Aakash Desai, Associate Director, Phase 1 and Precision Oncology Program at UAB O’Neal Comprehensive Cancer Center, shared a post on LinkedIn:
“American Society of Clinical Oncology (ASCO) 2026 is around the corner! And I have to say developmental therapeutics has never looked like this.
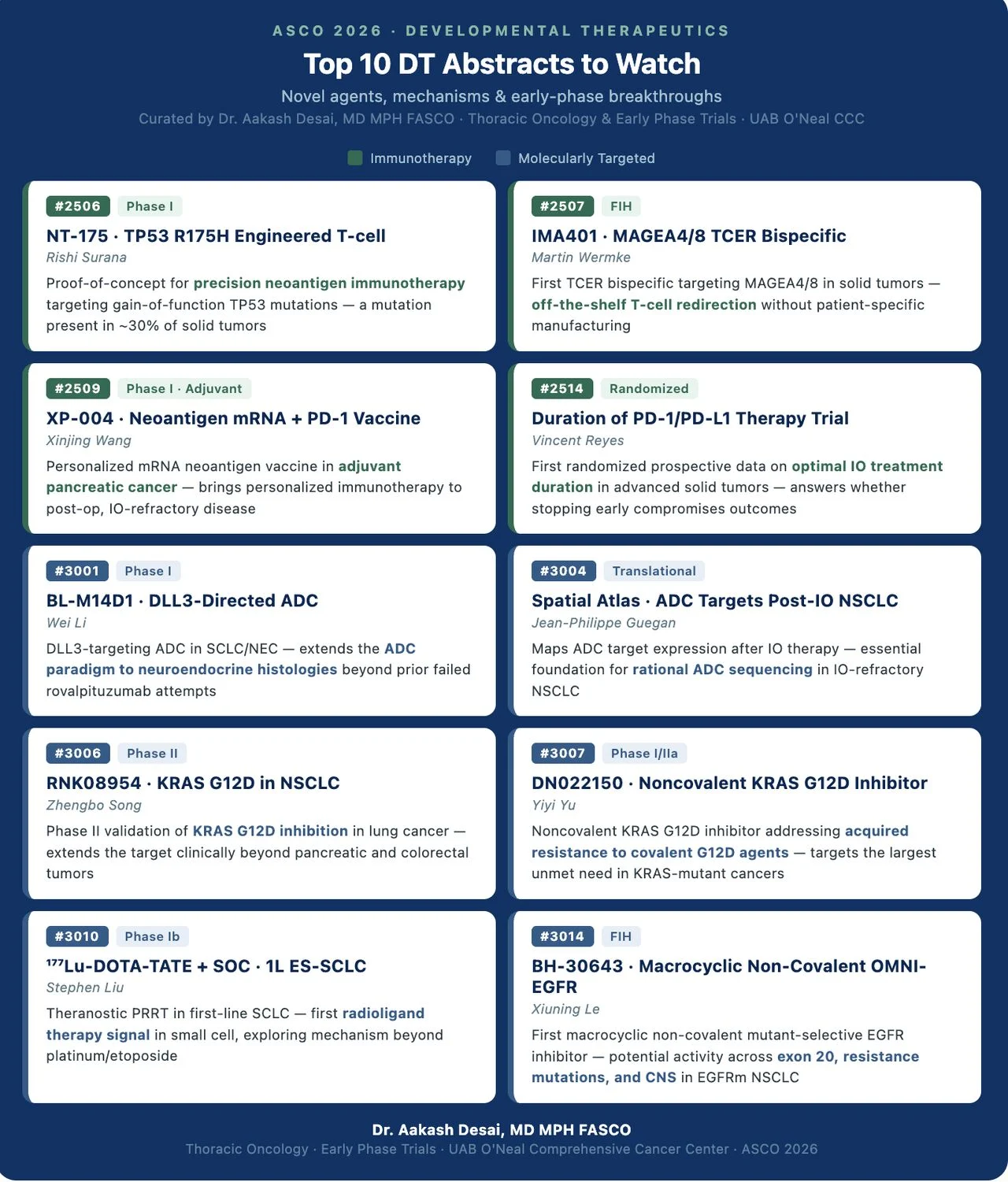
Here are my Top 10 DT abstracts: from a completely new class of EGFR inhibitor to radioligand therapy entering front-line small cell lung cancer.
The ones I’m watching most closely as a Phase I investigator:
- BH-30643 (N3014, Xiuning Le): First macrocyclic non-covalent OMNI-EGFR inhibitor, FIH. This is a mechanistically distinct molecule from prior TKIs. If the early signal holds, it could address exon 20, acquired resistance, and CNS disease simultaneously. BlossomHill Therapeutics, Inc.
- Two KRAS G12D agents in parallel (N3007 + N3006): Noncovalent DN022150 entering Phase I/IIa alongside Phase II data for RNK08954. KRAS G12D is the new EGFR. Watching two distinct binding approaches in the clinic at the same time is exactly how this field moves. Ranok Therapeutics
- 177Lu-DOTA-TATE in 1L ES-SCLC (N3010, Stephen Liu): Radioligand therapy moving to small cell. The theranostic approach is elegant. The key question is whether SSTR expression in SCLC is consistent enough to matter at scale. Novartis Advanced Accelerator Applications
- IMA401 TCER bispecific (N2507, Martin Wermke): Off-the-shelf T-cell redirection targeting MAGEA4/8. No patient-specific manufacturing. Immatics
As someone running early-phase trials, the density of genuinely novel mechanisms in this year’s developmental therapeutics session is striking. These aren’t incremental iterations- they are first-in-class.
Which DT abstract are you following most closely this American Society of Clinical Oncology (ASCO)?”
More posts featuring Aakash Desai.


