Maria Hafez, Assistant Professor at St. Luke’s University Health Network, shared a post on LinkedIn:
“Breast Cancer Monday Food for Thought
70%!
That’s the proportion of patients walking into my clinic with metastatic triple-negative breast cancer who are not candidates for immunotherapy, and for whom the 1L standard has not meaningfully moved in over a decade.
TROPION-Breast02 may have just changed that.
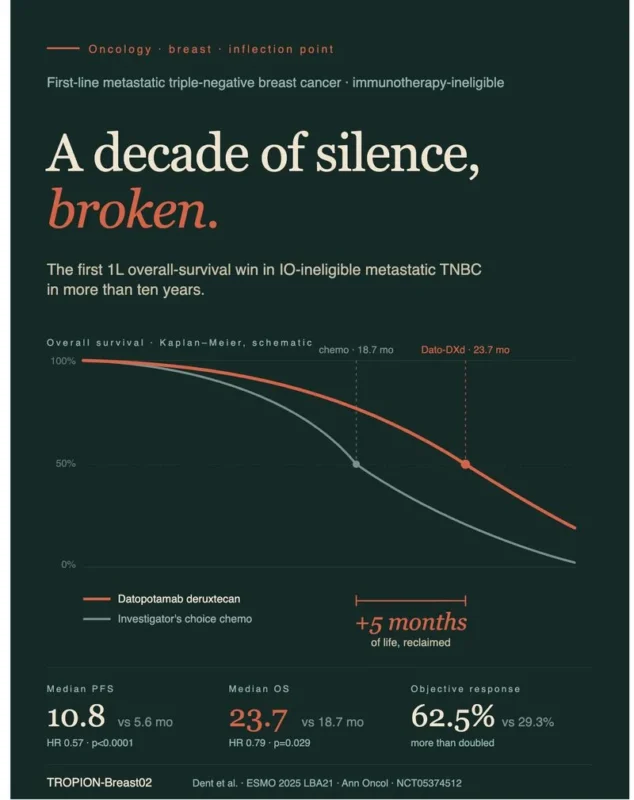
Phase 3 | N=644 | 1L IO-ineligible metastatic TNBC
Datopotamab deruxtecan (Dato-DXd) vs investigator’s choice chemotherapy
Dent et al., ESMO 2025 · Annals of Oncology
- Median PFS: 10.8 vs 5.6 months (HR 0.57)
- Median OS: 23.7 vs 18.7 months (HR 0.79) the first 1L OS signal in this population in over 10 years
- ORR: 62.5% vs 29.3%
- Duration of response nearly doubled: 12.3 vs 7.1 months
- Fewer discontinuations for toxicity on Dato-DXd than on chemotherapy
And the trial enrolled the patients we usually don’t see in registration studies: stable brain metastases, short disease-free intervals, and the sickest among the IO-ineligible.
What this means clinically:
- This is a replacement of standard chemotherapy, not a softer alternative. The comparator arm was the current SOC.
- The sequencing question is now urgent. ASCENT-03 showed a similar PFS signal to that of sacituzumab govitecan in the same population. Two TROP2-directed ADCs, one 1L slot. How we sequence post-progression — and whether cross-ADC activity holds is the next fight.
- Toxicity literacy has to catch up. Stomatitis, dry eye, and ILD will replace neutropenia and diarrhea as the AEs our teams manage most. Pre-treatment ophthalmology and dental baselines become reasonable.
- For our colleagues in LMICs and resource-constrained settings where IO access is already limited, a 1L regimen with an OS benefit in this exact population is not a marginal gain. It is the story of the year.
FDA granted Priority Review in February 2026. Approval is expected.
Food for thought for the breast oncology community this week.
What’s your take: new 1L standard, or do you want to see the real-world toxicity data first?”
SONIA Trial Challenges Routine First-Line CDK4/6 Use in Advanced Breast Cancer



