Rahul Gosain, Director of Regional Infusion Services and Medical Director at Wilmot Cancer Institute, shared a post on LinkedIn:
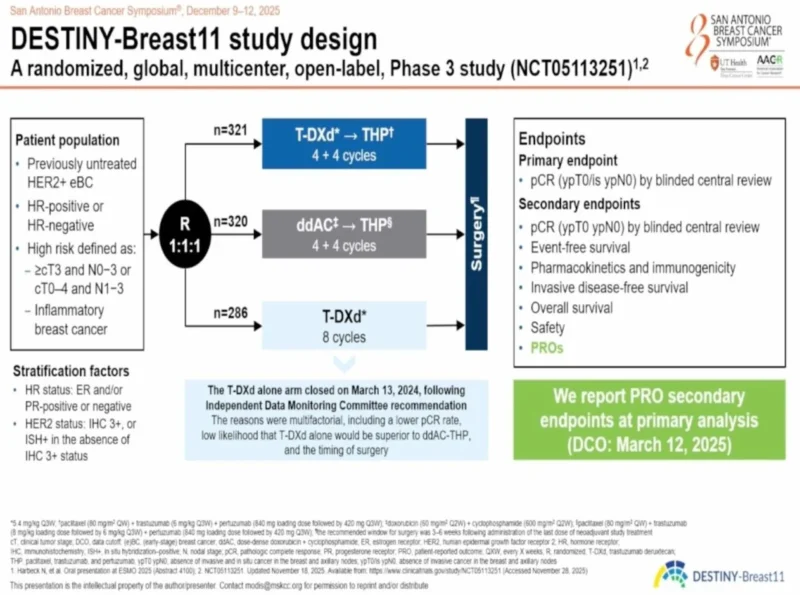
“Trastuzumab Deruxtecan is now FDA approved for 2 new indications in Her2 positive breast cancer: DestinyBreast11 (in NeoAdjuvant) and DestinyBreast05 (in adjuvant high risk residual)!
- pCR and iDFS with TDXd
- Timings of RT did NOT impact the incidence or severity of ILD
- New Soc for high risk residual disease. Are you going to use this broadly in neoadjuvant settings if meets inclusion criteria?”
FDA Approves T-DXd for Two Early HER2-Positive Breast Cancer Settings



