Mali Barbi, Medical Oncologist at Northwell Health, shared a post on X:
“AACR26 | Gyn Onc
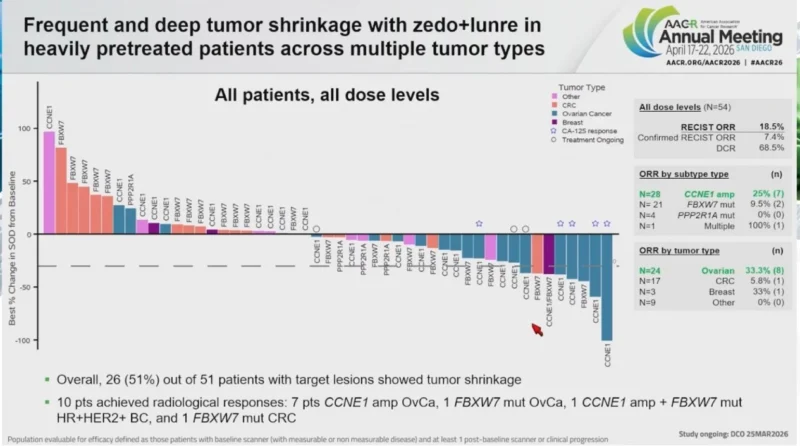
MYTHIC trial (CT022) – first-in-human data for WEE1i zedoresertib + PKMYT1i lunresertib in genomically selected solid tumors (CCNE1 amp, FBXW7 mut, PPP2R1A mut).
These alterations drive replication stress and have No approved targeted therapy – 24.5% of ovarian, 18.4% of uterine cancers. In resistant/refractory ovarian cancer at the lead dose:
- 50% ORR, 83% DCR
- 60% ORR in CCNE1-amp
- 67% ctDNA molecular response rate
- Responses after mirvetuximab, T-DXd, T-DM1
- No myelosuppression.
Zero discontinuations.
First clinical proof-of-concept for synthetic lethality in this space. FDA Fast Track granted.”
Other Articles Featuring Mali Barbi on OncoDaily.


