Dordaviprone (Modeyso) is an emerging therapeutic agent that has generated considerable interest within the oncology field due to its novel mechanism and potential to address unmet needs in cancer care. Ongoing research continues to define its role across different tumor types, with early clinical data highlighting its promise in difficult-to-treat malignancies.
This article will examine Dordaviprone’s therapeutic uses in oncology, its safety profile and potential side effects, recommended dosage considerations, and key expectations for patients and clinicians.
Which Company produced Dordaviprone?
Dordaviprone (Modeyso) was originally developed by Oncoceutics, a biotechnology company focused on novel cancer therapeutics. In 2021, Chimerix, Inc. acquired the ONC201 program, bringing dordaviprone into its oncology pipeline and advancing its clinical development. Under Chimerix’s leadership, the drug progressed through multiple trials, earning regulatory designations such as Orphan Drug, Fast Track, and Rare Pediatric Disease status for the treatment of recurrent H3 K27M-mutant diffuse glioma.
In April 2025, Jazz Pharmaceuticals completed the acquisition of Chimerix, incorporating dordaviprone into its oncology portfolio. This strategic move provided the drug with expanded development resources and global commercial infrastructure. The transition marked a collaborative effort between the Chimerix and Jazz teams, aiming to accelerate the drug’s clinical progress and bring it to patients sooner.
Through these successive partnerships—first between Oncoceutics and Chimerix, and now with Jazz Pharmaceuticals—dordaviprone has moved from an early-stage experimental therapy to a near-commercial product backed by robust scientific and corporate support.
How does Dordaviprone work?
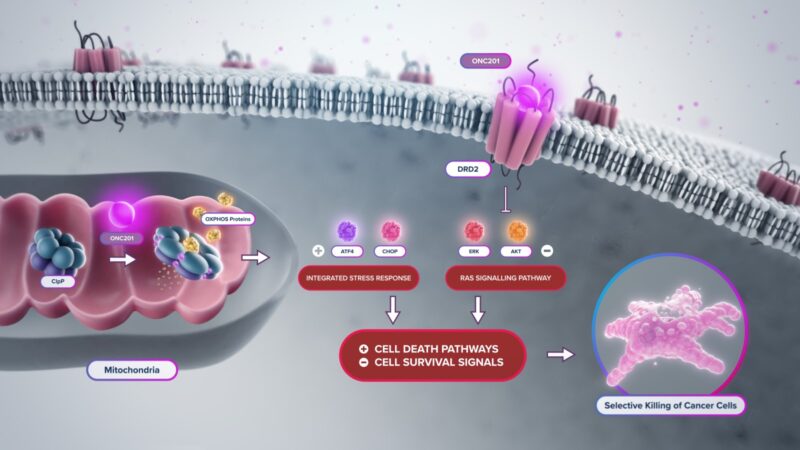
According to the official prescribing information and company data, Modeyso is a first-in-class small-molecule imipridone that acts through a dual, tumor-targeted mechanism. The drug selectively binds to and hyperactivates the mitochondrial caseinolytic protease P (ClpP), leading to uncontrolled proteolysis within the mitochondria. This disrupts oxidative phosphorylation, induces mitochondrial stress, and triggers the integrated stress response—effects that are particularly detrimental to tumor cells with high mitochondrial dependence.
In parallel, dordaviprone functions as a potent antagonist of the dopamine receptor D2 (DRD2), which is aberrantly expressed in several malignancies, including H3 K27M-mutant diffuse midline gliomas. DRD2 blockade interferes with key oncogenic signaling cascades, such as the PI3K/AKT and MAPK pathways, further promoting apoptosis.
The combination of mitochondrial proteotoxic stress and disruption of pro-survival GPCR signaling results in selective cytotoxicity in tumor cells while minimizing effects on normal tissue. This dual mechanism forms the scientific basis for the drug’s clinical activity in aggressive, treatment-resistant tumors.
Source: Chimerix Official Website, a part of Jazz Pharmaceuticals
What Cancers Is Dordaviprone Approved to Treat?
On August 6, 2025, Dordaviprone received accelerated approval from the U.S. Food and Drug Administration for the treatment of recurrent H3 K27M-mutant diffuse midline glioma in adult and pediatric patients aged one year and older whose disease has progressed following prior therapy. This represents a landmark development, as it is the first systemic therapy authorized for this aggressive and ultra-rare form of brain cancer. The decision was supported by clinical evidence from multiple open-label studies, reflecting years of collaborative research and development.
What research is behind the approval?
The FDA’s accelerated approval of Modeyso was based on an integrated analysis of 50 patients enrolled in five early-phase, open-label clinical trials in the U.S. These studies included adult and pediatric patients with progressive, measurable H3 K27M-mutant diffuse midline glioma who had received prior radiation and showed no cerebrospinal fluid dissemination.
The trials—ONC006, ONC013, ONC014, ONC016, and ONC018—evaluated safety, dosing, pharmacokinetics, and antitumor activity. ONC006 identified the recommended adult dose, ONC013 assessed early efficacy in mixed-age populations, ONC014 focused on pediatric dosing, ONC016 provided primary efficacy data via blinded independent review using RANO 2.0 criteria, and ONC018 monitored long-term response and safety.
Eligible patients had adequate performance status and were either off corticosteroids or tapering. Patients with DIPG, spinal cord gliomas, CSF dissemination, or atypical histologies were excluded.
Efficacy Results
These trials showed a 22% overall response rate (95% CI: 12–36), with a median duration of response of 10.3 months (95% CI: 7.3–15.2). Among responders, 73% maintained their response for at least six months, and 27% for over a year, demonstrating durable clinical benefit in this difficult-to-treat population.

Read more about FDA’s Accelerated Approval to Dordaviprone for H3 K27M-Mutant Diffuse Midline Glioma on Oncodaily.
Dordaviprone side effects and its management
As with any anticancer therapy, treatment with Modeyso is associated with a spectrum of adverse effects. Understanding the safety profile is essential for healthcare providers to optimize patient care, anticipate complications, and implement timely interventions that can maintain quality of life and adherence to therapy. While many patients tolerate dordaviprone well, clinicians should be vigilant for both common and less frequent toxicities, some of which may require dose modification or supportive care measures.
Common Side Effects
The most frequently reported adverse reactions in clinical trials included gastrointestinal symptoms such as nausea, vomiting, and diarrhea. Fatigue was also commonly observed, likely reflecting both the underlying disease and treatment effects. Headache and elevated liver enzymes (transaminases) occurred in a subset of patients and warrant routine monitoring. Mild to moderate hematologic abnormalities such as anemia and thrombocytopenia have been reported but are generally manageable.
Less Common Side Effects
Less frequent but clinically significant side effects include hypersensitivity reactions, which may present as rash, pruritus, or, rarely, more severe manifestations such as angioedema. QTc interval prolongation has been observed and necessitates careful electrocardiographic monitoring in patients with cardiac risk factors or those on concomitant QT-prolonging medications. Embryo-fetal toxicity is a critical concern, emphasizing the need for effective contraception and reproductive counseling for patients of childbearing potential.
Management of Side Effects
Management strategies focus on early recognition and supportive interventions. For gastrointestinal symptoms, prophylactic or therapeutic antiemetics and hydration are recommended. Liver function tests should be regularly monitored, and dose adjustments considered if significant elevations occur. Cardiac monitoring with ECGs is advised for at-risk patients, and drugs that prolong QTc should be avoided when possible. Hypersensitivity reactions may require antihistamines, corticosteroids, or discontinuation of therapy depending on severity.
Patients should receive counseling on reproductive risks and be advised on contraception during treatment and for an appropriate duration after discontinuation. Multidisciplinary collaboration among oncologists, cardiologists, and supportive care teams is essential to optimize safety and maintain treatment continuity.

What is the Recommended Dosage of Dordaviprone?
Dordaviprone is available as 125 mg capsules and is indicated for the treatment of diffuse midline glioma harboring the H3 K27M mutation in adults with progressive disease after prior therapy. The recommended dose is 625 mg orally once weekly and should be continued until disease progression or unacceptable toxicity occurs.
Treatment should be paused for suspected hypersensitivity or significant QTc prolongation and permanently discontinued for severe reactions or serious arrhythmias. For other severe toxicities, hold therapy until improvement and resume at a lower dose; recurrent severe events warrant stopping treatment.
How is Dordaviprone Administered?
Dordaviprone is administered orally once weekly on an empty stomach, at least one hour before or three hours after food, preferably at the same time each week. Capsules should be swallowed whole; however, for patients unable to swallow capsules, the contents may be dispersed in 15–30 mL of an appropriate liquid. The mixture should be gently swirled until uniform and administered promptly, within two hours of preparation, ensuring the entire dose is consumed.
For feeding tube administration, the same liquid preparation is used. The solution is drawn into a syringe and delivered slowly via the feeding tube, with flushing before and after dosing according to institutional protocols. In case of a missed dose within two days, administration should occur as soon as possible; if more than two days have elapsed, the dose should be skipped, resuming the regular schedule. If vomiting occurs post-dose, the next scheduled dose should be taken without repeating the missed dose.
Capsules should be stored at controlled room temperature (20–25ºC). Prepared liquid formulations should be discarded if not used within two hours.
What to Avoid During Dordaviprone Treatment?
During treatment with dordaviprone, several precautions are necessary to minimize risks and ensure patient safety.
- Avoid Concomitant Use of Strong or Moderate CYP3A4 Inhibitors – Dordaviprone is metabolized primarily via CYP3A4; coadministration with strong or moderate CYP3A4 inhibitors can significantly increase drug exposure, raising the risk of toxicity. Unless absolutely necessary, such combinations should be avoided. If unavoidable, dose reductions of dordaviprone are recommended, and patients should be closely monitored.
- Avoid Other QTc-Prolonging Agents – Due to the risk of QT interval prolongation, dordaviprone should not be used concurrently with medications known to prolong QTc. Careful cardiac monitoring is advised when any QTc-prolonging drugs cannot be avoided.
- Avoid Grapefruit and Grapefruit Juice – Grapefruit products can inhibit CYP3A4 and increase dordaviprone plasma levels, potentially exacerbating side effects. Patients should be counseled to avoid grapefruit and related citrus products during treatment.
- Avoid Pregnancy – Dordaviprone carries a risk of embryo-fetal toxicity. Effective contraception is essential for patients of reproductive potential during treatment and for an appropriate duration afterward. Pregnancy testing should be performed prior to therapy initiation.
- Avoid Abrupt Discontinuation Without Medical Advice – Stopping treatment suddenly without physician guidance may lead to disease progression or loss of therapeutic benefit. Any treatment modifications should be coordinated with the oncology care team.

Read more about Glioblastoma: Causes, Symptoms, Diagnosis, Treatment, and Latest Advances in 2025 on OncoDaily.
How Long Does the Drug Stay in the Body?
Dordaviprone is primarily metabolized in the liver through the cytochrome P450 enzyme CYP3A4, with minor contributions from several other CYP enzymes including CYP2B6, CYP2C8, CYP2C9, CYP2D6, and CYP3A5. This metabolic profile underlines the potential for drug-drug interactions, especially with strong CYP3A4 inhibitors or inducers.
The drug has an elimination half-life of approximately 11 hours, allowing for once-weekly dosing. Dordaviprone is cleared at a rate of about 27 liters per hour. Excretion occurs predominantly via the kidneys, with approximately 70% of the administered dose eliminated in urine.
Dordaviprone effectiveness over time
Dordaviprone has demonstrated durable clinical benefit in patients with recurrent H3 K27M-mutant diffuse midline glioma, a disease with historically poor prognosis and limited treatment options. Clinical trial data indicate that among patients who respond to therapy, many experience sustained tumor control extending beyond six months, with a subset maintaining responses for over a year. This durability suggests that dordaviprone can provide meaningful disease stabilization and improve clinical outcomes in a patient population with aggressive, treatment-refractory tumors.
Ongoing trials with Dordaviprone
The ACTION study is a global, randomized, double-blind Phase 3 trial evaluating dordaviprone (ONC201) versus placebo in patients with newly diagnosed H3 K27M-mutant diffuse glioma after standard radiotherapy. It aims to assess whether ONC201 improves survival outcomes in this aggressive tumor.
The ONC201 trial for refractory meningioma (NCT06012929) is currently suspended pending protocol revisions and regulatory approvals expected by April 2025. This University of Nebraska–sponsored pilot study evaluates ONC201 in patients undergoing meningioma surgery and those with refractory disease after standard treatments. Patients receive 625 mg orally once weekly, with pre-surgical patients given two doses before surgery and refractory patients continuing until progression or toxicity.
Primary goals include measuring drug levels in tumor tissue and progression-free survival; secondary goals assess tumor response, receptor expression, overall survival, and combination therapy with bevacizumab.
FAQ
How does Modeyso work against cancer?
Dordaviprone works through a dual mechanism: it hyperactivates the mitochondrial protease ClpP, causing stress that kills tumor cells, and blocks the dopamine receptor D2 (DRD2), disrupting survival pathways. This makes cancer cells more vulnerable while sparing healthy tissue.
What types of cancer is Modeyso used for?
As of 2025, dordaviprone is FDA-approved for recurrent H3 K27M-mutant diffuse midline glioma in adults and children one year and older.
How is Modeyso different from chemotherapy?
Unlike traditional chemotherapy, which indiscriminately attacks dividing cells, dordaviprone works through a targeted dual mechanism.
How should Dordaviprone be taken?
Modeyso should be taken on an empty stomach, at least one hour before or three hours after food.
What are the most common side effects of Dordaviprone?
The most frequently reported side effects include nausea, vomiting, diarrhea, fatigue, headache, and elevated liver enzymes.
What is the recommended dosage of Modeyso?
The standard recommended dose is 625 mg orally once weekly.
What is the half-life of Modeyso?
Modeyso has a half-life of about 11 hours, which allows for once-weekly dosing.


