James P. Crowley, Professor of Medicine Emeritus at Brown University, shared George Kumar, Senior Director at AstraZeneca, on LinkedIn, adding:
We don’t just need to ask
“Does a T cell have enough of a nutrient?”
— We need to ask
“Where is that nutrient going?”
Controlling intracellular metabolite routing could be a powerful new strategy for boosting cancer immunotherapy.
A team of scientists from Johns Hopkins, spanning the Kimmel Cancer Center, the Bloomberg~Kimmel Institute for Cancer Immunotherapy, and the Bloomberg School of Public Health, has unveiled a critical metabolic balancing act within our immune system’s most formidable cancer fighters: CD8+ T cells.
Their research, published online in the March 31, 2026, edition of Cell, reveals how the amino acid cysteine is partitioned within T-cells to control two essential, yet competing, functions—proliferation (the ability to multiply) and effector function (the ability to attack and kill cancer cells).”
Quoting George Kumar’s post on LinkedIn:
“One Amino Acid, Two Fates: The Metabolic Switch Behind T Cell Exhaustion
A single amino acid. Two very different fates. And a potential new lever for cancer immunotherapy.
New research reveals that CD8+ T cells — the immune system’s front-line cancer killers — use cysteine in two competing ways:
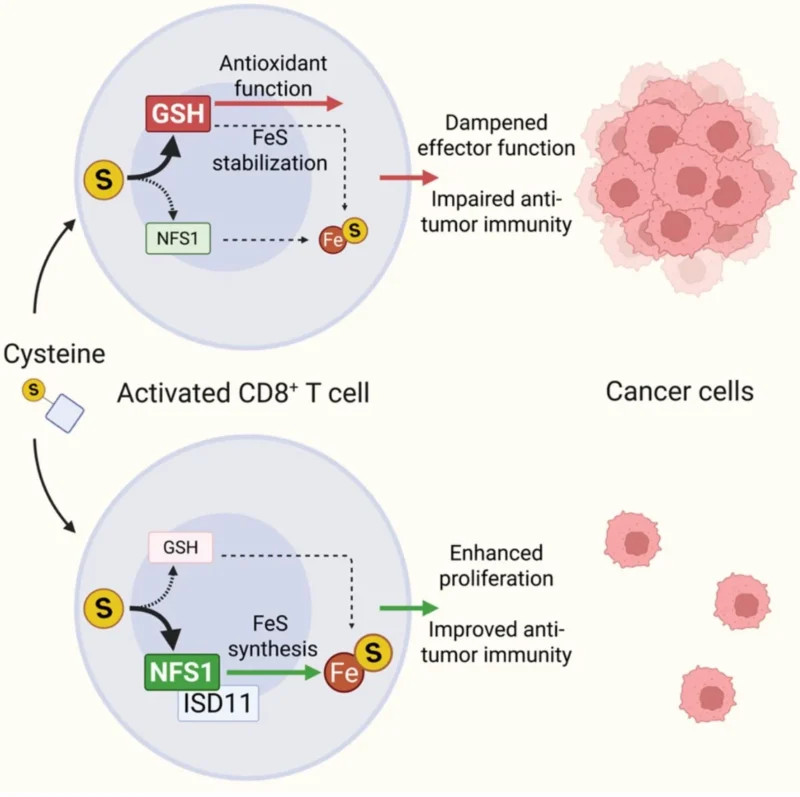
- Into glutathione (GSH): an antioxidant that actually dials down effector function in fully activated T cells
- Into FeS cluster synthesis via NFS1: a pathway that fuels T cell proliferation and sustains anti-tumor activity
When NFS1 is deleted, T cells exhaust faster and lose their ability to fight tumors. But when cysteine flux is redirected away from GSH and toward FeS metabolism, tumor control improves — validated in both mouse models and human hepatocellular carcinoma data.
The broader implication? We don’t just need to ask
“Does a T cell have enough of a nutrient?”
— We need to ask
“Where is that nutrient going?”
Controlling intracellular metabolite routing could be a powerful new strategy for boosting cancer immunotherapy.
Figure courtesy: Cell. Erika L. Pearce. Johns Hopkins University School of Medicine, Baltimore, MD, USA”
Title: Sulfur partitioning from cysteine controls T cell proliferation and effector function
Authors: Beth Kelly, Minsun Cha, Tatjana Gremelspacher, Jacob L. Martin, Massimo Andreis, Isha Maloo, Gustavo E. Carrizo, Mia Gidley, Michal A. Stanczak, Petya Apostolov, Joseph Longo, Lisa M. DeCamp, Eric H. Ma, Ryan D. Sheldon, Russell G. Jones, David E. Sanin, Ananya Majumdar, Erika L. Pearce.
Other articles featuring James P. Crowley and George Kumar on OncoDaily.


