Lurbinectedin has emerged as an important therapeutic option in small cell lung cancer (SCLC), a disease characterized by rapid proliferation, early dissemination, and high relapse rates after first-line therapy. Despite initial sensitivity to platinum-based chemotherapy, most patients experience disease progression within months, and outcomes in the relapsed setting remain poor.
Approved by the U.S. Food and Drug Administration (FDA) in 2020 for metastatic SCLC with disease progression on or after platinum-based chemotherapy, lurbinectedin represents a mechanistically distinct transcription inhibitor derived from marine compounds. Its development reflects a broader resurgence of DNA-interacting agents with targeted molecular effects beyond conventional cytotoxicity.
This review examines the pharmacology, clinical trial data, safety profile, resistance considerations, and emerging research directions for lurbinectedin, with emphasis on evidence from peer-reviewed clinical studies.
Read About Small Cell Lung Cancer on OncoDaily
Mechanism of Action
Lurbinectedin (PM1183) is a synthetic tetrahydroisoquinoline alkaloid structurally related to trabectedin. It binds selectively to guanine residues in the minor groove of DNA, particularly within CG-rich sequences, leading to disruption of oncogenic transcription programs (Leal et al., 2010, Molecular Cancer Therapeutics).
Unlike traditional alkylating agents, lurbinectedin interferes with RNA polymerase II activity, inducing DNA strand breaks and promoting apoptosis in transcriptionally active tumor cells. Additionally, it modulates the tumor microenvironment by reducing tumor-associated macrophages and inflammatory mediators, which may contribute to antitumor activity (Santamaria Nuñez et al., 2016, Clinical Cancer Research).
This dual effect, direct tumor cytotoxicity and microenvironmental modulation—distinguishes lurbinectedin from conventional cytotoxic chemotherapy.
Clinical Development in Small Cell Lung Cancer
Phase II Basket Trial (PM1183-B-005-14)
The pivotal phase II single-arm trial evaluated lurbinectedin in patients with relapsed SCLC after prior platinum-based chemotherapy (Trigo et al., 2020, The Lancet Oncology).
In this study of 105 patients receiving lurbinectedin 3.2 mg/m² every three weeks:
- Overall response rate (ORR): 35.2%
- Median progression-free survival (PFS): 3.5 months
- Median overall survival (OS): 9.3 months
Among patients with platinum-sensitive relapse (chemotherapy-free interval ≥90 days), the ORR was 45%, compared with 22% in platinum-resistant disease.
These results compared favorably to historical outcomes with topotecan, which demonstrated ORR ~16–24% and median OS approximately 6–8 months in prior studies (von Pawel et al., 1999, JCO).
Based on this trial, the FDA granted accelerated approval for lurbinectedin in June 2020.
Comparative Efficacy: Lurbinectedin vs Topotecan
Topotecan has long been the only FDA-approved second-line therapy for SCLC. However, its toxicity profile and modest efficacy have limited its clinical utility.
In a randomized phase III trial, topotecan demonstrated median OS of 6.5 months compared with CAV (cyclophosphamide, doxorubicin, vincristine) (von Pawel et al., 1999, JCO).
Although no head-to-head phase III comparison between lurbinectedin and topotecan exists, indirect comparisons suggest higher response rates and improved tolerability with lurbinectedin, particularly in platinum-sensitive relapse.
However, cross-trial comparisons must be interpreted cautiously due to differences in patient populations and trial designs.
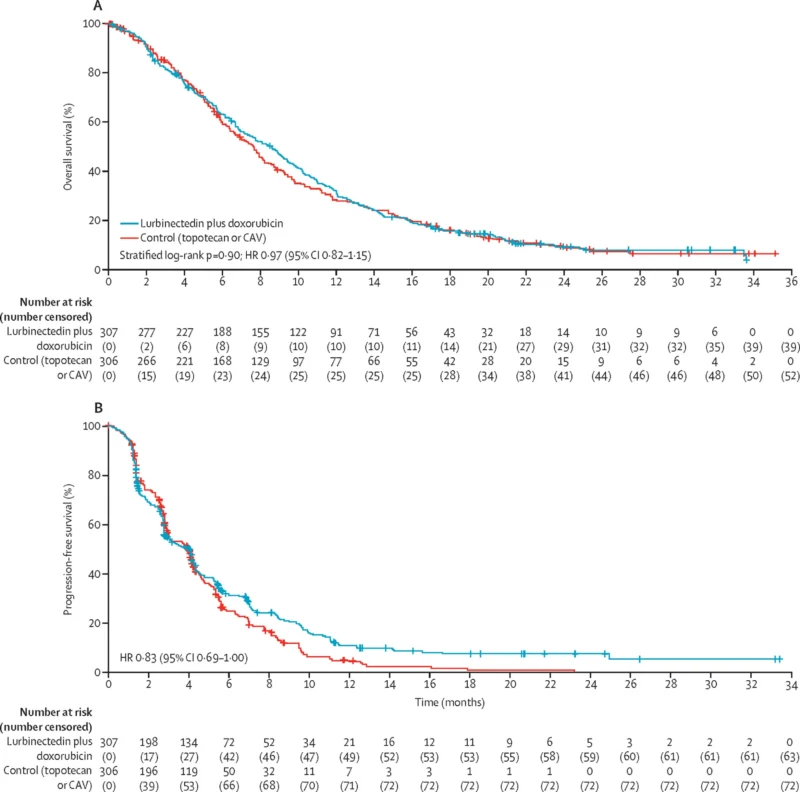
ATLANTIS Trial
The phase III ATLANTIS trial evaluated lurbinectedin in combination with doxorubicin versus physician’s choice (topotecan or CAV) in relapsed SCLC (Paz-Ares et al., 2021, JCO).
The combination regimen did not meet its primary endpoint of overall survival superiority. Median OS was 8.6 months in the lurbinectedin combination arm versus 7.6 months in the control arm, which was not statistically significant.
Importantly, the ATLANTIS trial used a lower lurbinectedin dose (2.0 mg/m²) than the approved single-agent dose (3.2 mg/m²), potentially influencing efficacy outcomes.
Despite the negative primary endpoint, ATLANTIS reinforced lurbinectedin’s manageable safety profile and suggested ongoing activity in platinum-sensitive subgroups.
Safety Profile
Across trials, lurbinectedin demonstrates a predictable and manageable toxicity profile.
The most common grade 3–4 adverse events in the phase II study included:
- Neutropenia (~46%)
- Leukopenia
- Anemia
- Thrombocytopenia
Febrile neutropenia occurred in approximately 5% of patients (Trigo et al., 2020). Non-hematologic toxicities were generally mild, including fatigue, nausea, and transaminase elevation.
Compared with topotecan, lurbinectedin appears associated with less severe thrombocytopenia and fewer treatment discontinuations due to toxicity. Primary prophylaxis with granulocyte colony-stimulating factor (G-CSF) may be considered in selected high-risk patients.
Role in Current Clinical Practice
Lurbinectedin is currently indicated for metastatic SCLC with progression after platinum-based therapy.
In platinum-sensitive relapse (chemotherapy-free interval ≥90 days), rechallenge with platinum doublet remains an option (O’Brien et al., 2006, JCO). However, lurbinectedin offers a non-platinum alternative with meaningful response rates.
In platinum-resistant disease, therapeutic options are limited, and lurbinectedin provides an evidence-supported second-line approach.
The optimal sequencing relative to topotecan, immunotherapy rechallenge, and emerging agents remains an area of ongoing investigation.
Biomarkers and Predictive Factors
To date, no validated predictive biomarkers for lurbinectedin response have been established.
Preclinical studies suggest that tumors with high transcriptional activity and DNA repair deficiencies may be particularly sensitive (Santamaria Nuñez et al., 2016).
Exploratory analyses of transcription factor dependencies in SCLC subtypes (ASCL1, NEUROD1, POU2F3, YAP1) may inform future patient selection strategies (Rudin et al., 2019, Nature Reviews Cancer).
Further translational studies are needed to clarify molecular predictors of response.
Combination Strategies
Given its transcriptional inhibition mechanism, lurbinectedin has been explored in combination with immunotherapy and cytotoxic agents.
Preclinical data suggest that transcription inhibition may enhance immunogenic cell death, potentially synergizing with immune checkpoint blockade.
Clinical trials evaluating lurbinectedin in combination with atezolizumab and other immunotherapies are ongoing. Combination approaches aim to address the aggressive biology of SCLC, which remains characterized by rapid resistance and limited durable responses.
Beyond Small Cell Lung Cancer
Lurbinectedin has also demonstrated activity in other malignancies.
In BRCA-mutated and platinum-resistant ovarian cancer, early-phase trials suggested antitumor activity (Leary et al., 2021, Clinical Cancer Research).
However, its primary clinical development remains focused on SCLC, where unmet need is greatest.
Resistance Mechanisms
Resistance to lurbinectedin likely involves enhanced DNA repair capacity and alterations in transcriptional regulation.
Upregulation of nucleotide excision repair pathways may attenuate drug-induced DNA damage. Understanding resistance biology will be critical in optimizing combination and sequencing strategies.

Read About Lung Cancer Cure Rate on OncoDaily
Future Directions
Key areas of investigation include:
- Optimizing sequencing in relapsed SCLC
- Identifying predictive biomarkers
- Exploring earlier-line integration
- Combining with immunotherapy
- Investigating activity in molecularly defined SCLC subtypes
While lurbinectedin is not curative in relapsed SCLC, it represents a meaningful advance in a historically stagnant therapeutic landscape.
Conclusion
Lurbinectedin is a mechanistically unique transcription inhibitor with demonstrated clinical activity in relapsed small cell lung cancer. Supported by phase II evidence and regulatory approval, it offers an important therapeutic option in a setting with limited alternatives.
Although the ATLANTIS trial did not confirm superiority in combination therapy, single-agent lurbinectedin at 3.2 mg/m² remains a standard option in platinum-pretreated SCLC.
Ongoing research into biomarkers, resistance mechanisms, and rational combinations will determine its ultimate positioning within the evolving SCLC treatment paradigm.
For clinicians managing relapsed SCLC, lurbinectedin represents a valuable addition grounded in mechanistic rationale and validated clinical activity.
Written by Armen Gevorgyan, MD


