Julian Chavarriaga, Urologic Oncologist at Penn State Urology, shared a post on X:
“AUA26 Bayesian reanalysis of 8 phase III mHSPC trials presented by Dr. Yoshida:
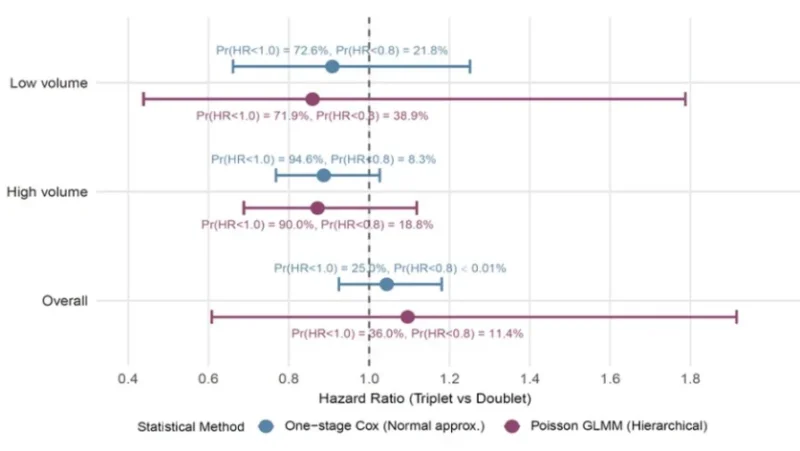
- ARASENS, PEACE-1, ARANOTE, TITAN, ENZAMET, ARCHES, STAMPEDE, and LATITUDE
- Triplet therapy (ADT + docetaxel + ARPI) vs modern doublets
- No clear OS advantage overall — (HR 1.04)
- Probability of ANY OS benefit: 36%
- Probability of clinically meaningful benefit (HR<0.8): only 11%
Signal may exist in high-volume disease… but perhaps less than many expected.”
To which Charles Jiang, Medical Oncologist at UT Southwestern Medical Center, added:
“Understand the motive, but….
These trials across different eras, populations, volume def, control arms, sequencing, and subsequent therapy — without biomarkers or individual-level data.
And if the target is “triplet for everyone,” that is already not how many of us practice…”
Charles Jiang also added:
“The bigger question: why don’t we have patient-level data from these landmark, practice-changing trials?
With a real open-data mandate for top journals, we might have answered “who truly benefits from triplet ?” years ago — instead of trying the answer via indirect reanalyses”
Other articles featuring Julian Chavarriaga and Charles Jiang on OncoDaily.


