Patients with advanced EGFR-mutated non-small cell lung cancer (NSCLC) face limited treatment options after progression on EGFR tyrosine kinase inhibitors (TKIs) and platinum-based chemotherapy. Outcomes in this setting remain poor, and standard therapies such as docetaxel offer modest benefit.
New data from the randomized OptiTROP-Lung03 trial introduce sacituzumab tirumotecan (sac-TMT), a next-generation TROP2-targeting antibody–drug conjugate (ADC), as a promising alternative with meaningful survival advantages.
 Background
Background
Sacituzumab tirumotecan is designed to deliver cytotoxic payloads directly to tumor cells expressing trophoblast cell surface antigen 2 (TROP2), using a bifunctional linker that enhances drug delivery and stability.
Earlier analyses of OptiTROP-Lung03 already demonstrated significant improvements in progression-free survival (PFS) and overall survival (OS) compared with docetaxel. This final analysis provides mature OS data along with updated efficacy and safety outcomes.
Methods
This randomized phase III trial enrolled patients with advanced EGFR-mutated NSCLC who had progressed after both EGFR-targeted therapy and platinum-based chemotherapy.
Participants were randomized in a 2:1 ratio to receive:
- Sacituzumab tirumotecan at 5 mg/kg every 2 weeks
- Docetaxel at 75 mg/m² every 3 weeks
Patients progressing on docetaxel were allowed to cross over to sac-TMT, and overall survival was adjusted using a rank-preserving structural failure time (RPSFT) model to account for this crossover.

Results
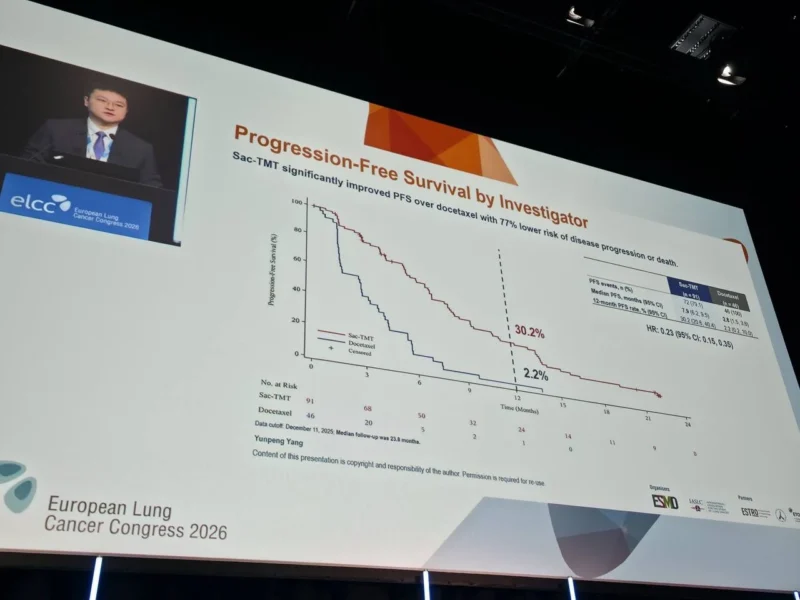
Progression-Free Survival
With a median follow-up of 23.8 months, sac-TMT demonstrated a striking improvement in disease control:
- Median PFS reached 7.9 months with sac-TMT compared to 2.8 months with docetaxel, corresponding to a hazard ratio of 0.23.
This reflects a 77% reduction in the risk of progression or death, highlighting the strong activity of this ADC in a heavily pretreated population.
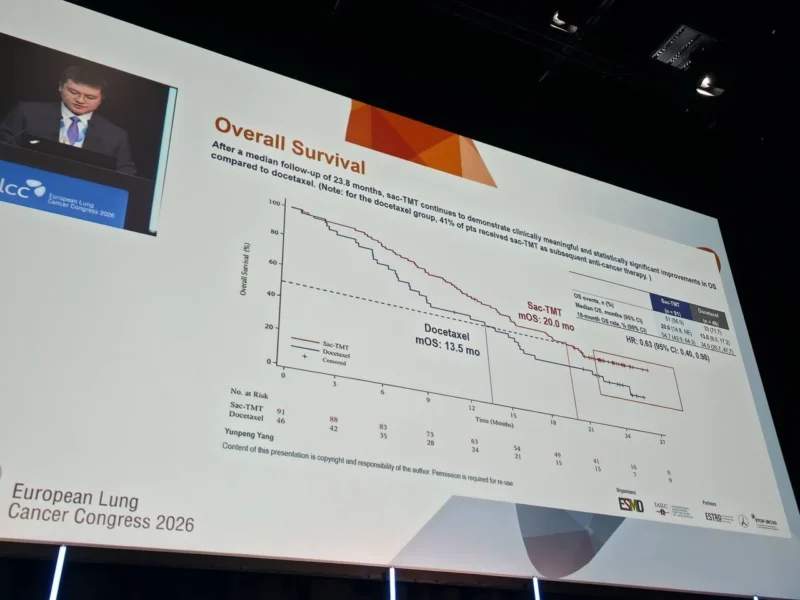
Overall Survival
The final overall survival analysis confirmed a clinically meaningful benefit:
- Median OS was 20.0 months with sac-TMT
- Median OS was 13.5 months with docetaxel
- Hazard ratio: 0.63
Given that more than 40% of patients in the docetaxel arm crossed over to receive sac-TMT, an adjusted analysis was performed.
After adjusting for crossover:
- Median OS remained 20.0 months with sac-TMT
- Reduced to 11.2 months in the docetaxel arm
- Adjusted hazard ratio: 0.45
The 18-month OS rate was 54.7% with sac-TMT versus only 9.1% with docetaxel, underscoring the magnitude of long-term benefit.
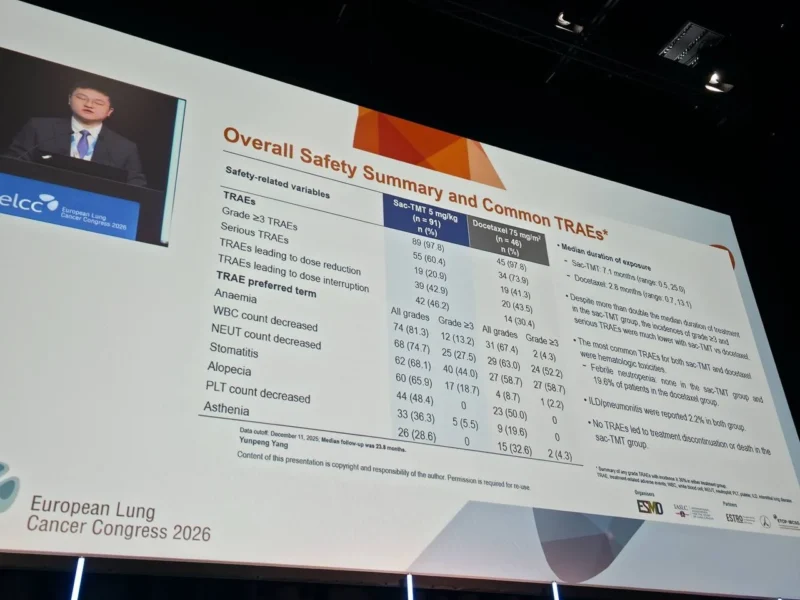
Safety Profile
Sacituzumab tirumotecan demonstrated a manageable and favorable safety profile compared to chemotherapy:
- Grade ≥3 treatment-related adverse events: 60.4% vs 73.9%
- Serious treatment-related adverse events: 20.9% vs 41.3%
Importantly, no cases of interstitial lung disease (ILD) were reported in the sac-TMT group, a key consideration in lung cancer therapies.
Overall, toxicity appeared consistent with expectations for ADCs and was lower than with docetaxel in several key categories.
Clinical Interpretation
These results establish sacituzumab tirumotecan as a highly active therapy in EGFR-mutant NSCLC after standard treatments.
The magnitude of benefit is particularly notable:
- A substantial improvement in both PFS and OS
- Durable survival advantage despite crossover
- A favorable safety profile compared to chemotherapy
In a treatment landscape where options are limited after TKI resistance, sac-TMT offers a new mechanism of action that bypasses traditional resistance pathways.
Implications for Practice
The OptiTROP-Lung03 trial supports a potential shift in the treatment paradigm for EGFR-mutated NSCLC.
Sacituzumab tirumotecan may:
- Replace docetaxel as a preferred option in later lines
- Expand the role of ADCs in lung cancer
- Provide a bridge between targeted therapy and immunotherapy strategies
As ADCs continue to gain traction across oncology, this study highlights their growing relevance in molecularly defined lung cancer populations.
Conclusion
The final analysis of OptiTROP-Lung03 confirms that sacituzumab tirumotecan significantly improves survival outcomes compared with docetaxel in previously treated EGFR-mutant NSCLC.
With meaningful gains in progression-free and overall survival, along with a manageable safety profile, sac-TMT emerges as a compelling new treatment option in a setting of high unmet need.
These findings mark an important step forward in expanding therapeutic strategies beyond TKIs and chemotherapy, reinforcing the role of antibody–drug conjugates in modern lung cancer care.
You can read full abstract here