patient phenotype rather than definitive comparative data.
The recently updated International Working Group–European LeukemiaNet criteria may help address this gap by introducing a more standardized definition of anemia response: absence of transfusions together with an average hemoglobin increase of ≥1.5 g/dL over any rolling 12-week period. If widely adopted, this framework could bring needed consistency.
Safety Profile
While the class-wide safety profile is well characterized, each agent has distinct individual considerations. In practice, optimal JAK inhibitor selection often means choosing the agent whose toxicity profile best matches the patient’s comorbidities and treatment priorities.
Ruxolitinib most commonly causes cytopenias, particularly early in treatment. Infections (including herpes zoster), weight gain, dyslipidemia, cardiac events, and non-melanoma skin cancers may also occur. Fedratinib can also cause anemia and thrombocytopenia, but is especially associated with gastrointestinal toxicity, elevated liver enzymes, or creatinine. Its key rare toxicity is Wernicke encephalopathy related to thiamine deficiency.
Pacritinib commonly causes gastrointestinal toxicity. Bleeding risk, QT prolongation, thrombosis, and cardiac events should also be considered. Momelotinib may improve anemia but can still cause thrombocytopenia or neutropenia. Non-hematologic effects include gastrointestinal toxicity, dizziness, hypotension, hepatotoxicity, and peripheral neuropathy. Secondary malignancies have been reported across JAK inhibitor therapy and should be considered during long-term monitoring.
What Comes Next in MF Therapy
Increasing attention is being directed toward true disease modification. Improvements in BMF grade, mutation burden, and long-term survival are emerging endpoints.
A systematic review of PubMed, ClinicalTrials.gov, and major hematology conference abstracts (2013-2024) was conducted to identify emerging Myelofibrosis therapies, with shortlisted agents selected based on positive phase II data, adequate enrollment, recent trial completion, and likelihood of regulatory advancement (Chan T.W. et al., 2025).
The search strategy identified 48 molecules, of which 16 met selection criteria: four approved JAK inhibitors, seven therapies in phase III development, and five in phase II.
Jaktinib, a pan-JAK/ACVR1 inhibitor, has shown encouraging interim phase III results versus hydroxyurea with fewer hematologic toxicities, including lower rates of anemia (25.5% and thrombocytopenia (17.0%). Navtemadlin (oral, selective MDM2 inhibitor for TP53 wild-type MF) demonstrated improved week-24 outcomes over best available therapy in patients relapsed or refractory to ruxolitinib, with biomarker reductions suggesting possible disease-modifying activity.
Imetelstat, a first‐in‐class telomerase inhibitor, has generated particular interest after phase II data showed median OS of 29.9 months, with matched comparisons suggesting survival improvement vs best available therapy, the phase III IMpactMF interim analysis is anticipated in the second half of 2026.
CALR-directed therapies are emerging as a mutation-specific approach. INCA33989, a monoclonal antibody targeting mutant CALR-positive CD34+ cells, has shown preclinical potential to reduce megakaryocyte accumulation and suppress self-renewal of malignant stem/progenitor cells. Early clinical trials are underway to develop bispecific antibodies that bind CD3 and mutant CALR cells, redirecting T cells against the malignant clone.
The Rise of Rational Combinations
Interim phase 3 MANIFEST-2 data showed that pelabresib (bromodomain inhibitor) plus ruxolitinib improved clinical outcomes with good tolerability vs ruxolitinib alone in JAK inhibitor-naïve patients with intermediate-1 or higher-risk MF, supporting a potential shift toward upfront combination therapy.
Navitoclax (Bcl‐2 inhibitor) plus ruxolitinib improved responses in the phase III TRANSFORM-1 trial, though thrombocytopenia and anemia remain notable adverse events. Selinexor (an XPO1 inhibitor that selectively induces apoptosis) combined with ruxolitinib has shown promising early efficacy and tolerability, while phase III evaluation is ongoing.
Among phase II therapies for MF, several agents are focused on anemia and disease modification. LCL161 (SMAC mimetic), studied in an older ruxolitinib-resistant population, showed median OS of 34 months, with anemia responses in 12% of patients. Sotatercept (ActR type IIA ligand trap) of 32% when combined with stable Ruxolitinib.
Elritercept has shown anemia-related activity in the ongoing RESTORE trial, with improved hemoglobin levels, reduced transfusion burden, and stable platelet counts. Among heavily transfusion-dependent patients, 73% achieved at least a 50% reduction in transfusion needs, and 45% reached transfusion independence when combined with ruxolitinib.
Bomedemstat (an LSD‐1 inhibitor), is being explored as a second-line strategy. Preliminary phase 2 data of bomedemstat plus ruxolitinib showed clinical activity in both patients with suboptimal ruxolitinib response and treatment-naïve MF, with spleen responses at week 24 of 7.4% and 38.5%, symptom responses of 25.9% and 30.7%, and stable or improved hemoglobin in about half of patients.
In the COMBI study, pegylated interferon alfa-2a plus ruxolitinib was associated with reductions in bone marrow cellularity, fibrosis, mutation burden, and symptom burden, with acceptable tolerability.
Imetelstat, pelabresib, navitoclax, and bomedemstat have shown fibrosis reduction, but their clinical significance is still uncertain. In contrast, SIMPLIFY-1 reported fibrosis improvement in about 20% of patients treated with ruxolitinib or momelotinib, without a clear survival benefit (S. B. Reynolds et al., Naseema Gangat 2025, Palandri, F., et al., 2026)
You Can Watch More on OncoDaily TV
Beyond JAK Inhibitors: Transplant and Supportive Treatment Strategies
Although MF treatment can be curative only with HSCT, its risk‐benefit ratio is such that it is reserved for carefully selected patients.
HSCT should be considered for all patients with PMF and an intermediate-2- or high-risk DIPSS score, a high-risk MIPSS70 /70+ v.2.0 score, and a low- or intermediate-risk Myelofibrosis Transplant Scoring System score.
In patients who are awaiting HCST, peritransplant JAK inhibition appears to be safe and advantageous, most studies in this setting focus on ruxolitinib and further evidence is needed for other JAKi.
Hydroxyurea retains a practical role in MF, mainly for patients with a proliferative phenotype characterized by leukocytosis, thrombocytosis, or modest splenomegaly, particularly when rapid cytoreduction is needed or JAK inhibitors are unsuitable. Responses are often partial and limited by worsening anemia or other cytopenias. In modern practice, it is used mainly as a supportive or bridging therapy rather than a long-term disease-modifying option.
Splenectomy is generally reserved for carefully selected patients, mainly for massive symptomatic splenomegaly refractory to medical therapy, severe splenic pain, portal hypertension, recurrent splenic infarction, or profound transfusion needs, often as a bridge to allogeneic transplant or when no medical alternative remains (Vachhani, P., et al., Masarova, L., et al., 2026)
Long story short
1. Allo-SCT remains the only curative therapy and should be considered early in eligible patients with higher-risk disease.
2. In transplant candidates, peritransplant JAK inhibition – particularly with ruxolitinib – appears feasible and may help optimize patients before HSCT.
3. Ruxolitinib remains a common first-line choice for reducing splenomegaly and symptom burden.
4. In cytopenic/myelodepletive myelofibrosis, pacritinib and momelotinib are important options because of their relative advantages in thrombocytopenia and anemia.
5. Genomic features such as ASXL1 and other high-risk mutations refine prognosis and may increasingly guide treatment selection
6. JAK inhibitor tapering or switching should be done carefully, with monitoring for withdrawal symptoms and early response loss.
7. Skin cancer surveillance and broader long-term safety monitoring are prudent during JAK inhibitor therapy.
You can also read: Myelodysplastic Neoplasms (MDS): From Ineffective Hematopoiesis to Sideroblastic Phenotype
Written by Susanna Mikayelyan, MD
FAQ
Can Myelofibrosis Turn Into Acute Leukemia?
Yes. A proportion of patients with myelofibrosis may progress to blast-phase disease or acute myeloid leukemia. This is one of the most serious complications and often reflects aggressive underlying biology.
Why Is Anemia So Common in Myelofibrosis?
Anemia can result from bone marrow fibrosis, chronic inflammation, enlarged spleen activity, nutritional factors, and treatment-related suppression of blood cell production.
Is Myelofibrosis Always Inherited?
No. Most cases are acquired during life through somatic mutations such as JAK2, CALR, or MPL, rather than inherited directly from parents.
What Is the Difference Between Primary and Secondary Myelofibrosis?
Primary myelofibrosis develops on its own, while secondary myelofibrosis evolves from another myeloproliferative neoplasm such as polycythemia vera or essential thrombocythemia.
Are JAK Inhibitors Chemotherapy?
Not in the traditional sense. JAK inhibitors are targeted therapies that block abnormal signaling pathways rather than broadly attacking rapidly dividing cells.
What Does the Future of Myelofibrosis Treatment Look Like?
The field is moving toward combination therapies, anemia-focused treatments, fibrosis-modifying agents, mutation-targeted approaches, and more personalized treatment sequencing.